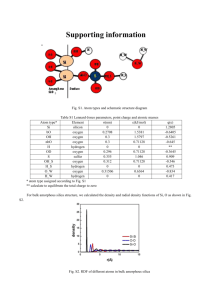
1. Synthetic route of dendrimer-like PAMAM polymer
advertisement

Supplementary Information Ionic liquid-assisted desorption of DNA from polyamidoamine-grafted silica nanoparticle surface by low-salt solution Nurgul Latep, Xiao Liang, Weidong Qin* 1. Synthetic route of dendrimer-like PAMAM polymer-grafted silica nanoparticles Fig. S1 Synthetic route of dendrimer-like PAMAM polymer-grafted silica nanoparticles The PAMAM-grafted silica nanoparticles were prepared according to previous reports [1, 2]: (1) Introduction of amino groups onto the silica nanoparticle surface Before use, the silica nanoparticle (SNPs) were dried in vacuum at 110 °C for 48 h. Then, to a 500-mL flask 5.0 g silica nanoparticle, 50 mL APES and 300 mL toluene were added and refluxed under stirring for 8 h. After reaction, the mixture was centrifuged at 4000 rpm for 20 min, the upper layer solution being discarded. The precipitation was suspended into 300 mL ethanol, then the suspension was ultrasonicated for 30 min 1 followed by centrifugation at 4000 rpm for 20 min. The suspension-precipitation process was repeated five times to remove residual starting materials. The product (SNP-PAMAM G 0) was dried under vacuum at 50 °C over 48 h before further reaction. (2) Synthesis of SNP-PAMAM G 0.5 The product was obtained by Michael addition of MA to amino groups on SNP-PAMAM G 0 surface. In a 500-mL flask, the mixture of 3.0 g SNP-PAMAM G 0, 100 mL methanol and 0.5 mL MA were stirred at 50 °C for 48 h. After reaction, the suspension was centrifuged; the precipitation SNP-PAMAM G 0.5 was washed five times with methanol, and then dried at 50 °C for 48h under vacuum. (3) Synthesis of SNP-PAMAM G 1.0 The product was synthesized via amidation of terminal ester groups of SNP-PAMAM G 0.5 by EDA. To a 500-mL flask containing 3.0 g SNP-PAMAM G 0.5 and 100 mL methanol, 2.2 mL EDA were added. After being stirred at 50 °C for 48 h, the mixture was centrifuged at 4000 rpm for 20 min and the precipitation was washed five times with methanol, dried at 50 °C under vacuum for 48 h. Higher generations of PAMAM-grafted SNPs were obtained through repeating the Michael addition/amidation cycles with proper amounts of MA and EDA. 2. FT-IR characterization Fig. S2 shows the IR spectra of dendrimer-like PAMAM-grafted silica nanoparticles of different generations. SNP-PAMAMs show new absorptions at 2950 cm-1, and 2853 cm-1, which are the characteristic stretching of asymmetric and symmetric –CH2 bands, respectively [3]. The absorption at 1645 cm-1 originates from bending of amide groups. The absorption at 1735 cm-1, suggesting the presence of ester groups (–CO2CH3), is visible in the spectra of all the half-generation products. Based on the above results, it was concluded that dendrimer-like highly branched polymers were 2 grafted on the SNP surface by repeating Michael addition/amidation cycles. Fig. S2 FT-IR spectra of different generation PAMAM-grafted SNPs. 1.silica nanoparticle; 2, SNP-PAMAM G 0; 3, SNP-PAMAM G 0.5; 4, SNP-PAMAM G 1; 5, SNP-PAMAM G 1.5; 6, SNP-PAMAM G 2.0 3. TGA analysis The thermal stability of the SNPs grafted with different generation PAMAMs was investigated by TGA (Figure S3). TGA curves of all the samples exhibit a slight weight loss before 120 °C, which could be attributed to adsorbed water removal from the samples [4]. When the temperature rising from 120 °C to 600 °C, SNP-PAMAMs showed two continuous weight-loss steps. The weight loss between 120 °C and 350 °C corresponded to the decomposition of PAMAM moieties, and the weight loss between 350 °C and 600 °C was predominantly caused by the degradation of the APES-grafted SNP core [5]. The increasing weight loss with higher generation SNP-PAMAMs suggests the growth of hyperbranched PAMAMs on the SNP surface. 