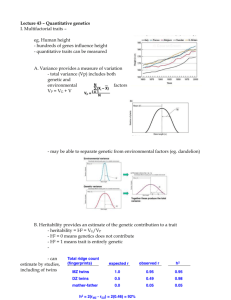
An Introduction to Genetic Analysis Chapter 25 Quantitative
advertisement

An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics Chapter 25 Quantitative Genetics Key Concepts In natural populations, variation in most characters takes the form of a continuous phenotypic range rather than discrete phenotypic classes. In other words, the variation is quantitative, not qualitative. Mendelian genetic analysis is extremely difficult to apply to such continuous phenotypic distributions, so statistical techniques are employed instead. A major task of quantitative genetics is to determine the ways in which genes interact with the environment to contribute to the formation of a given quantitative trait distribution. The genetic variation underlying a continuous character distribution can be the result of segregation at a single genetic locus or at numerous interacting loci that produce cumulative effects on the phenotype. The estimated ratio of genetic to environmental variation is not a measure of the relative contribution of genes and environment to phenotype. Estimates of genetic and environmental variance are specific to the single population and the particular set of environments in which the estimates are made. Introduction Ultimately, the goal of genetics is the analysis of the genotype of organisms. But the genotype can be identified — and therefore studied—only through its phenotypic effect. We recognize two genotypes as different from each other because the phenotypes of their carriers are different. Basic genetic experiments, then, depend on the existence of a simple relation between genotype and phenotype. That is why studies of DNA sequences are so important, because we can read off the genotype directly from this most basic of all phenotypes. In general, we hope to find a uniquely distinguishable phenotype for each genotype and only a simple genotype for each phenotype. At worst, when one allele is completely dominant, it may be necessary to perform a simple genetic cross to distinguish the heterozygote from the homozygote. Where possible, geneticists avoid studying genes that have only partial penetrance and incomplete expressivity (see Chapter 4) because of the difficulty of making genetic inferences from such traits. Imagine how difficult (if not impossible) it would have been for Benzer to study the fine structure of the gene in phage, if the only effect of the rII mutants was a 5 percent reduction from wild type in their ability to grow on E. coli K. For the most part, then, the study of genetics presented in the preceding chapters has been the study of allelic substitutions that cause qualitative differences in phenotype. However, the actual variation between organisms is usually quantitative, not qualitative. Wheat plants in a cultivated field or wild asters at the side of the road are not neatly sorted into categories of “tall” and “short,” any more than humans are neatly sorted into categories 1 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics of “black” and “white.” Height, weight, shape, color, metabolic activity, reproductive rate, and behavior are characteristics that vary more or less continuously over a range (Figure 25-1). Even when the character is intrinsically countable (such as eye facet or bristle number in Drosophila), the number of distinguishable classes may be so large that the variation is nearly continuous. If we consider extreme individuals—say, a corn plant 8 feet tall and another one 3 feet tall—a cross between them will not produce a Mendelian result. Such a corn cross will produce plants about 6 feet tall, with some clear variation among siblings. The F2 from selfing the F1 will not fall into two or three discrete height classes in ratios of 3:1 or 1:2:1. Instead, the F2 will be continuously distributed in height from one parental extreme to the other. This behavior of crosses is not an exception; it is the rule for most characters in most species. Mendel obtained his simple results because he worked with horticultural varieties of the garden pea that differed from one another by single allelic differences that had drastic phenotypic effects. Had Mendel conducted his experiments on the natural variation of the weeds in his garden, instead of abnormal pea varieties, he would never have discovered Mendel's laws. In general, size, shape, color, physiological activity, and behavior do not assort in a simple way in crosses. The fact that most phenotypic characters vary continuously does not mean that their variation is the result of some genetic mechanisms different from the Mendelian genes with which we have been dealing. The continuity of phenotype is a result of two phenomena. First, each genotype does not have a single phenotypic expression but a norm of reaction (see Chapter 1) that covers a wide phenotypic range. As a result, the phenotypic differences between genotypic classes become blurred, and we are not able to assign a particular phenotype unambiguously to a particular genotype. Second, many segregating loci may have alleles that make a difference in the phenotype under observation. Suppose, for example, that five equally important loci affect the number of flowers that will develop in an annual plant and that each locus has two alleles (call them + and −). For simplicity, also suppose that there is no dominance and that a + allele adds one flower, whereas a − allele adds nothing. Thus, there are 35 = 243 different possible genotypes [three possible genotypes (+/+, +/−, and −/−) at each of five loci], ranging from but there are only 11 phenotypic classes (10, 9, 8, . . . , 0) because many of the genotypes will have the same numbers of + and − alleles. For example, although there is only one genotype with 10 + alleles and therefore an average phenotypic value of 10, there are 51 different genotypes with 5 + alleles and 5 − alleles; for example, Thus, many different genotypes may have the same average phenotype. At the same time, because of environmental variation, two individuals of the same genotype may not have the 2 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics same phenotype. This lack of a one-to-one correspondence between genotype and phenotype obscures the underlying Mendelian mechanism. If we cannot study the behavior of the Mendelian factors controlling such traits directly, then what can we learn about their genetics? Using current experimental techniques, geneticists can answer the following questions about the genetics of a continuously varying character in a population (say, height in a human population). These questions constitute the study of quantitative genetics—the study of the genetics of continuously varying characters: 1. Is the observed variation in the character influenced at all by genetic variation? Are there alleles segregating in the population that produce some differential effect on the character or is all the variation simply the result of environmental variation and developmental noise (see Chapter 1)? 2. If there is genetic variation, what are the norms of reaction of the various genotypes? 3. How important is genetic variation as a source of total phenotypic variation? Are the norms of reaction and the environments such that nearly all the variation is a consequence of environmental difference and developmental instabilities or does genetic variation predominate? 4. Do many loci (or only a few) vary with respect to the character? How are they distributed over the genome? 5. How do the different loci interact with one another to influence the character? Is there dominance, and is there any epistasis (interaction between genes at different loci)? 6. Is there any nonnuclear inheritance (for example, any maternal effect)? In the end, the purpose of answering these questions is to be able to predict what kinds of offspring will be produced by crosses of different phenotypes. The precision with which these questions can be framed and answered varies greatly. In experimental organisms, on the one hand, it is relatively simple to determine whether there is any genetic influence at all, but extremely laborious experiments are required to localize the genes (even approximately). In humans, on the other hand, it is extremely difficult to answer even the question of the presence of genetic influence for most traits, because it is almost impossible to separate environmental from genetic effects in an organism that cannot be manipulated experimentally. As a consequence, we know a relatively large amount about the genetics of bristle number in Drosophila but virtually nothing about the genetics of complex human traits; a few (such as skin color) clearly are influenced by genes, whereas others (such as the specific language spoken) clearly are not. The purpose of this chapter is to develop the basic statistical and genetic concepts needed to answer these questions and to provide some examples of the applications of these concepts to particular characters in particular species. Some basic statistical notions 3 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics To consider the answers to these questions about the most common kinds of genetic variation, we must first examine a number of statistical tools that are essential in the study of quantitative genetics. Distributions The outcome of a cross for a Mendelian character can be described in terms of the proportions of the offspring that fall into several distinct phenotypic classes or often simply in terms of the presence or absence of a class. For example, a cross between a red-flowered plant and a white-flowered plant might be expected to yield all red-flowered plants or, if it were a backcross, 1/2 red-flowered plants and 1/2 white-flowered plants. However, we require a different mode of description for quantitative characters. The basic concept is that of the statistical distribution. If the heights of a large number of male undergraduates are measured to the nearest 5 centimeters (cm), they will vary (say, between 145 and 195 cm), but many more male undergraduates will be in the middle categories (say, between 170 and 180 cm) than at the extremes. Representing each measurement class as a bar, with its height proportional to the number of individuals in each class, we can graph the result as shown in Figure 25-2a. Such a graph of numbers of individuals observed against measurement class is a frequency histogram. Now suppose that five times as many individuals are measured, each to the nearest centimeter. The classes in Figure 25-2a are now subdivided to produce a histogram like the one shown in Figure 25-2b. If we continue this process, refining the measurement but proportionately increasing the number of individuals measured, then the histogram eventually takes on the continuous appearance of Figure 25-2c, which is the distribution function of heights in the population. This continuous curve is an idealization, because no measurement can be taken with infinite accuracy or on an unlimited number of individuals. Moreover, the measured variate itself may be intrinsically discontinuous because it is the count of some number of discrete objects such as eye facets or bristles. It is sometimes convenient, however, to develop concepts by using this slightly idealized picture as a shorthand for the more cumbersome observed frequency histogram (Figure 25-2a). Statistical measures Although a distribution contains all of the information about a set of measurements, we would like to be able to distill this information into a few characteristic numbers that convey the necessary information about the distribution without giving it in detail. The characteristics of a distribution that we would like to specify are as follows: 1. Where is the distribution located along the range of possible values? Are the observed values near 100 or near 1000? Therefore we need a measure of central tendency. 2. How much variation is there among individual measurements? Are they all concentrated around the central measurement or do they vary widely across a large range? That is, we need 4 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics a measure of dispersion. 