CH1 INTRODUCTION AND BASIC CONCEPTS (系上公布 BASIC CONCEPTS OF THERMODYNAMICS) EXAMPLE 1–1 Spotting Errors from Unit Inconsistencies While solving a problem, a person ended up with the following equation at some stage: where E is the total energy and has the unit of kilojoules. Determine how to correct the error and discuss what may have caused it. EXAMPLE 1–2 Obtaining Formulas from Unit Considerations A tank is filled with oil whose density isρ=850 kg/m3. If the volume of the tank is V = 2 m3, determine the amount of mass m in the tank. EXAMPLE 1–3 The Weight of One Pound-Mass Using unity conversion ratios, show that 1.00 lbm weighs 1.00 lbf on earth (Fig. 1–13). EXAMPLE 1–4 Expressing Temperature Rise in Different Units 1 During a heating process, the temperature of a system rises by 10°C. Express this rise in temperature in K, °F, and R. EXAMPLE 1–5 Absolute Pressure of a Vacuum Chamber A vacuum gage connected to a chamber reads 5.8 psi at a location where the atmospheric pressure is 14.5 psi. Determine the absolute pressure in the chamber. EXAMPLE 1–6 Measuring Pressure with a Manometer A manometer is used to measure the pressure in a tank. The fluid used has a specific gravity of 0.85, and the manometer column height is 55 cm, as shown in Fig. 1–46. If the local atmospheric pressure is 96 kPa, determine the absolute pressure within the tank. EXAMPLE 1–7 Measuring Pressure with a Multifluid Manometer The water in a tank is pressurized by air, and the pressure is measured by a multifluid manometer as shown in Fig. 1–49. The tank is located on a mountain at an altitude of 1400 m where the atmospheric pressure is 85.6 kPa. 2 Determine the air pressure in the tank if h1=0.1 m, h2=0.2 m, and h3=0.35 m. Take the densities of water, oil, and mercury to be 1000 kg/m3, 850 kg/m3, and 13,600 kg/m3, respectively. EXAMPLE 1–8 Measuring Atmospheric Pressure with a Barometer Determine the atmospheric pressure at a location where the barometric reading is 740 mm Hg and the gravitational acceleration is g=9.81 m/s2. Assume the temperature of mercury to be 10℃, at which its density is 13,570 kg/m3. EXAMPLE 1–9 Effect of Piston Weight on Pressure in a Cylinder The piston of a vertical piston–cylinder device containing a gas has a mass of 60 kg and a cross-sectional area of 0.04 m2, as shown in Fig. 1–54. The local atmospheric pressure is 0.97 bar, and the gravitational acceleration is 9.81 m/s2. (a) Determine the pressure inside the cylinder. (b) If some heat is transferred to the gas and its volume is doubled, do you expect the pressure inside the cylinder to change? 3 EXAMPLE 1–10 Hydrostatic Pressure in a Solar Pond with Variable Density Solar ponds are small artificial lakes of a few meters deep that are used to store solar energy. The rise of heated (and thus less dense) water to the surface is prevented by adding salt at the pond bottom. In a typical salt gradient solar pond, the density of water increases in the gradient zone, as shown in Fig. 1–55, and the density can be expressed as where r0 is the density on the water surface, z is the vertical distance measured downward from the top of the gradient zone, and H is the thickness of the gradient zone. For H=4 m,ρ0=1040 kg/m3, and a thickness of 0.8 m for the surface zone, calculate the gage pressure at the bottom of the gradient zone. 4 EXAMPLE 1–11 Solving a System of Equations with EES The difference of two numbers is 4, and the sum of the squares of these two numbers is equal to the sum of the numbers plus 20. Determine these two numbers. EXAMPLE 1–12 Analyzing a Multifluid Manometer with EES Reconsider the multifluid manometer discussed in Example 1–7 and replotted in Fig. 1–61. Determine the air pressure in the tank using EES. Also determine what the differential fluid height h3 would be for the same air pressure if the mercury in the last column were replaced by seawater with a density of 1030 kg/m3. 1–3C An office worker claims that a cup of cold coffee on his table warmed up to 80°C by picking up energy from the surrounding air, which is at 25°C. Is there any truth to his claim? Does this process violate any thermodynamic laws? 1–17C What is the difference between intensive and extensive properties? 5 1–20C Define the isothermal, isobaric, and isochoric processes. 1–39C Consider two identical fans, one at sea level and the other on top of a high mountain, running at identical speeds. How would you compare (a) the volume flow rates and (b) the mass flow rates of these two fans? 1–48 Consider a 70-kg woman who has a total foot imprint area of 400 cm2. She wishes to walk on the snow, but the snow cannot withstand pressures greater than 0.5 kPa. Determine the minimum size of the snowshoes needed (imprint area per shoe) to enable her to walk on the snow without sinking. 