3 Fig. S3 TGA curves of SNPs grafted with PAMAMs of different generations: A, G 0; B, G 0.5; C, G 1.0; D, G 1.5; E, G 2.0; F, G 2.5; G, G 3.0. Table S1. The PGs and amino group contents calculated from TGA results %Wt. change Observed Theoretical amino amino content groups (mmol/g) (mmol/g) %Wt. loss compared to Samples Observed Theoretical between the proceeding PG% PG % 120-600 °C generation G0 7.81 8.47 0.673 G 0.5 14.4 6.59 16.82 24.0 G 1.0 15.67 1.27 18.58 30.1 G 1.5 23.15 7.48 30.12 60.6 G 2.0 26.16 3.01 35.43 78.7 G 2.5 33.77 7.61 50.98 92.7 G 3.0 38.28 4.51 62.02 125.5 0.744 1.35 1.383 2.69 2.557 5.38 3.1 The content of surface amino groups [3] 3.1.1 SNP-PAMAM G 0 4 The weight loss of SNP-PAMAM G 0 in the range 120-600 °C was 7.81%. Therefore, taking molecular weight of 116 g/mol for the functional moiety, the content of surface amino group on SNP-PAMAM G 0 is 7.81/100/116×1000=0.673 mmol/g. 3.1.2 SNP-PAMAM G 1.0 The weight loss percentage was 14.4%, which was 6.59% more than that of SNP-PAMAM G 0. This is due to the amino group that underwent Michael addition from G 0. Because the molecular weight difference between G 0 and G 0.5 is 172, the content of amino groups reacted was calculated to be 0.383 mmol/g. Therefore, the amount of unreacted amino groups was 0.673-0.383 = 0.29 mmol/g. Further, the weight loss difference between SNP-PAMAM G 1.0 and SNP-PAMAM G 0.5 is 1.27%. Because the molecular weight of the PAMAM moiety increased by 56 Dalton during the reaction, it can be deduced that 0.454 mmol/g amino group was generated from SNP-PAMAM G 0.5 to SNP-PAMAM G 1.0. Therefore, the ultimate amino group content on the surface of SNP-PAMAM G 1.0 was 0.454+ 0.29 = 0.744 mmol/g. Using the similar calculation protocol, the contents of amino groups on the SNP-PAMAM G 2.0 and SNP-PAMAM G 3.0 were calculated to be 1.383 mmol/g and 2.557 mmol/g, respectively. 3.2 Theoretical amino group contents on the surface of full generation PAMAM grafted-SNPs According to weight loss in TGA analysis, SNP-PAMAM G 0 contains 0.673 mmol/g amino group on the surface after amination process. By assuming each following reaction was finished stoichiometrically completely, it can be inferred that the amino groups of SNP-PAMAM G 1.0 should be 0.673 mmol/g×2 = 1.346 mmol/g; the amino groups of SNP-PAMAM G 2.0 should be 0. 673 mmol/g×4 = 2.692 mmol/g; and the amino groups of SNP-PAMAM G 3.0 should be 0.673 mmol/g×8 = 5.384 mmol/g. 5 3.3 Grafting percentages (%) of different modified nanoparticles The percentage of grafting (PG) of dendrimer on the surface of silica nanoparticles was determined by the following equation [6]: PG(%) =A/B×100 where A is the weight of dendrimer grafted on silica nanoparticles (g) and B the weight of silica nanoparticles charged (g). The weight of dendrimer grafted onto the silica nanoparticles was calculated from the TGA results. The equation can be transfered to the formula: PG (%) = weight loss percentage /(100%-weight loss percentage). The grafting percentages of SNP-PAMAMs were thereby calculated to be 8.47%, 16.82%, 18.58%, 30.12%, 35.43%, 50.98% and 62.02%, respectively, for G 0, G 0.5, G1.0, G 1.5, G 2.0, G 2.5 and G 3.0. 3.4 The theoretical grafting percentage Calculations were based on the assumptions that, after the alkylation of silica surface (SNP-PAMAMs G 0, amino group content 0.673 mmol/g), each subsequent reaction took place stoichiometrically. For example, production of SNP-PAMAM G 0.5 resulted in an increase of 172 Dalton to every amino group; therefore, the mass of organic material grafted was 0.673 × (172+116) /1000 = 0.1938g, and the theoretical grafting percentage is 0.1938 / (1-0.1938) = 24.04%. Similarly, the theoretical grafting percentage for SNP-PAMAMs at G 1.0, 1.5, 2.0, 2.5 and 3.0 are calculated to be 30.1%, 60.6%, 78.7%, 92.7% and 125.5 %, respectively. 