3. If we are considering more than one characteristic, how are the values of these different characteristics related? Is there any relation between body size and fertility? Is it positive or negative? Do larger parents have larger offspring? If so, we would regard this as evidence that genes influence body size. Thus, we need measures of relation between measurements. Among the most commonly used measures of central tendency are the most frequent observation, the mode, and the arithmetic average of the observations, the mean. The dispersion of a distribution is almost always measured by the variance, which is the average squared distance of the observations from their mean. The relation between different variables is measured by their correlation, which is the average product of the deviation of one variable from its own mean times the deviation of the other variable from its own mean. These most commonly used measures of central tendency, dispersion, and relation are considered in detail in the Statistical Appendix to this chapter. The detailed discussion is in a separate section in order not to interrupt the flow of logic of the consideration of quantitative genetics. It should not be assumed, however, that an understanding of those statistical concepts is somehow secondary. A proper understanding of quantitative genetics requires a grasp of that material. Genotypes and phenotypic distribution Using the concepts of distribution, mean, and variance, we can understand the difference between quantitative and Mendelian genetic traits. Suppose that a population of plants contains three genotypes, each of which has some differential effect on growth rate. Furthermore, assume that there is some environmental variation from plant to plant because of inhomogeneity in the soil in which the population is growing and that there is some developmental noise (see Chapter 1). For each genotype, there will be a separate distribution of phenotypes with a mean and a standard deviation that depend on the genotype and the set of environments. Suppose that these distributions look like the three height distributions in Figure 25-3a. Finally, assume that the population consists of a mixture of the three genotypes but in the unequal proportions 1:2:3 (a/a:A/a:A/A). Then the phenotypic distribution of individuals in the population as a whole will look like the black line in Figure 25-3b, which is the result of summing the three underlying separate genotypic distributions, weighted by their frequencies in the population. This weighting by frequency is indicated in Figure 25-3b by the different heights of the component distributions that add up to the total distribution. The mean of this total distribution is the average of the three genotypic means, again weighted by the frequencies of the genotypes in the population. The variance of the total distribution is produced partly by the environmental variation within each genotype and partly by the slightly different means of the three genotypes. Two features of the total distribution are noteworthy. First, there is only a single mode. Despite the existence of three separate genotypic distributions underlying it, the population distribution as a whole does not reveal the separate modes. Second, any individual whose 5 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics height lies between the two arrows could have come from any one of the three genotypes, because they overlap so much. The result is that we cannot carry out any simple Mendelian analysis to determine the genotype of an individual organism. For example, suppose that the three genotypes are the two homozygotes and the heterozygote for a pair of alleles at a locus. Let a/a be the short homozygote and A/A be the tall one, with the heterozygote being of intermediate height. Because the phenotypic distributions overlap so much, we cannot know to which genotype a given individual belongs. Conversely, if we cross a homozygote a/a and a heterozygote A/a, the offspring will not fall into two discrete classes in a 1:1 ratio but will cover almost the entire range of phenotypes smoothly. Thus, we cannot know that the cross is in fact a/a × A/a and not a/a × A/A or A/a × A/a. Suppose we grew the hypothetical plants in Figure 25-3 in an environment that exaggerated the differences between genotypes—for example, by doubling the growth rate of all genotypes. At the same time, we were very careful to provide all plants with exactly the same environment. Then, the phenotypic variance of each separate genotype would be reduced because all the plants were grown under identical conditions; at the same time, the differences between genotypes would be exaggerated by the more rapid growth. The result (Figure 25-4b) would be a separation of the population as a whole into three nonoverlapping phenotypic distributions, each characteristic of one genotype. We could now carry out a perfectly conventional Mendelian analysis of plant height. A “quantitative” character has been converted into a “qualitative” one. This conversion has been accomplished by finding a way to make the differences between the means of the genotypes large compared with the variation within genotypes. MESSAGE A quantitative character is one for which the average phenotypic differences between genotypes are small compared with the variation between individuals within genotypes. It is sometimes assumed that continuous variation in a character is necessarily caused by a large number of segregating genes, so continuous variation is taken as evidence for control of the character by many genes. But, as we have just shown, this is not necessarily true. If the difference between genotypic means is small compared with the environmental variance, then even a simple one-gene–two-allele case can result in continuous phenotypic variation. If the range of a character is limited and if many segregating loci influence it, then we expect the character to show continuous variation, because each allelic substitution must account for only a small difference in the trait. This multiple-factor hypothesis (that large numbers of genes, each with a small effect, are segregating to produce quantitative variation) has long been the basic model of quantitative genetics, although there is no convincing evidence that such groups of genes really exist. A special name, polygenes, has been coined for these hypothetical factors of small-but-equal effect, in contrast to the genes of simple Mendelian analysis. 6 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics It is important to remember, however, that the number of segregating loci that influence a trait is not what separates quantitative and qualitative characters. Even in the absence of large environmental variation, it takes only a few genetically varying loci to produce variation that is indistinguishable from the effect of many loci of small effect. As an example, we can consider one of the earliest experiments in quantitative genetics, that of Wilhelm Johannsen on pure lines. By inbreeding (mating close relatives), Johannsen produced 19 homozygous lines of bean plants from an originally genetically heterogeneous population. Each line had a characteristic average seed weight ranging from 0.64 g per seed for the heaviest line to 0.35 g per seed for the lightest line. It is by no means clear that all these lines were genetically different (for example, five of the lines had seed weights of 0.450, 0.453, 0.454, 0.454, and 0.455 g), but let's take the most extreme position—that the lines were all different. These observations would be incompatible with a simple one-locus–two-allele model of gene action. In that case, if the original population were segregating for the two alleles A and a, all inbred lines derived from that population would have to fall into one of two classes: A/A or a/a. If, in contrast, there were, say, 100 loci, each of small effect, segregating in the original population, then a vast number of different inbred lines could be produced, each with a different combination of homozygotes at different loci. However, we do not need such a large number of loci to obtain the result observed by Johannsen. If there were only five loci, each with three alleles, then 35 = 243 different kinds of homozygotes could be produced from the inbreeding process. If we make 19 inbred lines at random, there is a good chance (about 50 percent) that each of the 19 lines will belong to a different one of the 243 classes. So Johannsen's experimental results can be easily explained by a relatively small number of genes. Thus, there is no real dividing line between polygenic traits and other traits. It is safe to say that no phenotypic trait above the level of the amino acid sequence in a polypeptide is influenced by only one gene. Moreover, traits influenced by many genes are not equally influenced by all of them. Some genes will have major effects on a trait; others, minor effects. MESSAGE The critical difference between Mendelian and quantitative traits is not the number of segregating loci but the size of phenotypic differences between genotypes compared with the individual variation within genotypic classes. Norm of reaction and phenotypic distribution The phenotypic distribution of a trait, as we have seen, is a function of the average differences between genotypes and of the variation between genotypically identical individuals. But both are in turn functions of the sequence of environments in which the organisms develop and live. For a given genotype, each environment will result in a given phenotype (for the moment, ignoring developmental noise). Then a distribution of environments will be reflected biologically as a distribution of phenotypes. The way in which the environmental distribution is transformed into the phenotypic distribution is determined by the norm of reaction, as shown in Figure 25-5. The horizontal axis is environment (say, temperature) and the vertical 7 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics axis is phenotype (say, plant height). The norm of reaction curve for the genotype shows how each particular temperature results in a particular plant height. So, the dashed lines from the 18°C point on the temperature axis is reflected off the norm of reaction curve to a corresponding plant height on the vertical phenotype axis, and so forth for each temperature. If a large number of individuals develop at, say, 20°C, then a large number of individuals will have the phenotype that corresponds to 20°C, as shown by the dashed line; and, if only small numbers develop at 18°C, few plants will have the corresponding plant height. Then the frequency distribution of developmental environments will be reflected as a frequency distribution of phenotypes as determined by the shape of the norm of reaction curve. It is as if an observer, standing at the vertical phenotype axis, were seeing the environmental distribution, not directly, but reflected in the curved mirror of the norm of reaction. The shape of the curvature will determine how the environmental distribution is distorted on the phenotype axis. So, the norm of reaction in Figure 25-5 falls very rapidly at lower temperatures (the phenotype changes rapidly with small changes in temperature) but flattens out at higher temperatures, so the plant height is much less sensitive to temperature differences at the higher temperatures. The result is that the symmetric environmental distribution is converted into an asymmetric phenotype distribution with a long tail at the larger plant heights, corresponding to the lower temperatures. By means of the same analysis, Figure 25-6 shows how a population consisting of two genotypes with different norms of reaction has a phenotypic distribution that depends on the distribution of environments. If the environments are distributed as shown by the black distribution curve, then the resulting population of plants will have a unimodal distribution, because the difference between genotypes is very small in this range of environments compared with the sensitivity of the norms of reaction to small changes in temperature. If the distribution of environments is shifted to the right, however, as shown by the gray distribution curve, a bimodal distribution of phenotypes results, because the norms of reaction are nearly flat in this environmental range but very different from each other. MESSAGE A distribution of environments is reflected biologically as a distribution of phenotypes. The transformation of environmental distribution into phenotypic distribution is determined by the norm of reaction. Determining norms of reaction Remarkably little is known about the norms of reaction for any quantitative traits in any species—partly because it is difficult in most sexually reproducing species to replicate a genotype so that it can be tested in different environments. For this reason, for example, we do not have a norm of reaction for any genotype for any human quantitative trait. In domesticated plants and animals 8 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics A few norm of reaction studies have been carried out with plants that can be clonally propagated. The results of one of these experiments are presented in Chapter 1. It is possible to replicate genotypes in sexually reproducing organisms by the technique of mating close relatives, or inbreeding. By selfing (where possible) or by mating brother and sister repeatedly generation after generation, a segregating line (one that contains both homozygotes and heterozygotes at a locus) can be made homozygous. The purpose of creating homozygous lines is to produce groups of organisms within which all individuals are genetically identical. These genetically identical individuals can then be allowed to develop in different environments to produce a norm of reaction. Alternatively, two different homozygous lines can be crossed and the F1 offspring, all genetically identical with one another, can be characterized in different environments. Ideally for a norm of reaction study all the individuals should be absolutely identical genetically, but the process of inbreeding increases the homozygosity of the group slowly, generation after generation, depending on the closeness of the relatives that are mated. In corn, for example, a single individual is chosen and self-pollinated. Then in the next generation, a single one of its offspring is chosen and self-pollinated. In the third generation, a single one of its offspring is chosen and self-pollinated, and so forth. Suppose that the original individual in the first generation is already a homozygote at some locus. Then all of its offspring from self-pollination will also be homozygous and identical at the locus. Future generations of self-pollination will simply preserve the homozygosity. If, on the other hand, the original individual is a heterozygote, then the selfing A/a × A/a will produce 1/4 A/A homozygotes and 1/4 a/a homozygotes. If a single offspring is chosen in this subsequent generation to propagate the line, then there is a 50 percent chance that it is now a homozygote. If, by