Cardiotonic Steroids Stimulate Cyclooxygenase

Supplementary data
Methods
DNA-EMSA analysis
Cells were harvested and nuclear extracts were prepared. For binding studies, oligonucleotides containing the CRE or NF-IL6 binding site of COX-2 promoter were used (Table E1). The complementary oligonucleotides were annealed in 20 mM Tris (pH 7.6), 50 mM NaCl, 10 mM MgCl
2
, and 1 mM dithiothreitol. The annealed oligonucleotide was phosphorylated at the 5’ end with [ 32 P] ATP and T
4
polynucleotide kinase. The binding reaction was performed by incubating 5-20 μg of nuclear protein in 20 mM HEPES (pH 7.9),
10% glycerol, 300 μg of bovine serum albumin and 1 μg of poly (dI:dC) in a final volume of 10 μl for 10 min at 25 °C. The labeled oligonucleotide was added to the reaction mixture and allowed to incubate for an additional 20 min at 25 °C. The samples were electrophoresed on a 4% non-denaturing polyacrylamide gel. The gel was then dried and scanned by using a Typhoon
8600 ( Amersham Biosciences, UK ) .
RNA electromobility shift assay (RNA-EMSA)
RNA gel shift assay for assessment of RNA binding of HuR was carried out as previously described with minor modification [23]. The RNA-EMSA experiment was performed by using Lightshift Chemiluminescent EMSA kit, based on the manufactures’ instruction (Pierce, Rockford, Illinois, USA). The sequence of RNA oligonucleotide is
1
5’-UUAAUUUAAUUAUUUAAUAAUAUUUAUAUU-3’, this sequence was synthesized and biotin-labeled (GenePharma, Shanghai, China) for RNA binding experiment.
Table E1: Primers and oligos used in this study
2
Figure E1. Effects of Na + , K + -ATPase inhibitors on PGE
2
release in A549 cells. Cells were treated with ouabain, oleandrin or foliandrin at 100 nM for 12 h. PGE
2
content in the culture medium was measured by ELISA method, and expressed as pg/ml. *** P < 0.001, as compared with the vehicle group. The experiment was performed at least in triplicate.
Figure E2. RT-PCR analysis of COX-2 mRNA level in MLE 12 cells after exposure to ouabain at 0, 25, 50 nM for 6 h. 28S was included as a control.
The experiment was performed at least in triplicate.
Figure E3.
Ouabain-induced PGE
2
release is suppressed in MLE 12 cells after knockdown of Na + , K + ATPase α1 subunit expression. Cells were left untransfected, or transfected with the scramble or Na + , K + ATPase α1 siRNA for 48 h, then treated with ouabain at 50 nM for an additional 12 h. PGE
2 content in the culture medium was measured by ELISA method, and expressed as pg/ml. ### P < 0.001 as compared with Na + , K + ATPase α1 siRNA group. The experiment was performed at least in triplicate.
Figure E4. Ouabain dose-dependently increases COX-2 promoter activity in A549 cells.
C ells were transiently transfected with 1 μg of human full-length
COX-2 promoter reporter plasmid and 50 ng pRL-TK. After transfection, cells were treated with ouabain at varied concentrations for an additional 12 h. The
3
luciferase activity in control cells without ouabain treatment was arbitrarily set at 1.0, fold increase of luciferase activity in cells after ouabain treatment is indicated. The experiment was performed at least in triplicate.
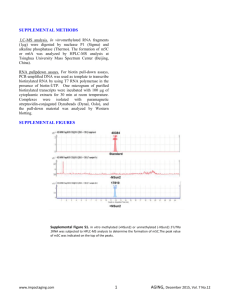
Figure E5. Ouabain time-dependently increases the binding of cells nuclear extract to CRE and NF-IL6/C/EBP binding sequences.
The EMSA experiment was performed as described in the Method. 6 μg nuclear extract was used for CRE binding assay, whereas 15 μg nuclear extract was used for
NF-IL6/C/EBP binding assay. The binding specificity was determined by the addition of 100 ×cold probes. The experiment was performed in duplicate.
Figure E6.
