Lecture 32
advertisement

Lon-Capa
8th (final) HW assignment due Tuesday,
December 2 by 5 pm.
Quiz #6 opens Friday at 5 pm. Due Tuesday,
December 2 by 10 pm.
Exams and Practice Exams on website
1
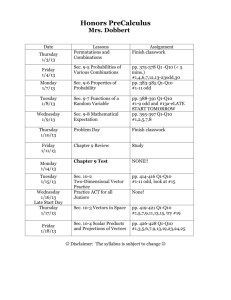
Clicker Question
Determine the missing
time given the following
orders of reaction for
aA Products
a)
b)
c)
d)
zero 1st
20
20
30
20
15
20
Not sure.
2nd
20
15
30
[A]
Time
1.00 M
0
0.500 M
10 sec
0.250 M
?? sec
2
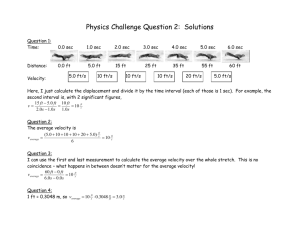
Chapter 15: 102
The following results were obtained
at 600K for the decomposition of
ethanol on an alumina (Al2O3)
surface,
C2H5OH(g) C2H4(g) + H2O(g)
a. Predict Ptotal in torr at t = 80. s
b. What is the value of the rate
constant, and what are the units?
c. What is the order of the reaction?
d. Calculate Ptotal at t = 300. s
Time (s)
0
Ptotal
(torr)
250.
10.
265
20.
280.
30.
295
40.
310.
50.
325
3
aA + bB Products
Given the following data, determine the rate
law and the value for the rate constant.
Expt 1 {[B] = 5.00 M}
Expt 2 {[B] = 10.0 M}
[A]
0.010 M
0.0050 M
0.0025 M
[A]
0.010 M
0.0050 M
0.0025 M
time
0
40. sec
120 sec.
time
0
20. sec
60. sec
4
Mechanisms
Series of elementary steps.
Must be chemically reasonable
Must have the correct stoichiometry. That is,
the steps must add up to the overall reaction.
How can we tell this?
Cannot prove but can support empirically.
Must agree with the overall rate law.
5
Rate Laws From Elementary
Steps
6
Evaluating Mechanisms
The rate law comes from the ratedetermining step (the “slow” step).
Fast equilibrium.
Rate forward = rate reverse.
Rate law cannot include intermediates.
Steady-state approximation.
[intermediate] = constant.
d[intermediate]/dt = 0
Rate law cannot include intermediates.
7
Chapter 15: 115
Do for next Friday.
8