bad luck, the chosen individual should still be a heterozygote, there is another 50 percent chance that the selected individual in the third generation is homozygous, and so forth. Of the ensemble of all heterozygous loci, then, after one generation of selfing, only 1/2 will still be heterozygous; after two generations, 1/4; after three, 1/8. In the nth generation, where Hetn is the proportion of heterozygous loci in the nth generation and Het0 is the proportion in the 0 generation. When selfing is not possible, brother–sister mating will accomplish the same end, although more slowly. Table 25-1 is a comparison of the amount of heterozygosity left after n generations of selfing and brother–sister mating. In natural populations To carry out a norm of reaction study of a natural population, a large number of lines are sampled from the population and inbred for a sufficient number of generations to guarantee that each line is virtually homozygous at all its loci. Each line is then homozygous at each locus for a randomly selected allele present in the original population. The inbred lines 9 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics themselves cannot be used to characterize norms of reaction in the natural population, because such totally homozygous genotypes do not exist in the original population. Each inbred line can be crossed to every other inbred line to produce heterozygotes that reconstitute the original population, and an arbitrary number of individuals from each cross can be produced. If inbred line 1 has the genetic constitution A/A·B/B·c/c·d/d·E/E . . . and inbred line 2 is a/a·B/B·C/C·d/d·e/e . . . , then a cross between them will produce a large number of offspring, all of whom are identically A/a·B/B·C/c·d/d·E/e . . . and can be raised in different environments. Inbreeding by mating of close relatives for many generations results in total homozygosity for the entire genome. In species such as Drosophila in which the necessary dominant markers and crossover suppressors are available, it is possible to produce lines that are homozygous for only a single chromosome, rather than for the whole set, as shown for an autosome in Figure 25-7. A single male from the population to be sampled is crossed to a female carrying a chromosome with a crossover suppressor C (usually a complex inversion), a recessive lethal l, and a dominant visible marker M1 heterozygous with a second dominant visible M2. In the F1, a single male carrying the ClM1 chromosome is chosen. This male, which is also carrying a wild-type chromosome from the population, is again crossed to the marker stock. In the F2, all flies showing the M1 trait but not M2 are necessarily all heterozygotes for copies of the original wild-type chromosome because ClM1/ClM1 is lethal, and no crossovers have taken place. In the F3, all wild-type flies are identically homozygous for the wild-type chromosome and are now available to make a stock for norm of reaction studies for crosses. (See Chapter 15 for another use of this technique.) Results of norm of reaction studies Very few norm of reaction studies have been carried out for quantitative characters found in natural populations, but many more have been carried out for domesticated species such as corn, which can be self-pollinated, or strawberries, which can be clonally propagated. The outcomes of such studies resemble those given in Figure 25-8, which shows the norms of reaction for abdominal bristle number as a function of temperature for second chromosome homozygotes of D. pseudoobscura. No genotype is consistently above or below the others. Instead, there are small differences between genotypes, and the direction of these differences is not consistent over a wide range of environments. These factors have two important consequences. First, the selection of “superior” genotypes in domesticated animals and cultivated plants will result in very specifically adapted varieties that may not show their superior proper-ties in other environments. To some extent, this problem is overcome by deliberately testing genotypes in a range of environments (for example, over several years and in several locations). It would be even better, however, if plant breeders could test their selections in a variety of controlled environments in which different environmental factors could be separately manipulated. The consequences of actual plant-breeding practices can be seen in Figure 25-9, in which the yields of two varieties of corn are shown as a function of different farm environments. Variety 1 is an older variety of 10 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics hybrid corn; variety 2 is a later “improved” hybrid. These performances are compared at a low planting density, which prevailed when variety 1 was developed, and at a high planting density characteristic of farming practice when hybrid 2 was selected. At the high density, the new variety is clearly superior to the old variety in all environments (Figure 25-9a). At the low density, however, the situation is quite different. First, note that the new variety is less sensitive to environment than is the older hybrid, as evidenced by its flatter norm of reaction. Second, the new “improved” variety is actually poorer under the best farm conditions. Third, the yield improvement of the new variety is not apparent under the low densities characteristic of earlier agricultural practice. The second consequence of the nature of reaction norms is that, even if it should turn out that there is genetic variation for various mental and emotional traits in the human species, which is by no means clear, this variation is unlikely to favor one genotype over another across a range of environments. We must beware of hypothetical norms of reaction for human cognitive traits that show one genotype unconditionally superior to another. Even putting aside all questions of moral and political judgment, there is simply no basis for describing different human genotypes as “better” or “worse” on any scale, unless the investigator is able to make a very exact specification of environment. MESSAGE Norm of reaction studies show only small differences between natural genotypes, and these differences are not consistent over a wide range of environments. Thus, “superior” genotypes in domesticated animals and cultivated plants may be superior only in certain environments. If it should turn out that humans exhibit genetic variation for various mental and emotional traits, this variation is unlikely to favor one genotype over another across a range of environments. The most basic question to be asked about a quantitative trait is whether the observed variation in the character is influenced by genes at all. It is important to note that this is not the same as asking whether genes play any role in the character's development. Gene-mediated developmental processes lie at the base of every character, but variation from individual to individual is not necessarily the result of genetic variation. Thus, the possibility of speaking any language at all depends critically on the structures of the central nervous system as well as of the vocal cords, tongue, mouth, and ears, which depend in turn on the nature of the human genome. There is no environment in which cows will speak. But, although the particular language that is spoken by humans varies from nation to nation, that variation is totally nongenetic. MESSAGE The question of whether a trait is heritable is a question about the role that differences in genes play in the phenotypic differences between individuals or groups. Familiality and heritability 11 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics In principle, it is easy to determine whether any genetic variation influences the phenotypic variation among organisms for a particular trait. If genes are involved, then (on average) biological relatives should resemble one another more than unrelated individuals do. This resemblance would be seen as a positive correlation between parents and offspring or between siblings (offspring of the same parents). Parents who are larger than the average would have offspring who are larger than the average; the more seeds that a plant produces, the more seeds that its siblings would produce. Such correlations between relatives, however, are evidence for genetic variation only if the relatives do not share common environments more than nonrelatives do. It is absolutely fundamental to distinguish familiality from heritability. Traits are familial if members of the same family share them, for whatever reason. Traits are heritable only if the similarity arises from shared genotypes. There are two general methods for establishing the heritability of a trait as distinct from its familial occurrence. The first depends on phenotypic similarity between relatives. For most of the history of genetics, this method has been the only one available; so nearly all the evidence about heritability for most traits in experimental organisms and in humans has been established by using this approach. The second method, using marker-gene segregation, depends on showing that genotypes carrying different alleles of marker genes also differ in their average phenotype for the quantitative character. If the marker genes (which have nothing to do with the character under study) are seen to vary in relation to the character, presumably they are linked to genes that do influence the character and its variation. Thus, heritability is demonstrated even if the actual genes causing the variation are not known. This method requires that the genome of the organism being studied have large numbers of detectable genetically variable marker loci spread throughout the genome. Such marker loci can be observed from electrophoretic studies of protein variation or, in vertebrates, from immunological studies of blood group genes. For example, within flocks, chickens of different blood groups show some difference in egg weight. Since the introduction of molecular methods for the study of DNA sequence variation, very large numbers of variable nucleotide positions have been discovered in a great variety of organisms. This molecular variation includes both single nucleotide replacements and insertions and deletions of longer nucleotide sequences. These variations are usually detected by the gain or loss of sites of cleavage of restriction enzymes or by length variation of DNA sequences between two fixed restriction sites, both of which are a form of restriction fragment length polymorphisms (RFLPs). In tomatoes, for example, strains carrying different RFLP variants differ in fruit characteristics. However, because so much of what is known or claimed about heritability still depends on phenotypic similarity between relatives, especially in human genetics, we will begin the examination of the problem of heritability by analyzing phenotypic similarity. Phenotypic similarity between relatives In experimental organisms, there is no problem in separating environmental from genetic similarities. The offspring of a cow producing milk at a high rate and the offspring of a cow 12 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics producing milk at a low rate can be raised together in the same environment to see whether, despite the environmental similarity, each resembles its own parent. In natural populations, and especially in humans, this is difficult to do. Because of the nature of human societies, members of the same family not only share genes, but also have similar environments. Thus, the observation of simple familiality of a trait is genetically uninterpretable. In general, people who speak Hungarian have Hungarian-speaking parents and people who speak Japanese have Japanese-speaking parents. Yet the massive experience of immigration to North America has demonstrated that these linguistic differences, although familial, are nongenetic. The highest correlations between parents and offspring for any social traits in the United States are those for political party and religious sect, but they are not heritable. The distinction between familiality and heredity is not always so obvious. The Public Health Commission, which originally studied the vitamindeficiency disease pellegra in the southern United States in 1910, came to the conclusion that it was genetic because it ran in families. To determine whether a trait is heritable in human populations, we must use adoption studies to avoid the usual environmental similarity between biological relatives. The ideal experimental subjects are identical twins reared apart, because they are genetically identical but environmentally different. Such adoption studies must be so contrived that there is no correlation between the social environment of the adopting family and that of the biological family. These requirements are exceedingly difficult to meet; so, in practice, we know very little about whether human quantitative traits that are familial are also heritable. Skin color is clearly heritable, as is adult height—but even for these traits we must be very careful. We know that skin color is affected by genes from studies of cross-racial adoptions and observations that the offspring of black African slaves were black even when they were born and reared in Canada. But are the differences in height between Japanese and Europeans affected by genes? The children of Japanese immigrants who are born and reared in North America are taller than their parents but shorter than the North American average, so we might conclude that there is some influence of genetic difference. However, second-generation Japanese