1–57 A gas is contained in a vertical, frictionless piston–cylinder device. The piston has a mass of 4 kg and a cross-sectional area of 35 cm2. A compressed spring above the piston exerts a force of 60 N on the piston. If the atmospheric pressure is 95 kPa, determine the pressure inside the cylinder. 1–71E The pressure in a natural gas pipeline is measured by the manometer shown in Fig. P1–71E with one of the arms open to the atmosphere where the local atmospheric pressure is 14.2 psia. Determine the absolute pressure in the pipeline. 6 1–76 Consider a double-fluid manometer attached to an air pipe shown in Fig. P1–76. If the specific gravity of one fluid is 13.55, determine the specific gravity of the other fluid for the indicated absolute pressure of air. Take the atmospheric pressure to be 100 kPa. CH3 PROPERTIES OF PURE SUBSTANCES (系上公布 PROPERTIES OF PURE SUBSTANCES) EXAMPLE 3–1 Pressure of Saturated Liquid in a Tank A rigid tank contains 50 kg of saturated liquid water at 90°C. Determine the pressure 7 in the tank and the volume of the tank. EXAMPLE 3–2 Temperature of Saturated Vapor in a Cylinder A piston–cylinder device contains 2 ft3 of saturated water vapor at 50-psia pressure. Determine the temperature and the mass of the vapor inside the cylinder. EXAMPLE 3–3 Volume and Energy Change during Evaporation A mass of 200 g of saturated liquid water is completely vaporized at a constant pressure of 100 kPa. Determine (a) the volume change and (b) the amount of energy transferred to the water. EXAMPLE 3–4 Pressure and Volume of a Saturated Mixture A rigid tank contains 10 kg of water at 90°C. If 8 kg of the water is in the liquid form and the rest is in the vapor form, determine (a) the pressure in the tank and (b) the volume of the tank. EXAMPLE 3–5 Properties of Saturated Liquid–Vapor Mixture An 80-L vessel contains 4 kg of refrigerant-134a at a pressure of 160 kPa. Determine (a) the temperature, (b) the quality, (c) the enthalpy of the refrigerant, and (d) the volume occupied by the vapor phase. EXAMPLE 3–6 Internal Energy of Superheated Vapor Determine the internal energy of water at 20 psia and 400°F. EXAMPLE 3–7 Temperature of Superheated Vapor 8 Determine the temperature of water at a state of P=0.5 MPa and h=2890 kJ/kg. EXAMPLE 3–8 Approximating Compressed Liquid as Saturated Liquid Determine the internal energy of compressed liquid water at 80°C and 5 MPa, using (a) data from the compressed liquid table and (b) saturated liquid data. What is the error involved in the second case? EXAMPLE 3–8 Approximating Compressed Liquid as Saturated Liquid Determine the internal energy of compressed liquid water at 80°C and 5 MPa, using (a) data from the compressed liquid table and (b) saturated liquid data. What is the error involved in the second case? EXAMPLE 3–9 The Use of Steam Tables to Determine Properties Determine the missing properties and the phase descriptions in the following table for water: EXAMPLE 3–10 Mass of Air in a Room Determine the mass of the air in a room whose dimensions are 4 m × 5 m ×6 m at 100 kPa and 25°C. EXAMPLE 3–11 The Use of Generalized Charts Determine the specific volume of refrigerant-134a at 1 MPa and 50°C, using (a) the 9 ideal-gas equation of state and (b) the generalized compressibility chart. Compare the values obtained to the actual value of 0.021796 m3/kg and determine the error involved in each case. EXAMPLE 3–12 Using Generalized Charts to Determine Pressure Determine the pressure of water vapor at 600°F and 0.51431 ft3/lbm, using (a) the steam tables, (b) the ideal-gas equation, and (c) the generalized compressibility chart. EXAMPLE 3–13 Different Methods of Evaluating Gas Pressure Predict the pressure of nitrogen gas at T =175 K and v = 0.00375 m3/kg on the basis of (a) the ideal-gas equation of state, (b) the van der Waals equation of state, (c) the Beattie-Bridgeman equation of state, and (d) the Benedict-Webb-Rubin equation of state. Compare the values obtained to the experimentally determined value of 10,000 kPa. EXAMPLE 3–14 Temperature Drop of a Lake Due to Evaporation On a summer day, the air temperature over a lake is measured to be 25°C. Determine water temperature of the lake when phase equilibrium conditions are established between the water in the lake and the vapor in the air for relative humidities of 10, 80, and 100 percent for the air (Fig. 3–65). 10 3–4C Is there any difference between the intensive properties of saturated vapor at a given temperature and the vapor of a saturated mixture at the same temperature? 