Na + , K + -ATPase inhibitors-induced COX-2 protein expression in A549 cells is suppressed by HuR silencing.
Cells were left untransfected, or transfected with HuR siRNA or scramble siRNA (100 pmol) for 36 h, treated with foliandrin or oleandrin (D) at 100 nM for an additional 12 h. GAPDH was included as a control. The experiment was performed at least in triplicate.
Figure E7. Ouabain fails to trigger HnRNP A1 and AUF1 nuclear export in
A549 cells. Indirect immunofluorescence analysis was performed to visualize changes in the subcellular localization of HuR, HnRNP A1 or AUF1 by ouabain.
Cells were stimulated for 12 h with either vehicle or 100 nM ouabain. After treatment, cells were fixed and stained with HuR, HnRNP A1 or AUF1 antibody, then incubated with FITC or Cy3-conjugated secondary IgG, cellular
4
distribution of HuR, HnRNP A1 or AUF1 was observed under fluorescent microscope. The experiment was performed at least in triplicate.
Figure E8. Na + , K + -ATPase inhibitors increase the RNA binding of cytoplasmic complexes to COX-2 ARE site I. The conditions for RNA binding were described under the Method. For competition analysis, biotin-labeled RNA oligonucleotides with 1000 fold excess nonlabeled oligonucleotides were mixed , and incubated with 10 μg cytoplasmic extracts.
Cytosolic HuR analyzed by western blotting was provided (above panel). The quantitative analysis of RNA binding is shown (bottom panel).The binding intensity in control cells without competition RNA oligonucleotides was arbitrarily set at 100%. ** P < 0.01, *** P < 0.001, drug treatment group vs. control group without competition RNA oligonucleotides; ### P < 0.001, competition RNA oligonucleotides vs. no competition RNA oligonucleotides in each drug-treated group.
The experiment was performed at least in triplicate.
Figure E9. Ouabain induces HuR nuclear export in MLE 12 cells. Cells were challenged with ouabain at 50 nM for 6 h. Representative fluorescent images of 6 independent experiments showing similar results are shown.
Scale bars: 20 μm.
Figure E10. ERK1/2 inhibitor U0126 fails to suppress ouabain-induced stabilization of COX2 3’-UTR.
A549 cells were transfected with 1 μg pC
5
(luciferase reporter plasmid with an SV40 promoter and COX-2 3
’-UTR,
Fig.4B) and 50 ng pRL-TK. After transfection, cells were preincubated with
U0126 (20 μM) for 30 min, then treated with ouabain at 100 nM for an additional 12 h. The luciferase activity in control cells without ouabain treatment was arbitrarily set at 1.0, fold increase of luciferase activity in cells after ouabain treatment is indicated. The experiment was performed at least in triplicate.
Figure E11. Foliandrin-induced HuR nuclear export is attenuated in the presence of SB203580.
A549 cells were preincubated with SB203580 at 20
μM for 1 h, treated with foliandrin (100 nM) for an additional 12 h. HuR cellular distribution was examined by indirect immunofluorescence analysis, as described in the Methods and Materials. The white arrows indicate the absence of HuR protein in the nucleus of A549 cells after foliandrin treatment, and HuR build-up in the nucleus of A549 cells after challenge of foliandrin and
SB203580. Cells treated with SB203580 alone served as a control.
Representative fluorescent images of three independent experiments with similar results are shown. Scale bars: 20 μm.
Figure E12. Effect of BAPTA/AM on the phosphorylation of ERK1/2 and p38 MAPKs in A549 cells challenged with ouabain.
Western blot analysis was performed to measure the amount of total ERK1/2 (t-ERK1/2),
6
phosphorylated ERK1/2 (p-ERK1/2), total p38 MAPK (t-p38) and phosphorylated p38 MAPK (p-p38). The quantitative analysis of ERK1/2 and p38 MAPK phosphorylation is shown in the right panel. ERK1/2 or p38 MAPK phosphorylation in the control group was arbitrarily set at 100%, ### P < 0.001, ouabain vs ouabain+BAPTA/AM. Cells were pre-incubated with BAPTA/AM at
20 μM for 1 h, further incubated with ouabain at 100 nM for 30 min.
7