Americans are even taller than their American-born parents. It appears that some environmental–cultural influence or perhaps a maternal effect is still felt in the first generation of births in North America. We cannot yet say whether genetic differences in height distinguish North Americans of, say, Japanese and Swedish ancestry. Personality traits, temperament, and cognitive performance (including IQ scores), as well as a whole variety of behaviors such as alcoholism and of mental disorders such as schizophrenia, have been the subject of heritability studies in human populations. Many show familiality. There is indeed a positive correlation between the IQ scores of parents and the scores of their children (the correlation is about 0.5 in white American families), but the correlation does not distinguish familiality from heritability. To make that distinction requires that the environmental correlation between parents and children be broken, so adoption studies are common. Because it is difficult to randomize the environments, even in cases of adoption, evidence of heritability for human personality and behavior traits remains equivocal despite the very large number of studies that exist. Prejudices about the causes of human differences are widespread and deep, and, as a result, the canons of evidence adhered to in studies of the 13 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics heritability of IQ, for example, have been much more lax than in studies of milk yield in cows. Figure 25-10 summarizes the usual method for testing heritability in experimental organisms. Individuals from both extremes of the distribution are mated with their own kind, and the offspring are raised in a common controlled environment. If there is an average difference between the two offspring groups, the trait is heritable. Most morphological traits in Drosophila, for example, turn out to be heritable—but not all of them. If flies with right wings that are slightly longer than their left wings are mated together, their offspring have no greater tendency to be “right winged” than do the offspring of “left winged” flies. As we shall see later, this method can also be used to obtain quantitative information about heritability. MESSAGE In experimental organisms, environmental similarity can often be readily distinguished from genetic similarity (heritability). In humans, however, it is very difficult to determine whether a particular trait is heritable. Heritability of a trait The most basic question to be asked about a quantitative trait is whether the observed variation in the character is influenced by genes at all. It is important to note that this is not the same as asking whether genes play any role in the character's development. Gene-mediated developmental processes lie at the base of every character, but variation from individual to individual is not necessarily the result of genetic variation. Thus, the possibility of speaking any language at all depends critically on the structures of the central nervous system as well as of the vocal cords, tongue, mouth, and ears, which depend in turn on the nature of the human genome. There is no environment in which cows will speak. But, although the particular language that is spoken by humans varies from nation to nation, that variation is totally nongenetic. MESSAGE The question of whether a trait is heritable is a question about the role that differences in genes play in the phenotypic differences between individuals or groups. Familiality and heritability In principle, it is easy to determine whether any genetic variation influences the phenotypic variation among organisms for a particular trait. If genes are involved, then (on average) biological relatives should resemble one another more than unrelated individuals do. This resemblance would be seen as a positive correlation between parents and offspring or between siblings (offspring of the same parents). Parents who are larger than the average would have offspring who are larger than the average; the more seeds that a plant produces, the more seeds that its siblings would produce. Such correlations between relatives, however, are evidence for genetic variation only if the relatives do not share common environments more 14 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics than nonrelatives do. It is absolutely fundamental to distinguish familiality from heritability. Traits are familial if members of the same family share them, for whatever reason. Traits are heritable only if the similarity arises from shared genotypes. There are two general methods for establishing the heritability of a trait as distinct from its familial occurrence. The first depends on phenotypic similarity between relatives. For most of the history of genetics, this method has been the only one available; so nearly all the evidence about heritability for most traits in experimental organisms and in humans has been established by using this approach. The second method, using marker-gene segregation, depends on showing that genotypes carrying different alleles of marker genes also differ in their average phenotype for the quantitative character. If the marker genes (which have nothing to do with the character under study) are seen to vary in relation to the character, presumably they are linked to genes that do influence the character and its variation. Thus, heritability is demonstrated even if the actual genes causing the variation are not known. This method requires that the genome of the organism being studied have large numbers of detectable genetically variable marker loci spread throughout the genome. Such marker loci can be observed from electrophoretic studies of protein variation or, in vertebrates, from immunological studies of blood group genes. For example, within flocks, chickens of different blood groups show some difference in egg weight. Since the introduction of molecular methods for the study of DNA sequence variation, very large numbers of variable nucleotide positions have been discovered in a great variety of organisms. This molecular variation includes both single nucleotide replacements and insertions and deletions of longer nucleotide sequences. These variations are usually detected by the gain or loss of sites of cleavage of restriction enzymes or by length variation of DNA sequences between two fixed restriction sites, both of which are a form of restriction fragment length polymorphisms (RFLPs). In tomatoes, for example, strains carrying different RFLP variants differ in fruit characteristics. However, because so much of what is known or claimed about heritability still depends on phenotypic similarity between relatives, especially in human genetics, we will begin the examination of the problem of heritability by analyzing phenotypic similarity. Phenotypic similarity between relatives In experimental organisms, there is no problem in separating environmental from genetic similarities. The offspring of a cow producing milk at a high rate and the offspring of a cow producing milk at a low rate can be raised together in the same environment to see whether, despite the environmental similarity, each resembles its own parent. In natural populations, and especially in humans, this is difficult to do. Because of the nature of human societies, members of the same family not only share genes, but also have similar environments. Thus, the observation of simple familiality of a trait is genetically uninterpretable. In general, people who speak Hungarian have Hungarian-speaking parents and people who speak Japanese have Japanese-speaking parents. Yet the massive experience of immigration to North America has demonstrated that these linguistic differences, although familial, are nongenetic. The highest 15 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics correlations between parents and offspring for any social traits in the United States are those for political party and religious sect, but they are not heritable. The distinction between familiality and heredity is not always so obvious. The Public Health Commission, which originally studied the vitamindeficiency disease pellegra in the southern United States in 1910, came to the conclusion that it was genetic because it ran in families. To determine whether a trait is heritable in human populations, we must use adoption studies to avoid the usual environmental similarity between biological relatives. The ideal experimental subjects are identical twins reared apart, because they are genetically identical but environmentally different. Such adoption studies must be so contrived that there is no correlation between the social environment of the adopting family and that of the biological family. These requirements are exceedingly difficult to meet; so, in practice, we know very little about whether human quantitative traits that are familial are also heritable. Skin color is clearly heritable, as is adult height—but even for these traits we must be very careful. We know that skin color is affected by genes from studies of cross-racial adoptions and observations that the offspring of black African slaves were black even when they were born and reared in Canada. But are the differences in height between Japanese and Europeans affected by genes? The children of Japanese immigrants who are born and reared in North America are taller than their parents but shorter than the North American average, so we might conclude that there is some influence of genetic difference. However, second-generation Japanese Americans are even taller than their American-born parents. It appears that some environmental–cultural influence or perhaps a maternal effect is still felt in the first generation of births in North America. We cannot yet say whether genetic differences in height distinguish North Americans of, say, Japanese and Swedish ancestry. Personality traits, temperament, and cognitive performance (including IQ scores), as well as a whole variety of behaviors such as alcoholism and of mental disorders such as schizophrenia, have been the subject of heritability studies in human populations. Many show familiality. There is indeed a positive correlation between the IQ scores of parents and the scores of their children (the correlation is about 0.5 in white American families), but the correlation does not distinguish familiality from heritability. To make that distinction requires that the environmental correlation between parents and children be broken, so adoption studies are common. Because it is difficult to randomize the environments, even in cases of adoption, evidence of heritability for human personality and behavior traits remains equivocal despite the very large number of studies that exist. Prejudices about the causes of human differences are widespread and deep, and, as a result, the canons of evidence adhered to in studies of the heritability of IQ, for example, have been much more lax than in studies of milk yield in cows. Figure 25-10 summarizes the usual method for testing heritability in experimental organisms. Individuals from both extremes of the distribution are mated with their own kind, and the offspring are raised in a common controlled environment. If there is an average difference between the two offspring groups, the trait is heritable. Most morphological traits in Drosophila, for example, turn out to be heritable—but not all of them. If flies with right 16 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics wings that are slightly longer than their left wings are mated together, their offspring have no greater tendency to be “right winged” than do the offspring of “left winged” flies. As we shall see later, this method can also be used to obtain quantitative information about heritability. MESSAGE In experimental organisms, environmental similarity can often be readily distinguished from genetic similarity (heritability). In humans, however, it is very difficult to determine whether a particular trait is heritable. Quantifying heritability If a trait is shown to have some heritability in a population, then it is possible to quantify the degree of heritability. In Figure 25-3, we saw that the variation between phenotypes in a population arises from two sources. First, there are average differences between the genotypes; second, each genotype exhibits phenotypic variance because of environmental variation. The total phenotypic variance of the population (S2p) can then be broken into two parts: the variance between genotypic means (S2g) and the remaining variance (S2e) The former is called the genetic variance, and the latter is called the environmental variance; however, as we shall see, these names are quite misleading. Moreover, the breakdown of the phenotypic variance into the sum of environmental and genetic variance leaves out the possibility of some covariance between genotype and environment. For example, suppose it were true (we do not know) that there are genes that influence musical ability. Parents with such genes might themselves be musicians, who would create a more musical environment for their children, who would then have both the genes and the environment promoting musical performance. The result would be an increase in the phenotypic variances of musical ability and an erroneous estimate of genetic and environmental variances. If the phenotype is the sum of a genetic and an environmental effect, P = G + E, then, as explained on page 768 of the Statistical Appendix, the variance of the phenotype is the sum of the genetic variance, the environmental variance, and twice the covariance between the genotypic and