4. SNP-PAMAM binding efficiency Preparation of the working solution: 9 μL DL 2000 DNA Marker and 15 μL of PAMAM-grafted SiO2 nanoparticles at 4 g/L were mixed (the molar ratio of DNA to PAMAM was the same as that described in the main text) and treated following the 6 procedures as per in the main text. Afterward, 17 μL of the supernate was added with appropriate amount of EtBr, and diluted with triple distilled water to 2 mL. The concentration of DNA in this solution, if no adsorption occurred during the treatment, should be 255 ng/mL, i.e.: 17 μL × 9 μL ÷ (9 μL + 15 μL) × 80 ng/μL (original DNA concentration) ÷ 2 mL = 255 ng/mL Fig. S4 Fluorescence spectra of DNA standards and post-adsorption supernate Experimental conditions: Excitation wavelength, 510 nm; emission wavelength, 620 nm. EtBr concentrations in blank and each DNA solution were 250 ng/mL. The concentration (ng/mL) in each spectrum: a, 252; b, 200; c, 160; d, 120; e, 80; f, 40; g, post-adsorption supernate; h, 10; i, blank Fig. S5 suggests the good binding ability of the synthesized SNP-PAMAM towards the DNA fragments. The results also indicate that the residual DNA concentration after adsorption was 17.97 ng/mL. Considering the initial concentration of 255ng/mL, the binding efficiency should be 7 255 17.97 100% 92.9% 255 Fig. S5 Calibration curve for the fluorescence measurements 5. The effect of release time Fig. S6 Effect of desorption time The additive in the desorption solution was 4 mM [C6MIM]Br. Other conditions were the same as in Fig. 2C of the main text. 8 6. Effect of release temperature Fig. S7 Dependence of peak area on release temperature The desorption time was 5 min. Other conditions were the same as in Fig. S6. 7. Stability of SNP-PAMAMs in working release buffer (pH 11) Although it was reported that surface modification of silica particles can improve their durability [7], the stability of the SNP-PAMAMs in the release buffer is still a concern because it may influence the recovery. To test its stability, SNP-PAMAM G 2.0 was suspended in the releasing buffer (5 mM phosphate containing 4 mM C6MIMBr at pH 11.0). After 24 h, the suspension was centrifuged at 4000 rpm for 20 min. The precipitate was washed for five times with 30 mL methanol, and then dried under vacuum at 50 °C for 48 h. The release buffer-treated SNP-PAMAM G 2.0 was tested by TGA and was used in DNA adsorption/releasing experiment; for comparison, the normal SNP-PAMAM G 2.0 were also tested and evaluated. 9 Fig. S8 Evaluation of the release buffer (pH 11)-treated and normal SNP-PAMAM G 2.0 TGA curves: A, normal; B, release buffer-treated. The inset: electropherograms of DNA standards prepared in release solution (a) and DNA fragments that underwent adsorption/release processes using the treated (b) and original (c). The TGA results (Fig. S8) suggests that the organic contents are similar for the normal and treated SNP-PAMAM G 2.0, with weight lose percentage between 120 – 600 °C being 26.16% and 26.83% for normal and treated SNP-cored PAMAMs respectively. Moreover, the electropherograms of DNA underwent adsorption/release process using the normal and the treated SNP-PAMAMs were highly similar, the mean combined recoveries of DNA fragments using the respective nanoparticles were 91.5% and 92.4%. It can be inferred from the above results that the as-prepared SNP-PAMAM G 2.0 was stable in the release buffer at pH 11. 