Solubility Data Tables
advertisement

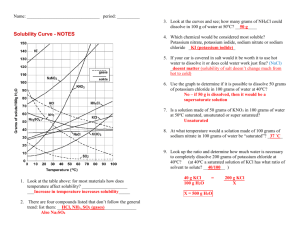
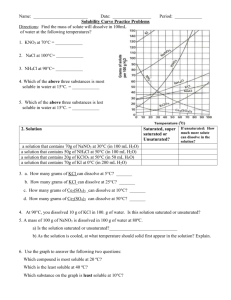
Solubility Data Tables Temperature oC 100 90 80 70 60 50 40 30 20 10 0 Solubility (g/100.0g of water) LiOH 17.5 15.3 13.8 13.3 13.0 12.9 12.8 12.7 12.7 CuSO4 75.4 55.0 40.0 33.3 28.5 25.0 20.7 17.4 14.3 KBr 104.0 99.2 95.0 90.0 85.5 80.2 75.5 70.6 65.2 59.5 53.5 SrBr2 222.5 181.8 150.0 135.8 123.2 111.9 102.4 93.0 85.2 Using the data table above solve the following problems. 1. Can 10g of copper (II) sulfate saturate 100g of water at 40oC? Explain your answer 2. At what temperature will 25g of copper (II) sulfate saturate 100g of water? 3. A solution of strontium bromide is saturated at 20 oC. How much additional solute must be added to keep the solution saturated as the temperature increases to 30 oC? 4. At 100 oC, 100g of water is saturated with lithium hydroxide. The solution is cooled to 0 oC. How many grams of solid lithium hydroxide will settle out of solution? What kind of solution is left after all of the excess settles out? 5. How many grams of potassium bromide will dissolve in 450g of water at 80 oC? 6. How many grams of strontium bromide will dissolve in 258g if water at 50 oC? 7. How many grams of water are needed to dissolve 45g of lithium hydroxide at 100 oC? 8. How many grams water are needed to dissolve 23g of copper (II) sulfate at 60 oC?