3–37 A piston–cylinder device contains 0.85 kg of refrigerant- 134a at _10°C. The piston that is free to move has a mass of 12 kg and a diameter of 25 cm. The local atmospheric pressure is 88 kPa. Now, heat is transferred to refrigerant-134a until the temperature is 15°C. Determine (a) the final pressure, (b) the change in the volume of the cylinder, and (c) the change in the enthalpy of the refrigerant-134a. 3–52 A rigid vessel contains 2 kg of refrigerant-134a at 800 kPa and 120°C. Determine the volume of the vessel and the total internal energy. 3–54 A 0.5-m3 vessel contains 10 kg of refrigerant-134a at 20°C. Determine (a) the pressure, (b) the total internal energy, and (c) the volume occupied by the liquid 11 phase. 3–68 A piston–cylinder device initially contains steam at 3.5 MPa, superheated by 5°C. Now, steam loses heat to the surroundings and the piston moves down hitting a set of stops at which point the cylinder contains saturated liquid water. The cooling continues until the cylinder contains water at 200°C. Determine (a) the initial temperature, (b) the enthalpy change per unit mass of the steam by the time the piston first hits the stops, and (c) the final pressure and the quality (if mixture). 3–97 A 1-m3 tank contains 2.841 kg of steam at 0.6 MPa. Determine the temperature of the steam, using (a) the idealgas equation, (b) the van der Waals equation, and (c) the steam tables. CH2 ENERGY, ENERGY TRANSFER, AND GENERAL ENERGY ANALYSIS CH4 ENERGY ANALYSIS OF CLOSED SYSTEMS CH5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES (系上公布 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES、 12 THIS FIRST LAW OF THERMODYNAMICS) EXAMPLE 2–1 A Car Powered by Nuclear Fuel An average car consumes about 5 L of gasoline a day, and the capacity of the fuel tank of a car is about 50 L. Therefore, a car needs to be refueled once every 10 days. Also, the density of gasoline ranges from 0.68 to 0.78 kg/L, and its lower heating value is about 44,000 kJ/kg (that is, 44,000 Kj of heat is released when 1 kg of gasoline is completely burned). Suppose all the problems associated with the radioactivity and waste disposal of nuclear fuels are resolved, and a car is to be powered by U-235. If a new car comes equipped with 0.1-kg of the nuclear fuel U-235, determine if this car will ever need refueling under average driving conditions (Fig. 2–9). EXAMPLE 2–2 Wind Energy A site evaluated for a wind farm is observed to have steady winds at a speed of 8.5 m/s (Fig. 2–10). Determine the wind energy (a) per unit mass, (b) for a mass of 10 kg, and (c) for a flow rate of 1154 kg/s for air. EXAMPLE 2–3 Burning of a Candle in an Insulated Room A candle is burning in a well-insulated room. Taking the room (the air plus the candle) as the system, determine (a) if there is any heat transfer during this burning process and (b) if there is any change in the internal energy of the system. 13 EXAMPLE 2–4 Heating of a Potato in an Oven A potato initially at room temperature (25°C) is being baked in an oven that is maintained at 200°C, as shown in Fig. 2–21. Is there any heat transfer during this baking process? EXAMPLE 2–5 Heating of an Oven by Work Transfer A well-insulated electric oven is being heated through its heating element. If the entire oven, including the heating element, is taken to be the system, determine whether this is a heat or work interaction. EXAMPLE 2–6 Heating of an Oven by Heat Transfer Answer the question in Example 2–5 if the system is taken as only the air in the oven 14 without the heating element. EXAMPLE 2–7 Power Transmission by the Shaft of a Car Determine the power transmitted through the shaft of a car when the torque applied is 200 N · m and the shaft rotates at a rate of 4000 revolutions per minute (rpm). EXAMPLE 2–8 Power Needs of a Car to Climb a Hill Consider a 1200-kg car cruising steadily on a level road at 90 km/h. Now the car starts climbing a hill that is sloped 30° from the horizontal (Fig. 2–35). If the velocity of the car is to remain constant during climbing, determine the additional power that must be delivered by the engine. 15 EXAMPLE 2–9 Power Needs of a Car to Accelerate Determine the power required to accelerate a 900-kg car shown in Fig. 2–36 from rest to a velocity of 80 km/h in 20 s on a level road. EXAMPLE 2–10 Cooling of a Hot Fluid in a Tank A rigid tank contains a hot fluid that is cooled while being stirred by a paddle wheel. Initially, the internal energy of the fluid is 800 kJ. During the cooling process, the fluid loses 500 kJ of heat, and the paddle wheel does 100 kJ of work on the fluid. Determine the final internal energy of the fluid. Neglect the energy stored in the paddle wheel. 