environmental effects. If genotypes are not distributed randomly across environments, there will be some covariance between genotype and environmental values, and the covariance will be hidden in the genetic and environmental variances. The degree of heritability can be defined as the part of the total variance that is due to genetic variance: 17 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics H2, so defined, is called the broad heritability of the character. It must be stressed that this measure of “genetic influence” tells us what part of the population's variation in phenotype can be assigned to variation in genotype. It does not tell us what parts of an individual's phenotype can be ascribed to its heredity and to its environment. This latter distinction is not a reasonable one. An individual's phenotype is a consequence of the interaction between its genes and its sequence of environments. It clearly would be silly to say that you owe 60 inches of your height to genes and 10 inches to environment. All measures of the “importance” of genes are framed in terms of the proportion of variance ascribable to their variation. This approach is a special application of the more general technique of the analysis of variance for apportioning relative weight to contributing causes. The method was, in fact, invented originally to deal with experiments in which different environmental and genetic factors were influencing the growth of plants. (For a sophisticated but accessible treatment of the analysis of variance written for biologists, see R. Sokal and J. Rohlf, Biometry, 3d ed. W. H. Freeman and Company, 1995.) Methods of estimating H2 Genetic variance and heritability can be estimated in several ways. Most directly, we can obtain an estimate of (S2e) by making a number of homozygous lines from the population, crossing them in pairs to reconstitute individual heterozygotes, and measuring the phenotypic variance within each heterozygous genotype. Because there is no genetic variance within a genotypic class, these variances will (when averaged) provide an estimate of (S2e). This value can then be subtracted from the value of (S2p) in the original population to give (S2g). With the use of this method, any covariance between genotype and environment in the original population will be hidden in the estimate of genetic variance and will inflate it. Other estimates of genetic variance can be obtained by considering the genetic similarities between relatives. Using simple Mendelian principles, we can see that half the genes of full siblings will (on average) be identical. For identification purposes, we can label the alleles at a locus carried by the parents differently, so that they are, say, A1/A2 and A3/A4. Now the older sibling has a probability of 1/2 of getting A1 from its father, as does the younger sibling, so the two siblings have a chance of 1/2 × 1/2 = 1/4 of both carrying A1. On the other hand, they might both have received an A2 from their father; so, again, they have a probability of 1/4 of carrying a gene in common that they inherited from their father. Thus, the chance is 1/4 + 1/4 = 1/2 that both siblings will carry an A1 or that both siblings will carry an A2. The other half of the time, one sibling will inherit an A1 and the other will inherit an A2. So, as far as paternally inherited genes are concerned, full siblings have a 50 percent chance of carrying the same allele. But the same reasoning applies to their maternally inherited gene. Averaging over their paternally and maternally inherited genes, half the genes of full siblings are identical between them. Their genetic correlation, which is equal to the chance that they carry the same allele, is 1/2. If we apply this reasoning to half-siblings, say, with a common father but with different mothers, we get a different result. Again, the two siblings have a 50 percent chance of 18 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics inheriting an identical gene from their father, but this time they have no way of inheriting the same gene from their mothers because they have two different mothers. Averaging the maternally inherited and paternally inherited genes thus gives a probability of (1/2 + 0)/2 = 1/4 that these half-siblings will carry the same gene. We might be tempted to use the theoretical correlation between, say, siblings to estimate H2. If the observed phenotypic correlation were, for example, 0.4 and we expect on purely genetic grounds a correlation of .05, then an estimate of heritability would be 0.4/0.5 = 0.8. But such an estimate fails to take into account the fact that siblings may also be environmentally correlated. Unless we are careful to raise the siblings in independent environments, the estimate of H2 would be too large and could even exceed 1 if the observed phenotypic correlation were greater than 0.5. To get around this problem, we use the differences between phenotypic correlations of different relatives. For example, the difference in genetic correlation between full and half-siblings is 1/2 − 1/4 = 1/4. Let's contrast this with their phenotypic correlations. If the environmental similarity is the same for half- and full siblings—a very important condition for estimating heritability—then environmental similarities will cancel out if we take the difference in correlation between the two kinds of siblings. This difference in phenotypic correlation will then be proportional to how much of the variance is genetic. Thus: but so an estimate of H2 is: where the correlation here is the phenotypic correlation. We can use similar arguments about genetic similarities between parents and offspring and between twins to obtain two other estimates of H2: and 19 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics These formulas are derived from considering the genetic similarities between relatives. They are only approximate and depend on assumptions about the ways in which genes act. The first two formulas, for example, assume that genes at different loci add together in their effect on the character. The last formula also assumes that the alleles at each locus show no dominance (see the discussion of components of variance on pages 760–762). All these estimates, as well as others based on correlations between relatives, depend critically on the assumption that environmental correlations between individuals are the same for all degrees of relationship. If closer relatives have more similar environments, as they do in humans, the estimates of heritability are biased. It is reasonable to assume that most environmental correlations between relatives are positive, in which case the heritabilities would be overestimated. Negative environmental correlations also can exist. For example, if the members of a litter must compete for food that is in short supply, there could be negative correlations in growth rates among siblings. The difference in correlation between monozygotic and dizygotic twins is commonly used in human genetics to estimate H2 for cognitive or personality traits. Here the problem of degree of environmental similarity is very severe. Identical (monozygotic) twins are generally treated more similarly to each other than are fraternal (dizygotic) twins. People often give their identical twins names that are similar, dress them alike, treat them identically, and, in general, accent their similarities. As a result, heritability is overestimated. Meaning of H2 Attention to the problems of estimating broad heritability distracts from the deeper questions about the meaning of the ratio when it can be estimated. Despite its widespread use as a measure of how “important” genes are in influencing a trait, H2 actually has a special and limited meaning. There are two conclusions that can be drawn from a properly designed heritability study. First, if there is a nonzero heritability, then, in the population measured and in the environments in which the organisms have developed, genetic differences have influenced the variation between individuals, so genetic differences do matter to the trait. This finding is not trivial and is a first step in a more detailed investigation of the role of genes. It is important to notice that the reverse is not true. Finding no heritability for the trait is not a demonstration that genes are irrelevant; rather, it demonstrates that, in the particular population studied, there is no genetic variation at the relevant loci or that the environments in which the population developed were such that different genotypes had the same phenotype. In other populations or other environments, the trait might be heritable. 20 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics MESSAGE In general, the heritability of a trait is different in each population and in each set of environnents; it cannot be extrapolated from one population and set of environments to another. Moreover, we must distinguish between genes being relevant to a trait and genetic differences being relevant to differences in the trait. The experiment of immigration to North America has proved that the ability to pronounce the sounds of North American English, rather than French, Swedish, or Russian, is not a consequence of genetic differences between our immigrant ancestors. But, without the appropriate genes, we could not speak any language at all. Second, the value of the H2 provides a limited prediction of the effect of environmental modification under particular circumstances. If all the relevant environmental variation is eliminated and the new constant environment is the same as the mean environment in the original population, then H2 estimates how much phenotypic variation will still be present. So, if the heritability of performance on an IQ test were, say, 0.4, then, if all children had the same developmental and social environment as the “average child,” about 60 percent of the variation in IQ test performance would disappear and 40 percent would remain. The requirement that the new constant environment be at the mean of the old environmental distribution is absolutely essential to this prediction. If the environment is shifted toward one end or the other of the environmental distribution or a new environment is introduced, nothing at all can be predicted. In the example of IQ performance, the heritability gives us no information at all about how variable performance would be if children's developmental and social environments were generally enriched. To understand why this is so, we must return to the concept of the norm of reaction. The separation of variance into genetic and environmental components S2g and S2e does not really separate the genetic and environmental causes of variation. Consider Figure 25-9b. When the environment is poor (50), corn variety 2 is much higher yielding than variety 1, so a population made up of a mixture of the two varieties would have a lot of genetic variance for yield. But, in an environment scoring 80, there is no difference in yield between genotypes 1 and 2, so a mixed population would have no genetic variance at all for yield in that environment. Thus, genetic variance has been changed by changing the environment. On the other hand, variety 2 is less sensitive to environment than variety 1, as shown by the slopes of the lines. So a population made up mostly of genotype 2 would have a lower environmental variance than one made up mostly of genotype 1. So, environmental variance in the population is changed by changing the proportion of genotypes. MESSAGE Because genotype and environment interact to produce phenotype, no partition of variation can actually separate causes of variation. 