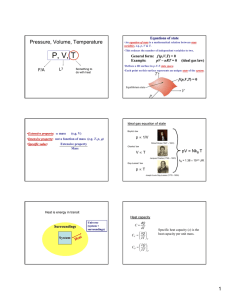
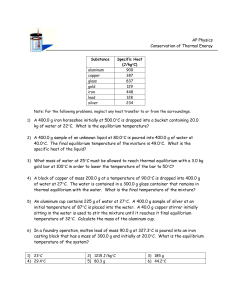
Thermal Equilibrium Lab
advertisement

If a section is highlighted, it needs your attention. Do not turn in your lab with anything highlighted! Names 1. Thermal Equilibrium 2. Purpose-The purpose of this lab is… Questions: a. What is the direction of energy flow when you put two objects of different temperatures together? b. What is the temperature of thermal equilibrium for room temperature water and one ice cube? Two ice cubes? c. In general, what is the temperature of thermal equilibrium between two objects with differing initial temperatures? 3. Hypothesis (predictions) a. I predict that… b. I predict that… c. I predict that… 4. Materials: use bullet points 5. Background: Write anything here that someone would need to know to understand your lab (definitions, etc) 6. Procedure 1. 2. 3. 4. 5. 6. 7. 8. 9. 7. Data Table-Complete the data tables. Initial Volume of Water (mL) Initial Temperature of Water (C◦) Initial Volume of Ice (mL) Initial Thermal Time to Temperature Equilibrium Achieve Thermal of Ice (C◦) (C◦) Equilibrium Final Volume of Water (mL) Final Volume of Ice (mL) Observations-Make your own observations about temperature, thermal equilibrium, or energy flow. Make sure you record in bullet points or in a table. 6. Results (graph here) Click on insert, picture, and chart. Format the cells to reflect the above data. Make sure that your graph makes sense and is presented appropriately in terms of the lab questions. 7. Conclusion-FOLLOW THE RERUN FORMAT and address the questions. Go beyond the basics (the ice cube melted). When you are finished, post your lab on your UNIT 4 webpage. Also, remember to post your conclusion on nicenet. No paper needed!