16 EXAMPLE 2–11 Acceleration of Air by a Fan A fan that consumes 20 W of electric power when operating is claimed to discharge air from a ventilated room at a rate of 0.25 kg/s at a discharge velocity of 8 m/s (Fig. 2–48). Determine if this claim is reasonable. EXAMPLE 2–12 Heating Effect of a Fan A room is initially at the outdoor temperature of 25°C. Now a large fan that consumes 200 W of electricity when running is turned on (Fig. 2–49). The heat transfer rate between the room and the outdoor air is given as Q=UA(Ti -To) where U =6 W/m2 · °C is the overall heat transfer coefficient, A =30 m2 is the exposed surface area of the room, and Ti and To are the indoor and outdoor air temperatures, respectively. Determine the indoor air temperature when steady operating conditions are established. 17 EXAMPLE 2–13 Annual Lighting Cost of a Classroom The lighting needs of a classroom are met by 30 fluorescent lamps, each consuming 80 W of electricity (Fig. 2–50). The lights in the classroom are kept on for 12 hours a day and 250 days a year. For a unit electricity cost of 7 cents per kWh, determine annual energy cost of lighting for this classroom. Also, discuss the effect of lighting on the heating and air-conditioning requirements of the room. EXAMPLE 2–14 Conservation of Energy for an Oscillating Steel Ball The motion of a steel ball in a hemispherical bowl of radius h shown in Fig. 2–51 is to be analyzed. The ball is initially held at the highest location at point A, and then it is 18 released. Obtain relations for the conservation of energy of the ball for the cases of frictionless and actual motions. EXAMPLE 2–15 Cost of Cooking with Electric and Gas Ranges The efficiency of cooking appliances affects the internal heat gain from them since an inefficient appliance consumes a greater amount of energy for the same task, and the excess energy consumed shows up as heat in the living space. The efficiency of open burners is determined to be 73 percent for electric units and 38 percent for gas units (Fig. 2–57). Consider a 2-kW electric burner at a location where the unit costs of electricity and natural gas are $0.09/kWh and $0.55/therm, respectively. Determine the rate of energy consumption by the burner and the unit cost of utilized energy for both electric and gas burners. 19 EXAMPLE 2–16 Performance of a Hydraulic Turbine–Generator The water in a large lake is to be used to generate electricity by the installation of a hydraulic turbine–generator at a location where the depth of the water is 50 m (Fig. 2–60). Water is to be supplied at a rate of 5000 kg/s. If the electric power generated is measured to be 1862 kW and the generator efficiency is 95 percent, determine (a) the overall efficiency of the turbine– generator, (b) the mechanical efficiency of the turbine, and (c) the shaft power supplied by the turbine to the generator. EXAMPLE 2–17 Cost Savings Associated with High-Efficiency Motors A 60-hp electric motor (a motor that delivers 60 hp of shaft power at full load) that has an efficiency of 89.0 percent is worn out and is to be replaced by a 93.2 percent 20 efficient high-efficiency motor (Fig. 2–61). The motor operates 3500 hours a year at full load. Taking the unit cost of electricity to be $0.08/kWh, determine the amount of energy and money saved as a result of installing the high-efficiency motor instead of the standard motor. Also, determine the simple payback period if the purchase prices of the standard and high-efficiency motors are $4520 and $5160, respectively. EXAMPLE 2–18 Reducing Air Pollution by Geothermal Heating A geothermal power plant in Nevada is generating electricity using geothermal water extracted at 180°C, and reinjected back to the ground at 85°C. It is proposed to utilize the reinjected brine for heating the residential and commercial buildings in the area, and calculations show that the geothermal heating system can save 18 million therms of natural gas a year. Determine the amount of NOx and CO2 emissions the geothermal system will save a year. Take the average NOx and CO2 emissions of gas furnaces to be 0.0047 kg/therm and 6.4 kg/therm, respectively. 21 EXAMPLE 2–19 Heat Transfer from a Person Consider a person standing in a breezy room at 20°C. Determine the total rate of heat transfer from this person if the exposed surface area and the average outer surface temperature of the person are 1.6 m2 and 29°C, respectively, and the convection heat transfer coefficient is 6 W/m2 · °C (Fig. 2–75). 