21 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics As a consequence of the argument just given, knowledge of the heritability of a trait does not permit us to predict how the distribution of that trait will change if either genotypic frequencies or environmental factors change markedly. MESSAGE A high heritability does not mean that a trait is unaffected by its environment. All that high heritability means is that, for the particular population developing in the particular distribution of environments in which the heritability was measured, average differences between genotypes are large compared with environmental variation within genotypes. If the environment is changed, there may be large differences in phenotype. Perhaps the most well known example of the erroneous use of heritability arguments to make claims about the changeability of a trait is the case of human IQ performance and social success. In 1969, an educational psychologist, A. R. Jensen, published a long paper in the Harvard Educational Review, asking the question (in its title) “How much can we boost IQ and scholastic achievement?” Jensen's conclusion was “not much.” As an explanation and evidence of this unchangeability, he offered a claim of high heritability for IQ performance. A great deal of criticism has been made of the evidence offered by Jensen for the high heritability of IQ scores. But, irrespective of the correct value of H2 for IQ performance, the real error of Jensen's argument lies in his equation of high heritability with unchangeability. In fact, the heritability of IQ is irrelevant to the question raised in the title of his article. To see why this is so, let us consider the results of adoption studies in which children are separated from their biological parents in infancy and reared by adoptive parents. Although results may vary quantitatively from study to study, there are three characteristics in common. First, adopting parents generally have higher IQ scores than those of the biological parents. Second, the adopted children have higher IQ scores than those of their biological parents. Third, the adopted children show a higher correlation of IQ scores with their biological parents than with their adoptive families. The following table is a hypothetical data set that shows all these characteristics, in idealized form, to illustrate the concepts. The scores given for parents are meant to be the average of mother and father. First, we can see that the children have a high correlation with their biological parents but a low correlation with their adoptive parents. In fact, in our hypothetical example, the 22 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics correlation of children with biological parents is r = 1.00, but, with adoptive parents, it is r = 0. (The correlation between two sets of numbers does not mean that the two sets are identical but that, for each unit increase in one set, there is a constant proportion increase in the other set. See page 768 of the Statistical Appendix at the end of this chapter.) This perfect correlation with biological parents and zero correlation with adoptive parents means that H2 = 1, given the arguments developed on page 755. All the variation in IQ score between the children is explained by the variation between the biological parents. Second, however, we notice that each of the IQ scores of the children is 20 points higher than the IQ scores of their respective biological parents and that the mean IQ of the children is equal to the mean IQ of the adoptive parents. Thus, adoption has raised the average IQ of the children 20 points higher than the average IQ of their biological parents; so, as a group, the children resemble their adoptive parents. So we have perfect heritability, yet high environmental plasticity. An investigator who is seriously interested in knowing how genes might constrain or influence the course of development of any trait in any organism must study directly the norms of reaction of the various genotypes in the population over the range of projected environments. No less detailed information will do. Summary measures such as H2 are not first steps toward a more complete analysis and therefore are not valuable in themselves. MESSAGE Heritability is not the opposite of phenotypic plasticity. A character may have perfect heritability in a population and still be subject to great changes resulting from environmental variation. Locating the genes It is not possible with purely genetic techniques to identify all the genes that influence the development of a given trait. This is true even for simple qualitative traits—for example, the genes that determine the total antigenic configuration of the membrane of the human red blood cell. About 40 loci determining human blood groups are known at present; each has been discovered by finding at least one person with an immunological specificity that differs from the specificities of other people. Many other loci that determine red-cell membrane structure may remain undiscovered because all the individuals studied are genetically identical. Genetic analysis detects genes only when there is some allelic variation. In contrast, molecular analysis, by dealing directly with DNA and its translated information, can identify genes even when they do not vary—provided the gene products can be identified. Even though a trait may show continuous phenotypic variation, the genetic basis for the differences may be allelic variation at a single locus. Most of the classical mutations in Drosophila are phenotypically variable in their expression, and in many cases the mutant class differs little from wild type, so many individuals that carry the mutation are indistinguishable from normal. Even the genes of the bithorax gene complex, which have dramatic homeotic mutations that turn halteres into wings (see pages 698–699), also have 23 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics weak alleles that increase the size of the haltere only slightly on the average, so individuals of the mutant genotype may appear to be wild type. It is sometimes possible to use prior knowledge of the biochemistry and development of an organism to guess that variation at a known locus is responsible for at least some of the variation in phenotype. This locus then is a candidate gene for investigation of continuous phenotypic variation. An example is the variation in activity of the enzyme acid phosphatase in human red blood cells. Because we are dealing with variation in enzyme activity, a good hypothesis would be that there is allelic variation at the locus that encodes this enzyme. When H. Harris and D. Hopkinson sampled an English population, they found that there were, indeed, three allelic forms, A, B, and C, with different activities. Table 25-2 shows the mean activity, the variance in activity, and the population frequency of the six genotypes. Figure 25-11 shows the distribution of activity in the entire population and how it is composed of the distributions of the different genotypes. Table 25-2 shows that, of the variance in activity in the total distribution (607.8), about half is explained by the average variance within genotypes (310.7), so half (607.8 − 310.7 = 297.1) is accounted for by the variance between the means of the six genotypes. Although much of the variation in activity is explained by the mean differences between the genotypes, there remains variation within each genotype that may be the result of environmental influences or of the segregation of other, as yet unidentified, genes. This partial explanation of variation by alleles at a single identified locus is typical of what is found by the candidate gene method, and the proportion of variance associated with the single locus is usually less than what was found for acid phosphatase. For example, the three common alleles for the gene apoE that encodes the protein apolipoprotein E account for only about 16 percent of the variance in blood levels of low-density lipoproteins that carry cholesterol and are implicated in excess cholesterol levels. Marker-gene segregation The genes segregating for a quantitative trait, so-called quantitative trait loci, or QTLs, cannot be individually identified in most cases. It is possible, however, to localize those regions of the genome in which the relevant loci lie and to estimate how much of the total variation is accounted for by QTL variation in each region. This analysis is done in experimental organisms by crossing two lines that differ markedly in the quantitative trait and differ in alleles at well-known loci, marker genes, where the different genotypes can be distinguished by criteria such as some visible phenotypic effect that is not confused with the quantitative trait (say, eye color in Drosophila) or by the electrophoretic mobility of the proteins that they encode or by the DNA sequence of the genes themselves. The F1 between the two lines is then crossed with itself to make a segregating F2 or it may be backcrossed to one of the parental lines. If there are QTLs closely linked to a marker gene, then the different marker genotypes in the segregating generation will also carry the QTL alleles that were linked to them in the original parental lines. Thus different marker genotypes in the F2 or backcross will have different average phenotypes for the quantitative character. Linkage analysis 24 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics The localization of QTLs to small regions within a chromosome requires that there be closely spaced marker loci along the chromosome. Moreover, it must be possible to have parental lines that differ from each other in the alleles carried at these loci. For most of the history of genetics, these requirements could not be met, even in a genetically well known species such as Drosophila, because most marker loci were known from severe morphological mutants that had deleterious effects on the viability and fecundity of their carriers. As a result, it was not possible to create a line that carried large numbers of mutant alleles that would distinguish it from an alternative line carrying the wild-type alleles. With the advent of molecular techniques that can detect genetic polymorphism at the DNA level (see pages 718–721), very high densities of variant loci have been discovered along the chromosomes of all species. Especially useful are restriction-site polymorphisms and tandem repeats in DNA (see pages 718–719, and 720–721). Such polymorphisms are so common that any two lines selected for a difference in quantitative traits are also sure to differ from each other at known molecular marker loci spaced a few crossover units from each other along each chromosome. An experimental protocol for localizing the genes uses groups of individuals that differ markedly in the quantitative trait and differ at marker loci. These groups may be created by a number of generations of divergent selection to create extreme lines or advantage may be taken of already existing varieties of family groups that differ markedly in the trait. These lines must then be surveyed for marker loci that differ between them. A cross is made between the two lines, and the F1 is crossed with itself to produce a segregating F2 or is crossed back to one of the parental lines to produce a segregating backcross. A large number of offspring from the segregating generation are then measured for the quantitative trait and characterized for their genotype at the marker loci. A marker locus that is unlinked or very loosely linked to any QTLs will have the same average value of the quantitative trait for all its genotypes, whereas one that is closely linked to some QTLs will differ in its mean phenotype from one of its genotypes to another. How much difference there is in the mean phenotype between the marker-locus genotypes depends both on the strength of the effect of the QTLs and on the tightness of linkage between the QTLs and the marker locus. Suppose, for example, that there are two selected lines that differ by a total of 100 units in some quantitative character, that the high line is homozygous +/+ at a QTL, whereas the low line is homozygous −/−, and that each + allele at this QTL accounts for 5 units of the total difference between the lines. Further suppose that the high line is M/M and the low line is m/m at a marker locus 10 crossover units away from the QTL. Then, as shown in Figure 25-12, there are 4 units of difference between the average gamete carrying an M allele and an average gamete carrying an m allele in the segregating F2, or 8 units of the difference between the two original homozygous lines. Thus we have accounted for 8 percent of the average difference between the lines, although the QTL actually accounts for 10 percent of the difference. The discrepancy comes from the recombination between the marker gene and the QTL. This technique has been used to locate chromosomal segments associated with traits such as fruit weight in tomatoes, bristle number in Drosophila, and vegetative characters in maize. In the maize case, 82 vegetative characters were examined in a cross between lines that differed in 20 DNA markers. On the average, each trait was significantly associated with 14 different 25 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics markers, but the proportion of the trait difference between the lines that was explained by any given marker was usually very small. Figure 25-13 shows the proportion of the significant marker–trait associations (on the y-axis) that accounted for different proportions of trait difference between the lines. As Figure 25-13 shows, most associations accounted for less than 1 percent of the trait difference between lines. For many organisms (for example, humans), it is not possible to make homozygous lines differing in some trait and then cross them to produce a segregating generation. For such organisms, one can use the differences among sibs carrying different marker alleles from heterozygous parents. This method has much less power to find QTLs especially when the number of sibs in any family is small, as it is in human families. As a consequence, the attempts to map QTLs for human traits have not been very successful, although the segregating marker technique has been a success in finding loci whose mutations are responsible for single-gene disorders. More on analyzing variance Knowledge of the broad heritability (H2) of a trait in a population is not very useful in itself, but a finer subdivision of phenotypic variance can provide important information for plant and animal breeders. The genetic variation and the environmental variation can themselves each be further subdivided to provide information about gene action and the possibility of shaping the genetic composition of a population. Additive and dominance variance Our previous consideration of gene action suggests that the phenotypes of homozygotes and heterozygotes ought to have a simple relation. If one of the alleles encoded a less active gene product or one with no activity at all and if one unit of gene product were sufficient to allow full physiological activity of the organism, then we would expect complete dominance of one allele over the other, as Mendel observed for flower color in peas. If, on the other hand, physiological activity were proportional to the amount of active gene product, we would expect the heterozygote phenotype to be exactly intermediate between the homozygotes (show no dominance). For many quantitative traits, however, neither of these simple cases is the rule. In general, heterozygotes are not exactly