8. Stability of SNP-PAMAM G 2.0 in release buffer at higher pH (pH 12) The buffer used for treatment: 5 mM phosphate and 4 mM [C6MIM]Br at pH 12. The SNP-PAMAM G 2.0 was suspended in buffer at pH 12 and stored for 12 h or 24 h. Afterward, the nanoparticle was used for adsorbing DNA fragments, which was desorbed with the optimum release buffer (pH 11) as stated in the main text, and analyzed with CE-UV. For comparison, the normal SNP-PAMAM G 2.0 was also evaluated, and a DNA standard solution analyzed. It was determined that the recovery 10 with 12-h treated SNP-PAMAM G 2.0 was 91.6%, similar to the value of 92.0% with normal one. However, the value with 24-h treated SNP-PAMAM G 2.0 was 74.8%, suggesting the slow degradation of the outer PAMAM moieties. However, due to the short releasing time of ca. 10 min, such degradation should not be the main contributor to the low recovery as shown in the main text (Fig. 3A). Fig. S9 Evaluation of the buffer (pH 12)-treated and normal SNP-PAMAM G 2.0 A, SNP-PAMAM G 2.0 treated for 12 h; B, DNA standard prepared in release solution; C, normal SNP-PAMAM G 2.0; D, SNP-PAMAM G 2.0 treated for 24 h. 9. Effect of release buffer on the electropherogram In our experiments, the separation buffer comprised 44.5 mM Tris, 44.5 mM boric acid, 1 mM EDTA and 3% PVP at pH 11. It was reported that EDTA was UV-active at 254 nm [8]; therefore, EDTA anion could act as background chromophore for the anions in the release buffer, i.e., bromide (from [C6MIM]Br), phosphate and carbonate (from the dissolved carbon dioxide). It can be found from Fig. S10 that the peak profiles of the release buffer components (peaks 1, 2 and 3) were similar in panels A and B. However, probably due to the high-concentration carbonate presented in panel B, the peak of this anion broadened. Fortunately, the peaks of these inorganic anions did not interfere with the detection and quantification of the DNA fragments. 11 Fig. S10 Influence of release buffer on the electropherograms A, Electropherograms of the release buffer constituents. a, 4 mM [C6MIM]Br; b, 5 mM phosphate at pH 11; c, the mixture of 4 mM [C6MIM]Br and 5 mM phosphate. B, Fig. 4 of the main text. The running buffer: 44.5 mM Tris, 44.5 mM boric acid, 1 mM EDTA and 3% PVP at pH 11. Peak identities: 1, bromide; 2, phosphate; 3, carbonate. Other conditions were the same as in Fig. S9. 10. Mean combined recovery and precision of the method The precision of the recovery for the process of SNP-PAMAM-initiated adsorption/IL-assisted desorption was evaluated over three days, with 5 experiments every day. 12 Table S2 Mean combined recovery and precision of the method Intra-day Mean recovery (%) Intra-day precision average 1# 2# 3# 4# 5# (RSD%, n=5) (n=5) Day 1 91.99 92.83 91.99 92.90 91.50 99.292 0.67 Day 2 92.18 91.05 85.96 90.48 93.18 97.494 3.073 Day 3 92.99 92.37 86.75 92.05 88.81 97.518 2.978 98.1 2.984 Inter-day precision(RSD%, 3 days, n=3×5=15) References [1] Qu, R. J., Niu, Y. Z., Sun, C. M., Ji, C. N., Wang, C. H., Cheng, G. X., Microporous Mesoporous Mater. 2006, 97, 58-65. [2] Wu, X. Z., Liu, P., Pu, Q. S., Sun, Q. Y., Su, Z. X., Talanta 2004, 62, 918-923. [3] Gao, J. J., Latep, N., Ge, Y., Tian, J., Wu, J. Q., Qin, W. D., J. Sep. Sci. 2013, 36, 1575-1581. [4] Neelgund, G. M., Oki, A., Appl Catal B-Environ 2011, 110, 99-107. [5] Jin, L., Liu, H. B., Yang, W. L., Wang, C. C., Yu, K., J. Polym. Sci., Part A: Polym. Chem. 2008, 46, 2948-2959. [6] Tsubokawa, N., Ichioka, H., Satoh, T., Hayashi, S., Fujiki, K., React Funct Polym 1998, 37, 75-82. [7] Qu, Q., Peng, S., Mangelings, D., Hu, X., Yan, C., Electrophoresis 2010, 31, 556-562. [8] Vassil, A. D., Kapulnik, Y., Raskin, I., Salt, D. E., Plant Physiol. 1998, 117, 447-453. 13