2–68 A wind turbine is rotating at 15 rpm under steady winds flowing through the turbine at a rate of 42,000 kg/s. The tip velocity of the turbine blade is measured to be 250 km/h. If 180 kW power is produced by the turbine, determine (a) the average velocity of the air and (b) the conversion efficiency of the turbine. Take the density of air to be 1.31 kg/m3. EXAMPLE 4–1 Boundary Work for a Constant-Volume Process A rigid tank contains air at 500 kPa and 150°C. As a result of heat transfer to the surroundings, the temperature and pressure inside the tank drop to 65°C and 400 kPa, respectively. Determine the boundary work done during this process. EXAMPLE 4–2 Boundary Work for a Constant-Pressure Process A frictionless piston–cylinder device contains 10 lbm of steam at 60 psia and 320F. Heat is now transferred to the steam until the temperature reaches 400F. If the piston 22 is not attached to a shaft and its mass is constant, determine the work done by the steam during this process. EXAMPLE 4–3 Isothermal Compression of an Ideal Gas A piston–cylinder device initially contains 0.4 m3 of air at 100 kPa and 80°C. The air is now compressed to 0.1 m3 in such a way that the temperature inside the cylinder remains constant. Determine the work done during this process. EXAMPLE 4–4 Expansion of a Gas against a Spring A piston–cylinder device contains 0.05 m3 of a gas initially at 200 kPa. At this state, a linear spring that has a spring constant of 150 kN/m is touching the piston but exerting no force on it. Now heat is transferred to the gas, causing the piston to rise and to compress the spring until the volume inside the cylinder doubles. If the cross-sectional area of the piston is 0.25 m2, determine (a) the final pressure inside the cylinder, (b) the total work done by the gas, and (c) the fraction of this work done against the spring to compress it. EXAMPLE 4–5 Electric Heating of a Gas at Constant Pressure A piston–cylinder device contains 25 g of saturated water vapor that is maintained at a constant pressure of 300 kPa. A resistance heater within the cylinder is turned on and passes a current of 0.2 A for 5 min from a 120-V source. At the same time, a heat loss of 3.7 kJ occurs. (a) Show that for a closed system the boundary work Wb and the change in internal energy U in the first-law relation can be combined into one term, H, for a constantpressure process. (b) Determine the final temperature of the steam. 23 EXAMPLE 4–6 Unrestrained Expansion of Water A rigid tank is divided into two equal parts by a partition. Initially, one side of the tank contains 5 kg of water at 200 kPa and 25°C, and the other side is evacuated. The partition is then removed, and the water expands into the entire tank. The water is allowed to exchange heat with its surroundings until the temperature in the tank returns to the initial value of 25°C. Determine (a) the volume of the tank, (b) the final pressure, and (c) the heat transfer for this process. EXAMPLE 4–7 Evaluation of the _u of an Ideal Gas Air at 300 K and 200 kPa is heated at constant pressure to 600 K. Determine the change in internal energy of air per unit mass, using (a) data from the air table (Table A–17), (b) the functional form of the specific heat (Table A–2c), and (c) the average specific heat value (Table A–2b). EXAMPLE 4–8 Heating of a Gas in a Tank by Stirring An insulated rigid tank initially contains 1.5 lbm of helium at 80°F and 50 psia. A paddle wheel with a power rating of 0.02 hp is operated within the tank for 30 min. Determine (a) the final temperature and (b) the final pressure of the helium gas. EXAMPLE 4–9 Heating of a Gas by a Resistance Heater A piston–cylinder device initially contains 0.5 m3 of nitrogen gas at 400 kPa and 27°C. An electric heater within the device is turned on and is allowed to pass a current of 2 A for 5 min from a 120 V source. Nitrogen expands at constant pressure, and a heat loss of 2800 J occurs during the process. Determine the final temperature of nitrogen. EXAMPLE 4–10 Heating of a Gas at Constant Pressure 24 A piston–cylinder device initially contains air at 150 kPa and 27°C. At this state, the piston is resting on a pair of stops, as shown in Fig. 4–32, and the enclosed volume is 400 L. The mass of the piston is such that a 350 kPa pressure is required to move it. The air is now heated until its volume has doubled. Determine (a) the final temperature, (b) the work done by the air, and (c) the total heat transferred to the air. EXAMPLE 4–11 Enthalpy of Compressed Liquid Determine the enthalpy of liquid water at 100°C and 15 MPa (a) by using compressed liquid tables, (b) by approximating it as a saturated liquid, and (c) by using the correction given by Eq. 4–38. EXAMPLE 4–12 Cooling of an Iron Block by Water A 50-kg iron