intermediate between the two homozygotes but are closer to one or the other (show partial dominance), even though there is an equal mixture of the primary products of the two alleles in the heterozygote. Indeed, in some cases, the heterozygote phenotype may lie outside the phenotypic range of the homozygotes altogether—a feature termed overdominance. For example, newborn babies who are intermediate in size have a higher chance of survival than very large or very small newborns. Thus, if survival were the phenotype of interest, heterozygotes for genes influencing growth rate would show overdominance for fitness although not for growth rate. 26 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics Suppose that two alleles, a and A, segregate at a locus influencing height. In the environments encountered by the population, the mean phenotypes (heights) and frequencies of the three genotypes might be: There is genetic variance in the population; the phenotypic means of the three genotypic classes are different. Some of the variance arises because there is an average effect on phenotype of substituting an allele A for an allele a; that is, the average height of all individuals with A alleles is greater than that of all individuals with a alleles. By defining the average effect of an allele as the average phenotype of all individuals that carry it, we necessarily make the average effect of the allele depend on the frequencies of the genotypes. The average effect is calculated by simply counting the a and A alleles and multiplying them by the heights of the individuals in which they appear. Thus, 0.36 of all the individuals are homozygous a/a, each a/a individual has two a alleles, and the average height of a/a individuals is 10 cm. Heterozygotes make up 0.48 of the population, each has only one a allele, and the average phenotypic measurement of A/a individuals is 18 cm. The total “number” of a alleles is 2(0.36) + 1(0.48). Thus, the average effect of all the a alleles is: and, by a similar argument, This average difference in effect between A and a alleles of 5.60 cm accounts for some of the variance in phenotype—but not for all of it. The heterozygote is not exactly intermediate between the homozygotes; there is some dominance. We would like to separate the so-called additive effect caused by substituting a alleles for A alleles from the variation caused by dominance. The reason is that the effect of selective breeding depends on the additive variation and not on the variation caused by dominance. Thus, for purposes of plant and animal breeding or for making predictions about evolution by natural selection, we must determine the additive variation. An extreme example will illustrate the principle. Suppose that there is overdominance and that the phenotypic means and frequencies of three genotypes are: 27 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics It is apparent (and a calculation like the preceding one will confirm) that there is no average difference between the a and A alleles, because each has an effect of 11 units. So there is no additive variation, although there is obviously variation in phenotype between the genotypes. The largest individuals are heterozygotes. If a breeder attempts to increase height in this population by selective breeding, mating these heterozygotes together will simply reconstitute the original population. Selection will be totally ineffective. This example illustrates the general law that the effect of selection depends on the additive genetic variation and not on genetic variation in general. We partition the total genetic variance in a population into additive genetic variation(S2a, the variance that arises because there is an average difference between the carriers of a alleles and the carriers of A alleles, and a component called the dominance variance(S2d which results from the fact that heterozygotes are not exactly intermediate between the monozygotes. Thus: The components of variance in the first example, where a/a = 10, A/a = 18, and A/A = 20, can be calculated by using the definitions of mean and variance developed earlier in this chapter. Remembering that a mean is the sum of the values of a variable, each weighted by the frequency with which that value occurs (see page 765), we can calculate the mean phenotype to be: The total genetic variance that arises from the variation among the mean phenotypes of the three genotypes is: The frequency of allele a is (by counting alleles): 28 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics and the frequency of the A allele is: The variance of allelic means is then But we want the variance among diploid individuals that results from the allelic effects, and every diploid individual carries two alleles; so: and The total phenotypic variance can now be written as We define a new kind of heritability, the heritability in the narrow sense (h2), as It is this heritability, not to be confused with H2, that is useful in determining whether a program of selective breeding will succeed in changing the population. The greater the h2 is, the greater the difference is between selected parents and the population as a whole that will be preserved in the offspring of the selected parents. MESSAGE The effect of selection depends on the amount of additive genetic variance and not on the genetic variance in general. Therefore, the narrow heritability, h2, not the broad heritability, H2, is relevant for a prediction of response to selection. 29 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics What has been described as the “dominance” variance is really more complicated. It is all the genetic variation that cannot be explained by the average effect of substituting A for a. If more than one locus affects the character, then any epistatic interactions between loci will appear as variance not associated with the average effect of substituting alleles at the A locus. In principle, we can separate this interaction variance(S2i from the dominance variance (S2d In practice, however, this separation cannot be done with any semblance of accuracy, so all the nonadditive variance appears as “dominance” variance. Estimating genetic variance components Genetic components of variance can be estimated from covariance between relatives, but the derivation of these estimates is beyond the scope of an elementary text. There is, however, another way to estimate h2 that reveals its real meaning. If we plot the phenotypes of the offspring against the average phenotypes of their two parents (the midparent value), we may observe a relation like the one illustrated in Figure 25-14. The regression line will pass through the mean of all the parents and the mean of all the offspring, which will be equal to each other because no change has occurred in the population between generations. Moreover, taller parents have taller children and shorter parents have shorter children, so the slope of the line is positive. But the slope is not unity; very short parents have children who are somewhat taller and very tall parents have children who are somewhat shorter than they themselves are. This slope of less than unity for the regression line arises because heritability is less than perfect. If the phenotype were additively inherited with complete fidelity, then the height of the offspring would be identical with the midparent value and the slope of the line would be 1. On the other hand, if the offspring had no heritable similarity to their parents, all parents would have offspring of the same average height and the slope of the line would be 0. This suggests that the slope of the regression line of the offspring value on the midparent value is an estimate of additive heritability. In fact, the relation is precise. The fact that the slope equals the additive heritability now allows us to use h2 to predict the effects of artificial selection. Suppose that we select parents for the next generation who are on the average 2 units above the general mean of the population from which they were chosen. If h2 = 0.5, then the offspring who form the next, selected generation will lie 0.5(2.0) = 1.0 unit above the mean of the present population, because the regression coefficient predicts how much increase in y will result from a unit increase in x. We can define the selection differential as the difference between the selected parents and the unselected mean and the selection response as the difference between their offspring and the preceding generation. Then: or 30 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics The second expression provides us with yet another way to estimate h2: by selecting for one generation and comparing the response with the selection differential. Usually this is carried out for several generations, and the average response is used. Remember that any estimate of h2, just as for H2, depends on the assumption of no greater environmental correlation between closer relatives. Moreover, h2 in one population in one set of environments will not be the same as h2 in a different population at a different time. Figure 25-15 shows the range of heritabilities reported in various studies for a number of traits in chickens. The very small ranges are generally close to zero. For most traits for which a substantial heritability has been reported in some population, there are big differences from study to study. Use of h2 in breeding Even though h2 is a number that applies only to a particular population and a given set of environments, it is still of great practical importance to breeders. A poultry geneticist interested in increasing, say, growth rate is not concerned with the genetic variance over all possible flocks and all environmental distributions. Given a particular flock (or a choice between a few particular flocks) under the environmental conditions approximating present husbandry practice, the question becomes: Can a selection scheme be devised to increase growth rate and, if so, how fast? If one flock has a lot of genetic variance and another only a little, the breeder will choose the former to carry out selection. If the heritability in the chosen flock is very high, then the mean of the population will respond quickly to the selection imposed, because most of the superiority of the selected parents will appear in the offspring. The higher h2 is, the higher the parent–offspring correlation is. If, on the other hand, h2 is low, then only a small fraction of the increased growth rate of the selected parents will appear in the next generation. If h2 is very low, some alternative scheme of selection or husbandry may be needed. In this case, H2 together with h2 can be of use to the breeder. Suppose that h2 and H2 are both low, which means that there is a lot of environmental variance compared with genetic variance. Some scheme of reducing (S2a must be used. One method is to change the husbandry conditions so that environmental variance is lowered. Another is to use family selection. Rather than choosing the best individuals, the breeder allows pairs to produce several progeny, and the mating is selected on the basis of the average performance of the progeny. Averaging over progeny allows uncontrolled environmental and developmental noise variation to be canceled out, and a better estimate of the genotypic difference between pairs can be made so that the best pairs can be chosen as parents of the next generation. If, on the other hand, h2 is low but H2 is high, then there is not much environmental variance. The low h2 is the result of a small amount of additive genetic variance compared with dominance and interaction variance. Such a situation calls for special breeding schemes that 31 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics make use of nonadditive variance. One such scheme is the hybridinbred method, which is used almost universally for corn. A large number of inbred lines are created by selfing. These inbred lines are then crossed in many different combinations (all possible combinations, if this is economically feasible), and the cross that gives the best hybrid is chosen. Then new inbred lines are developed from this best hybrid, and again crosses are made to find the best second-cycle hybrid. This scheme selects for dominance effects because it takes the best heterozygotes; it has been the basis of major genetic advances in hybrid maize yield in North America since 1930. Yield in corn does not appear to have large amounts of nonadditive genetic variance, so it is debatable whether this technique ultimately produces higher-yielding varieties than those that would have resulted from years of simple selection techniques based on additive variance. The hybrid method has been introduced into the breeding of all kinds of plants and animals. Tomatoes and chickens, as examples, are now almost exclusively hybrids. Attempts also have been made to breed hybrid wheat, but thus far the wheat hybrids obtained do not yield consistently better than the nonhybrid varieties now used. MESSAGE The subdivision of genetic variation and environmental variation provides important information about gene action that can be used in plant and animal breeding. Summary Many—perhaps most—of the phenotypic traits that we observe in organisms vary continuously. In many cases, the variation of the trait is determined by more than a single segregating locus. Each of these loci may contribute equally to a particular phenotype, but it is more likely that they contribute unequally. The measurement of these phenotypes and the determination of the contributions of specific alleles to the distribution must be made on a statistical