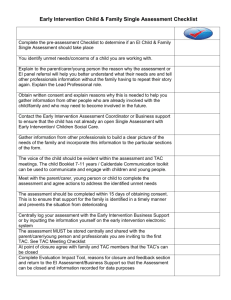
ATW - Michigan Manufacturers Association
advertisement

DRAFT Air Toxics Workgroup (ATW) Meeting Summary September 25, 2013 Members Present: James Clift, MI Environmental Council Steve Kohl, Warner Norcross & Judd Andy Such, MMA for John Caudell, Fishbeck Thompson Carr & Huber Stuart Batterman, UM Brad Venman, NTH Bob Sills, AQD Joy Taylor Morgan, AQD, Facilitator Kory Groetsch, MDCH Greg Ryan, DTE Energy Kim Essenmacher, GM Carrie Houtman, Dow Chemical Company Mark Mitchell, Permit Unit Supervisor for Mary Ann Dolehanty, AQD Members Absent: Mary Ann Dolehanty, DEQ and John Caudell, Fishbeck Thompson Carr & Huber Guests/Observers Present: Dave Fiedler, Regulatory Affairs Officer, DEQ; Mike Depa, AQD; Breanna Bukowski, SIP Unit; Chris Flaga, Toxics Unit Supervisor of Remediation and Redevelopment Division. The meeting began with the Facilitator informing the Members that she will be finalizing meeting number eight and nine and these summaries will be placed on the ATW web site. She noted that Andy Such was in attendance for John Caudell and he will be voting in his place. She mentioned that the Division is coordinating rule revision efforts with the exemption workgroup and the rule changes will be moved along together. The Division will submit two RFRs (request for rule making), one for Part 1 (definition changes) and another for Part 2. Only one rules package under each Part can be submitted at a time. There is a current Part 2 rules set (2012107EQ) that is currently at JCAR and once it is filed at Secretary of State (expected October 25, 2013), then the next RFR can be submitted. She mentioned that there will be a public hearing for both and there will be a 30 day public comment period. Once the process begins, the rules are expected to be finalized within a year. A-1(7) Consistent with Nearby States The Facilitator began discussing A-1(7) to clarify the degree of support and non-support, since some Members were not entirely supportive of the proposal and draft rule language. Removal of the default ITSL was not supported by two of the Members. Some Members stressed that with the removal of a default ITSL and with a defined TAC list that it is critical that an internal procedure be followed that allows toxicologist review of non-TACs. A question was asked, what happens when there is no ITSL? The response was that an ITSL can be set based on toxicity data for the substance or supplemental data for similar substances and the application of computational toxicology, but the approach must be defensible. Two Members stated that a back-stop is needed for all non-TACs. One Member stated that the burden of evaluating nonTACs is now placed on the Department, which the Member did not think was good. Unfortunately unanimous agreement was not reached, and the Facilitator stated that this will be noted in the final report with a mention of the importance of developing an effective internal policy on non-TAC reviews. The following revisions will be made in the final report: 1 Degree of Consensus with this ATW Recommendation: ATW discussions indicated that most Members support all of the recommended changes, although two Members disagreed with the proposed elimination of the default-based ITSLs and with the burden shifting to the Department for evaluating non-TACs. Those two Members supported the current AQD policy and rule for setting a default ITSL for air toxics with inadequate toxicological data. A-1(9) Rule 228 The Facilitator then moved the discussion along to Rule 228. She gave a quick overview of the draft rule language (see below) and mentioned that one of the Members submitted proposed alternative (see below) which was discussed. The proposed alternative was to make Rule 228(2) less comprehensive by limiting the applicability to only those non-TACs that the agency proposes to add to the TAC list. The proposed alternative also refers to the conduct of the evaluation as described in Rule 228(1). Draft Rule 228(2) The department may determine on a case-by-case basis that an emission rate limitation is needed for a non-TAC air contaminant to provide adequate protection of human health and the environment. The department shall establish this emission rate consistent with the provisions of Rule 225, Rule 227, and Rule 229 or any other methodology determined by the department to be more appropriate considering all relevant scientific data. Proposed alternative language for Rule 228(2): (2) The department may determine on a case-by-case basis that an emission rate limitation is needed for a non-TAC air contaminant proposed to be regulated as a TAC under Rule 230(3) to provide adequate protection of human health and the environment. The department shall establish this emission rate consistent with the provisions of Rule 225, Rule 227, and Rule 229 or any other methodology determined by the department to be more appropriate after an evaluation conducted under Rule 228(1). The Facilitator stated that the Department was tentatively accepting of the proposed alternative, pending discussion and input from the Workgroup. Three Members were concerned that under the proposed alternative