block at 80°C is dropped into an insulated tank that contains 0.5 m3 of liquid water at 25°C. Determine the temperature when thermal equilibrium is reached. EXAMPLE 4–13 Temperature Rise due to Slapping If you ever slapped someone or got slapped yourself, you probably remember the burning sensation. Imagine you had the unfortunate occasion of being slapped by an 25 angry person, which caused the temperature of the affected area of your face to rise by 1.8°C (ouch!). Assuming the slapping hand has a mass of 1.2 kg and about 0.150 kg of the tissue on the face and the hand is affected by the incident, estimate the velocity of the hand just before impact. Take the specific heat of the tissue to be 3.8 kJ/kg · °C. EXAMPLE 4–14 Burning Off Lunch Calories A 90 kg man had two hamburgers, a regular serving of french fries, and a 200 ml Coke for lunch (Fig. 4–43). Determine how long it will take for him to burn the lunch calories off (a) by watching TV and (b) by fast swimming. What would your answers be for a 45-kg man? EXAMPLE 4–15 Losing Weight by Switching to Fat-Free Chips The fake fat olestra passes through the body undigested, and thus adds zero calorie to the diet. Although foods cooked with olestra taste pretty good, they may cause abdominal discomfort and the long-term effects are unknown. A 1 oz (28.3 g) serving of regular potato chips has 10 g of fat and 150 Calories, whereas 1 oz of the so-called fat-free chips fried in olestra has only 75 Calories. Consider a person who eats 1 oz of regular potato chips every day at lunch without gaining or losing any weight. Determine how much weight this person will lose in one year if he or she switches to fat-free chips (Fig. 4–44 ). 26 4–6 A piston–cylinder device with a set of stops initially contains 0.3 kg of steam at 1.0 MPa and 400°C. The location of the stops corresponds to 60 percent of the initial volume. Now the steam is cooled. Determine the compression work if the final state is (a) 1.0 MPa and 250°C and (b) 500 kPa. (c) Also determine the temperature at the final state in part (b). 4–12 A mass of 2.4 kg of air at 150 kPa and 12°C is contained in a gas-tight, frictionless piston–cylinder device. The air is now compressed to a final pressure of 600 kPa. During the process, heat is transferred from the air such that the temperature inside the cylinder remains constant. Calculate the work input during this process. 27 4–26 A piston–cylinder device initially contains 0.25 kg of nitrogen gas at 130 kPa and 120°C. The nitrogen is now expanded isothermally to a pressure of 100 kPa. Determine the boundary work done during this process. 4–36 An insulated piston–cylinder device contains 5 L of saturated liquid water at a constant pressure of 175 kPa. Water is stirred by a paddle wheel while a current of 8 A flows for 45 min through a resistor placed in the water. If one-half of the liquid is evaporated during this constantpressure process and the paddle-wheel work amounts to 400 kJ, determine the voltage of the source. Also, show the process on a P-v diagram with respect to saturation lines. 4–69 A room is heated by a baseboard resistance heater. When the heat losses from the room on a winter day amount to 6500 kJ/h, the air temperature in the room remains constant even though the heater operates continuously. Determine the power 28 rating of the heater, in kW. EXAMPLE 5–1 Water Flow through a Garden Hose Nozzle A garden hose attached with a nozzle is used to fill a 10-gal bucket. The inner diameter of the hose is 2 cm, and it reduces to 0.8 cm at the nozzle exit (Fig. 5–9). If it takes 50 s to fill the bucket with water, determine (a) the volume and mass flow rates of water through the hose, and (b) the average velocity of water at the nozzle exit. EXAMPLE 5–2 Discharge of Water from a Tank A 4-ft-high, 3-ft-diameter cylindrical water tank whose top is open to the atmosphere is initially filled with water. Now the discharge plug near the bottom of the tank is pulled out, and a water jet whose diameter is 0.5 in streams out (Fig. 5–10). The 29 average velocity of the jet is given by where h is the height of water in the tank measured from the center of the hole (a variable) and g is the gravitational acceleration. Determine how long it will take for the water level in the tank to drop to 2 ft from the bottom. EXAMPLE 5–3 Energy Transport by Mass Steam is leaving a 4-L pressure cooker whose operating pressure is 150 kPa (Fig. 5–16). It is observed that the amount of liquid in the cooker has decreased by 0.6 L in 40 min after the steady operating conditions are established, and the cross-sectional area of the exit opening is 8 mm2. Determine (a) the