basis in these cases. Some of these variations of phenotype (such as height in some plants) may show a normal distribution around a mean value; others (such as seed weight in some plants) will illustrate a skewed distribution around a mean value. In other characters, the variation in one phenotype may be correlated with the variation in another. A correlation coefficient may be calculated for these two variables. A quantitative character is one for which the average phenotypic differences between genotypes are small compared with the variation between the individuals within the genotypes. This situation may be true even for characters that are influenced by alleles at one locus. The distribution of environments is reflected biologically as a distribution of phenotypes. The transformation of environmental distribution into phenotypic distribution is determined by the norm of reaction. Norms of reaction can be characterized in organisms in which large numbers of genetically identical individuals can be produced. With the use of genetically marked chromosomes, it is possible to determine the relative contributions of different chromosomes to variation in a quantitative trait, to observe 32 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics dominance and epistasis from whole chromosomes, and, in some cases, to map genes that are segregating for a trait. Traits are familial if they are common to members of the same family, for whatever reason. Traits are heritable, however, only if the similarity arises from common genotypes. In experimental organisms, environmental similarities may be readily distinguished from genetic similarities, or heritability. In humans, however, it is very difficult to determine whether a particular trait is heritable. Norm of reaction studies show only small differences between genotypes, and these differences are not consistent over a wide range of environments. Thus, “superior” genotypes in domesticated animals and cultivated plants may be superior only in certain environments. If it should turn out that humans exhibit genetic variation for various mental and emotional traits, this variation is unlikely to favor one genotype over another across a range of environments. The attempt to quantify the influence of genes on a particular trait has led to the determination of heritability in the broad sense (H2). In general, the heritability of a trait is different in each population and each set of environments and cannot be extrapolated from one population and set of environments to another. Because H2 characterizes present populations in present environments only, it is fundamentally flawed as a predictive device. Heritability in the narrow sense, h2, measures the proportion of phenotypic variation that results from substituting one allele for another. This quantity, if large, predicts that selection for a trait will succeed rapidly. If h2 is small, special forms of selection are required. Solved Problems 1. Two inbred lines of beans are intercrossed. In the F1, the variance in bean weight is measured at 1.5. The F1 is selfed; in the F2, the variance in bean weight is 6.1. Estimate the broad heritability of bean weight in the F2 population of this experiment. Solution The key here is to recognize that all the variance in the F1 population must be environmental because all individuals must be of identical genotype. Furthermore, the F2 variance must be a combination of environmental and genetic components, because all the genes that are heterozygous in the F1 will segregate in the F2 to give an array of different genotypes that relate to bean weight. Hence, we can estimate Therefore and broad heritability is 33 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics 2. In an experimental population of Tribolium (flour beetles), the body length shows a continuous distribution with a mean of 6 mm. A group of males and females with body lengths of 9 mm are removed and interbred. The body lengths of their offspring average 7.2 mm. From these data, calculate the heritability in the narrow sense for body length in this population. Solution The selection differential is 9 − 6 = 3 mm, and the selection response is 7.2 − 6 = 1.2 mm. Therefore, the heritability in the narrow sense is: Problems 1. Distinguish between continuous and discontinuous variation in a population, and give some examples of each. 2. In a large herd of cattle, three different characters showing continuous distribution are measured, and the variances in the following table are calculated: a. Calculate the broad- and narrow-sense heritabilities for each character. b. In the population of animals studied, which character would respond best to selection? Why? c. A project is undertaken to decrease mean fat content in the herd. The mean fat content is currently 10.5 percent. Animals of 6.5 percent fat content are interbred as parents of the next generation. What mean fat content can be expected in the descendants of these animals?See answer 3. Suppose that two triple heterozygotes A/a ; B/b ; C/c are crossed. Assume that the three loci 34 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics are in different chromosomes. a. What proportions of the offspring are homozygous at one, two, and three loci, respectively? b. What proportions of the offspring carry 0, 1, 2, 3, 4, 5, and 6 alleles (represented by capital letters), respectively?See answer 4. In Problem 3, suppose that the average phenotypic effect of the three genotypes at the A locus is A/A = 4, A/a = 3, and a/a = 1 and that similar effects exist for the B and C loci. Moreover, suppose that the effects of loci add to each other. Calculate and graph the distribution of phenotypes in the population (assuming no environmental variance). 5. In Problem 4, suppose that there is a threshold in the phenotypic character so that, when the phenotypic value is above 9, the individual Drosophila has three bristles; when it is between 5 and 9, the individual has two bristles; and when the value is 4 or less, the individual has one bristle. Describe the outcome of crosses within and between bristle classes. Given the result, could you infer the underlying genetic situation?See answer 6. Suppose that the general form of a distribution of a trait for a given genotype is: over the range of x where f is positive. a. On the same scale, plot the distributions for three genotypes with the following means and environmental variances: b. Plot the phenotypic distribution that would result if the three genotypes were equally frequent in a population. Can you see distinct modes? If so, what are they? 7. The following table shows a distribution of bristle number in Drosophila: 35 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics Calculate the mean, variance, and standard deviation of this distribution. See answer 8. The following sets of hypothetical data represent paired observations on two variables (x, y). Plot each set of data pairs as a scatter diagram. Look at the plot of the points, and make an intuitive guess about the correlation between x and y. Then calculate the correlation coefficient for each set of data pairs, and compare this value with your estimate. a. (1, 1); (2, 2); (3, 3); (4, 4); (5, 5); (6, 6). b. (1, 2); (2, 1); (3, 4); (4, 3); (5, 6); (6, 5). c. (1, 3); (2, 1); (3, 2); (4, 6); (5, 4); (6, 5). d. (1, 5); (2, 3); (3, 1); (4, 6); (5, 4); (6, 2). 9. A book on the problem of heritability of IQ makes the following three statements. Discuss the validity of each statement and its implications about the authors' understanding of h2 and H2. a. “The interesting question then is . . . ‘How heritable?' The answer [0.01] has a very different theoretical and practical application from the answer [0.99].” [The authors are talking about H2.] b. “As a rule of thumb, when education is at issue, H2 is usually the more relevant coefficient, and when eugenics and dysgenics (reproduction of selected individuals) are being discussed, h2 is ordinarily what is called for.” c. “But whether the different ability patterns derive from differences in genes . . . is not relevant to assessing discrimination in hiring. Where it could be relevant is in deciding what, in the long run, might be done to change the situation.” (From J. C. Loehlin, G. Lindzey, and J. N. Spuhler, Race Differences in Intelligence. Copyright © 1975 by W. H. Freeman and Company.)See answer 10. Using the concepts of norms of reaction, environmental distribution, genotypic distribution, and phenotypic distribution, try to restate the following statement in more exact terms: “80 percent of the difference in IQ performance between the two groups is genetic.” What would it mean to talk about the heritability of a difference between two groups? 36 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics 11. Describe an experimental protocol for studies of relatives that could estimate the broad heritability of alcoholism. Remember that you must make an adequate observational definition of the trait itself.See answer 12. A line selected for high bristle number in Drosophila has a mean of 25 sternopleural bristles, whereas a low-selected line has a mean of only 2. Marker stocks involving the two large autosomes II and III are used to create stocks with various mixtures of chromosomes from the high (h) and low (l) lines. The mean number of bristles for each chromosomal combination is as follows: What conclusions can you reach about the distribution of genetic factors and their actions from these data? 13. Suppose that number of eye facets is measured in a population of Drosophila under various temperature conditions. Further suppose that it is possible to estimate total genetic variance (S2g) as well as the phenotypic distribution. Finally, suppose that there are only two genotypes in the population. Draw pairs of norms of reaction that would lead to the following results: a. An increase in mean temperature decreases the phenotypic variance. b. An increase in mean temperature increases H2. c. An increase in mean temperature increases S2g but decreases H2. d. An increase in temperature variance changes a unimodal into a bimodal phenotypic distribution (one norm of reaction is sufficient here). 14. Francis Galton compared the heights of male undergraduates with the heights of their fathers, with the results shown in the graph below. 37 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics The average height of all fathers is the same as the average height of all sons, but the individual height classes are not equal across generations. The very tallest fathers had somewhat shorter sons, whereas the very short fathers had somewhat taller sons. As a result, the best line that can be drawn through the points on the scatter diagram has a slope of about 0.67 (solid line) rather than 1.00 (dashed line). Galton used the term regression to describe this tendency for the phenotype of the sons to be closer than the phenotype of their fathers to the population mean. a. Propose an explanation for this regression. b. How are regression and heritability related here? (Graph after W. F. Bodmer and L. L. Cavalli-Sforza, Genetics, Evolution, and Man. Copyright © 1976 by W. H. Freeman and Company.) Chapter 25* 2. a. Shank length: H2 = 0.200 h2 = 0.150 Neck length: H2 = 0.600 h2 = 0.010 Fat content: H2 = 0.500 h2 = 0.400 b. Fat content would respond best to selection. c. 8.9% 3. a. p(homozygous at 3 loci = 21/23 = 2/8 p(homozygotic at two loci) = 31/23 = 3/8 p(homozygotic at three loci) = 1/23 = 1/8 b. p(0 capital letters) = 1/64 p(4 capital letters) = 15/64 p(1 capital letter) = 3/32 p(5 capital letters) = 3/32 p(2 capital letters) = 15/64 p(6 capital letters) = 1/64 p(3 38 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西. An Introduction to Genetic Analysis Chapter 25 Quantitative Genetics capital letters) = 10/32 5. The population described would be distributed as follows: Note that the three-bristle class contains 7 different genotypes, the two-bristle class contains 19 different genotypes, and the one-bristle class contains only 1 genotype. It would be very difficult to determine the underlying genetic situation by doing controlled crosses and determining progeny frequencies. 7. Mean = 4.7;Variance = 0.2619;Standard deviation = 0.5117 9. a. H2 has meaning only with respect to the population that was studied in the environment in which it was studied. Otherwise, it has no meaning. b. Neither H2 nor h2 is a reliable measure that can be used to generalize from a particular sample to a “universe” of the human population. They certainly should not be used in social decision-making (as implied by the terms eugenics and dysgenics). c. Again, H2 and h2 are not reliable measures, and they should not be used in any decision-making with regard to social problems. 11. First, define alcoholism in behavioral terms. Next, realize that all observations must be limited to the behavior that you used in the definition and that all conclusions from your observations are applicable to only that behavior. To do your data gathering, you must work with a population in which familiarity is distinguished from heritability. In practical terms, this population must consist of persons who are genetically close but who are found in all environments possible. 14. a. If you assume that individuals at the extreme of any spectrum are homozygous, then their offspring are more likely to be heterozygous than the original individuals. That is, they will be less extreme. b. For Galton's data, regression is an estimate of heritability (h2), assuming that there were few environmental differences between father and son. 39 勇者并非无所畏惧,而是判断出有比恐惧更重要的东西.