there would be no clear back-stop for all non-TACs. An example was given that while fluorocarbons are extremely toxic to birds, they are not listed as TACs and do not meet TAC criteria. Another concern was for high emissions of non-TACs. Staff noted that Rule 901(a) could serve as the back-stop in these cases, however, some Members felt that Rule 901(a) was not an effective and sufficient back-stop and that if the Rule language is revised then Rule 228(2) should either repeat the Rule 901(a) language or cite Rule 901(a). A concern was brought up that since the AQD’s Air Advisory Council has asked for a curtailment of the use of Rule 901 in permitting, the ATW should not rely on Rule 901(a) as a back-stop for the effects of air toxics. One Member thought that as written Rule 228(2) could limit criteria pollutants. Staff stated that was not the intent. The Facilitator asked if it would suffice to explicitly exclude criteria pollutants, but some Members still had a concern that the Rule 228(2) language could still bring back into regulation all chemicals that have dropped off the TAC list. There was discussion regarding bringing the Rule 901 language into Rule 228 as back-stop language. A couple Members thought this was redundant so there was discussion regarding possibly revising the recent Rule 901 Policy and Procedure to address Rule 228. A Member stated they were not comfortable with the change to the draft Rule 228(2) language since this 2 was intended to be the back–stop for all non-TACs, and Rule 901 is vague and has not been very effective. It was decided that staff would discuss with AQD management a potential revision to the Rule 901 Policy and Procedure. However, 33% (3/9) of the Members were not in agreement with the proposed revision to the draft Rule 228(2) language. A-1(2) Meaningful Change The Facilitator then began the discussion on A-1(2). It was clarified that this exemption cannot be used if the change would cause a violation of a permit limit. The baseline issue was discussed. The baseline is established in a permit application and review that occurred since the air toxics rules were promulgated in 1992. There are separate baselines for carcinogens and noncarcinogens. Even if there has been a change in a screening level, the established baseline is still valid and should still be utilized, as described in the examples in the discussion paper. Changes to the draft report’s discussion of the degree of consensus were discussed. One Member stated that when a company uses the exemption, the Department should be notified. Several Members stated that they do already notify the Department voluntarily, and that this may not be too difficult to implement. However, AQD staff contacted district staff after the meeting and the district staff stated that they address the use of exemptions at the time of performing routine inspections, and do not want required notifications which suggest that a pseudo-permit review would be conducted. Another Member stated that in the report, the wording should be changed from, “..the Department should develop a Policy and Procedure…” to “….the Department will develop a Policy and Procedure…”. The Facilitator then took a poll of the Members utilizing the gradients of agreement tool. Seven members endorsed the A-1(2) rule changes with one agreeing with reservation and one having mixed feelings. Based on the discussion, the draft report will be revised as follows: Degree of Consensus with this ATW Recommendation: There was consensus agreement, but not unanimous support. Representatives of the regulated community supported the recommendation, but some other Members were not supportive due to concerns that the exemption definitions and procedure do not address potential interactive effects of air toxics, do not account for non-TACs, do not account for cumulative impacts, and allow up to a 10% increase. A-1(6) TAC List The Facilitator then began a discussion on the TAC list issue. She mentioned that there were a few suggested changes to the language that was included in the draft final report. Some Members and MMA requested that the current rule’s list of 41 non-TACs also be included in the proposed rule as exemptions. A Member asked if the agency had concerns with any of those substances continuing to be explicitly exempted from the TAC definition; staff said that they did not. There was also discussion on further clarifying the exemption of “nuisance particulates”, expanding the listing of “vegetable oil mist” to include plant and animal oils, and/or adding an additional exemption addressing food manufacturing. A Member did not agree with clarifying 3 the “nuisance particulate” exemption by stating that it only included human food, because that would narrow it. One company that was interested in adding a food related exemption was the Amway Company, as they are currently looking at moving a company from California to Michigan and they believe that a new, broader exemption for human food and food supplements is warranted. The group considered the draft new exemption proposed by a Member and Amway: “Animal or plant derived ingredients used in a process whose products