mass flow rate of the steam and the exit velocity, (b) the total and flow energies of the steam per unit mass, and (c) the rate at which energy leaves the cooker by steam. EXAMPLE 5–4 Deceleration of Air in a Diffuser Air at 10°C and 80 kPa enters the diffuser of a jet engine steadily with a velocity of 200 m/s. The inlet area of the diffuser is 0.4 m2. The air leaves the diffuser with a 30 velocity that is very small compared with the inlet velocity. Determine (a) the mass flow rate of the air and (b) the temperature of the air leaving the diffuser. EXAMPLE 5–5 Acceleration of Steam in a Nozzle Steam at 250 psia and 700°F steadily enters a nozzle whose inlet area is 0.2 ft2. The mass flow rate of steam through the nozzle is 10 lbm/s. Steam leaves the nozzle at 200 psia with a velocity of 900 ft/s. Heat losses from the nozzle per unit mass of the steam are estimated to be 1.2 Btu/lbm. Determine (a) the inlet velocity and (b) the exit temperature of the steam. EXAMPLE 5–6 Compressing Air by a Compressor Air at 100 kPa and 280 K is compressed steadily to 600 kPa and 400 K. The mass flow rate of the air is 0.02 kg/s, and a heat loss of 16 kJ/kg occurs during the process. Assuming the changes in kinetic and potential energies are negligible, determine the necessary power input to the compressor. EXAMPLE 5–7 Power Generation by a Steam Turbine The power output of an adiabatic steam turbine is 5 MW, and the inlet and the exit conditions of the steam are as indicated in Fig. 5–28. (a) Compare the magnitudes of h, ke, and pe. (b) Determine the work done per unit mass of the steam flowing through the turbine. (c) Calculate the mass flow rate of the steam. 31 EXAMPLE 5–8 Expansion of Refrigerant-134a in a Refrigerator Refrigerant-134a enters the capillary tube of a refrigerator as saturated liquid at 0.8 MPa and is throttled to a pressure of 0.12 MPa. Determine the quality of the refrigerant at the final state and the temperature drop during this process. EXAMPLE 5–9 Mixing of Hot and Cold Waters in a Shower Consider an ordinary shower where hot water at 140°F is mixed with cold water at 50°F. If it is desired that a steady stream of warm water at 110°F be supplied, determine the ratio of the mass flow rates of the hot to cold water. Assume the heat losses from the mixing chamber to be negligible and the mixing to take place at a pressure of 20 psia. EXAMPLE 5–10 Cooling of Refrigerant-134a by Water Refrigerant-134a is to be cooled by water in a condenser. The refrigerant enters the condenser with a mass flow rate of 6 kg/min at 1 MPa and 70°C and leaves at 35°C. The cooling water enters at 300 kPa and 15°C and leaves at 25°C. Neglecting any 32 pressure drops, determine (a) the mass flow rate of the cooling water required and (b) the heat transfer rate from the refrigerant to water. EXAMPLE 5–11 Electric Heating of Air in a House The electric heating systems used in many houses consist of a simple duct with resistance heaters. Air is heated as it flows over resistance wires. Consider a 15 kW electric heating system. Air enters the heating section at 100 kPa and 17°C with a volume flow rate of 150 m3/min. If heat is lost from the air in the duct to the surroundings at a rate of 200 W, determine the exit temperature of air. EXAMPLE 5–12 Charging of a Rigid Tank by Steam A rigid, insulated tank that is initially evacuated is connected through a valve to a supply line that carries steam at 1 MPa and 300°C. Now the valve is opened, and steam is allowed to flow slowly into the tank until the pressure reaches 1 MPa, at which point the valve is closed. Determine the final temperature of the steam in the tank. EXAMPLE 5–13 Cooking with a Pressure Cooker A pressure cooker is a pot that cooks food much faster than ordinary pots by maintaining a higher pressure and temperature during cooking. The pressure inside the pot is controlled by a pressure regulator (the petcock) that keeps the pressure at a constant level by periodically allowing some steam to escape, thus preventing any excess pressure buildup. Pressure cookers, in general, maintain a gage pressure of 2 atm (or 3 atm absolute) inside. Therefore, pressure cookers cook at a temperature of about 133°C (or 271°F) instead of 100°C (or 212°F), cutting the cooking time by as much as 70 percent while 33 minimizing the loss of nutrients. The newer pressure cookers use a spring valve with several pressure settings rather than a weight on the cover. A certain pressure cooker has a volume of 6 L and an operating pressure of 75 kPa gage. Initially, it contains 1 kg of water. Heat is supplied to the pressure cooker at a rate of 500 W for 30 min after the operating pressure is reached. Assuming an atmospheric pressure of 100 kPa, determine (a) the temperature at which cooking takes place and (b) the amount of water left in the pressure cooker at the end of the process. 