are intended for human consumption.” Staff noted that more input was needed by AQD staff and management, and that there could be some concerns with the scope of inclusion. It was agreed that this topic will be mentioned in the Final Report and the agency will consider broadening the TAC exemptions as appropriate to address the food exemption issue. Another change that was recommended was changing the schedule for public noticing the changes in screening levels from annually to, at a minimum, semi-annually. It was also recommended to post the list of air contaminants determined by AQD not to be a TAC. The AQD has agreed with these recommendations. The following revised draft language was agreed upon: (4) The department may at any time make changes to the screening levels for TACs based on best available information. The department shall, at a minimum, semi-annually provide public notice of changes to any TAC screening levels that have occurred over the previous posting and accept comments thereon for 30 days and thereafter post on the department website the modification or a decision to not make a modification and a response to substantive comments received. (5) The department shall maintain on the department website a list of air contaminants which it has determined not to regulate as a toxic air contaminant based on a screening level review. The Members agreed with Rule 230(5) above, and the change to Rule 230(4). Two of the Members wanted it noted in the Final Report that they do not support the use of the 75th percentile values as cutoffs for ITSLs for the TAC list. A Member said that if the Rule 228(2) language is changed so that it is not a back-stop to address all non-TACs, then AQD staff needs to change that discussion in the draft final report (on p. 14). The Facilitator then took a poll of the Members utilizing the gradients of agreement tool. Five Members endorsed the A-1(6) draft rule changes; one Member agreed with reservation; one Member had mixed feelings (since the sources subject to a MACT were not being exempted); one Member disagreed (because the agency would not be able to sufficiently address all chemicals that pose a risk); and, one Member strongly disagreed (they wanted the approach currently in place to be retained, with no defined TAC list, and with use of default ITSLs). A-1(5) Pollution Control Projects The Facilitator presented the language that is included in the Final Report to make sure that the Members were in agreement with the draft language. The draft language in the report reads: “ATW Recommendation: Considering the significant regulatory streamlining steps being recommended by the ATW elsewhere in this Report, and those currently under development by the AQD’s Permit Exemptions Workgroup, the ATW does not recommend additional specific pollution control projects for exemptions to provide significant additional streamlining.” 4 The Members’ agreement with this language was unanimous. Other Issues The Facilitator asked the Workgroup Members if there were any other air toxics rule issues that they wanted to discuss (as mentioned in the ATW “charge”). One Member said, as previously mentioned, that they would like the non-TAC list to include a food related exemption. Also, there was a question as to why the RRD program utilizes a 10-5 acceptable risk level versus 10-6 in AQD’s risk assessments. Staff explained that the RRD program addresses contaminant levels present in each media, with a 10-5 risk target for each media, which is more analogous to AQD’s SRSL (10-5 for facility-wide impacts per chemical) than to AQD’s IRSL (10-6 risk target for a proposed project, not facility-wide emissions). Staff then presented a handout, “MDEQ-AQD Air Toxics Workgroup (ATW) “Other Issues” September 19, 2013”. The proposed rule changes would update the risk assessment procedures in the air toxics rules to be consistent with the current EPA guidance for cancer and noncancer risk assessment, including age-dependent adjustment factors (ADAFs) for carcinogens with a mutagenic mode of action, and benchmark dose modeling. There were no concerns expressed; the handout will be added to the final report as Appendix Q. Action Items to be Completed Prior to the Next ATW Meeting: Bob will visit MMA and/or the Chamber to present the other air toxic rule revisions discussed today. AQD staff will investigate the possibility of revising the Rule 901 Policy and Procedure or look into Rule 228(2) language that can be more agreeable to all of the Members. AQD staff will continue consider the possibility of adding a food related exemption to the TAC definition. AQD staff to discuss with the AQD Field Supervisor the idea of notification under meaningful change. AQD staff will post meeting numbers 7 and 8 to the web site and will send out a summary of meeting number 9 to review. AQD staff will revise the draft final report according to recommended changes made today and will get a revised draft to Members by 10/1/13. Members will send back their recommended changes (in track changes) to the Facilitator by 10/16/13. Meeting Summary prepared by: Joy Taylor Morgan, Facilitator, and Bob Sills; October 1, 2013. 5