5–3C Does the amount of mass entering a control volume have to be equal to the amount of mass leaving during an unsteady-flow process? 5–8 A hair dryer is basically a duct of constant diameter in which a few layers of electric resistors are placed. A small fan pulls the air in and forces it through the resistors where it is heated. If the density of air is 1.20 kg/m 3 at the inlet and 1.05 kg/m3 at the exit, determine the percent increase in the velocity of air as it flows through the dryer. 34 5–12 A desktop computer is to be cooled by a fan whose flow rate is 0.34 m3/min. Determine the mass flow rate of air through the fan at an elevation of 3400 m where the air density is 0.7 kg/m3. Also, if the average velocity of air is not to exceed 110 m/min, determine the diameter of the casing of the fan. 5–17 Consider a 300-L storage tank of a solar water heating system initially filled with warm water at 45°C. Warm water is withdrawn from the tank through a 2-cm diameter hose at an average velocity of 0.5 m/s while cold water enters the tank at 20°C at a rate of 5 L/min. Determine the amount of water in the tank after a 20-minute period. Assume the pressure in the tank remains constant at 1 atm. 5–30 Air enters an adiabatic nozzle steadily at 300 kPa, 200°C, and 30 m/s and leaves at 100 kPa and 180 m/s. The inlet area of the nozzle is 80 cm2. Determine (a) the mass flow rate through the nozzle, (b) the exit temperature of the air, and (c) the exit area of the nozzle. 35 5–54 Argon gas enters an adiabatic turbine steadily at 900 kPa and 450°C with a velocity of 80 m/s and leaves at 150 kPa with a velocity of 150 m/s. The inlet area of the turbine is 60 cm2. If the power output of the turbine is 250 kW, determine the exit temperature of the argon. 5–66 Refrigerant-134a is throttled from the saturated liquid state at 700 kPa to a pressure of 160 kPa. Determine the temperature drop during this process and the final specific volume of the refrigerant. CH6 THE SECOND LAW OF THERMODYNAMICS (系上公布 THE SECOND LAW OF THERMODYNAMICS) EXAMPLE 6–1 Net Power Production of a Heat Engine 36 Heat is transferred to a heat engine from a furnace at a rate of 80 MW. If the rate of waste heat rejection to a nearby river is 50 MW, determine the net power output and the thermal efficiency for this heat engine. EXAMPLE 6–2 Fuel Consumption Rate of a Car A car engine with a power output of 65 hp has a thermal efficiency of 24 percent. Determine the fuel consumption rate of this car if the fuel has a heating value of 19,000 Btu/lbm (that is, 19,000 Btu of energy is released for each lbm of fuel burned). EXAMPLE 6–3 Heat Rejection by a Refrigerator The food compartment of a refrigerator, shown in Fig. 6–24, is maintained at 4°C by removing heat from it at a rate of 360 kJ/min. If the required power input to the refrigerator is 2 kW, determine (a) the coefficient of performance of the refrigerator and (b) the rate of heat rejection to the room that houses the refrigerator. EXAMPLE 6–4 Heating a House by a Heat Pump A heat pump is used to meet the heating requirements of a house and maintain it at 20°C. On a day when the outdoor air temperature drops to 2°C, the house is estimated to lose heat at a rate of 80,000 kJ/h. If the heat pump under these conditions has a COP of 2.5, determine (a) the power consumed by the heat pump and (b) the rate at 37 which heat is absorbed from the cold outdoor air. EXAMPLE 6–5 Analysis of a Carnot Heat Engine A Carnot heat engine, shown in Fig. 6–48, receives 500 kJ of heat per cycle from a high-temperature source at 652°C and rejects heat to a low-temperature sink at 30°C. Determine (a) the thermal efficiency of this Carnot engine and (b) the amount of heat rejected to the sink per cycle. EXAMPLE 6–6 A Questionable Claim for a Refrigerator An inventor claims to have developed a refrigerator that maintains the refrigerated space at 35°F while operating in a room where the temperature is 75°F and that has a COP of 13.5. Is this claim reasonable? EXAMPLE 6–7 Heating a House by a Carnot Heat Pump A heat pump is to be used to heat a house during the winter, as shown in Fig. 6–53. The house is to be maintained at 21°C at all times. The house is estimated to be losing heat at a rate of 135,000 kJ/h when the outside temperature drops to 5°C. Determine the minimum power required to drive this heat pump. 38 EXAMPLE 6–8 Malfunction of a Refrigerator Light Switch The interior lighting of refrigerators is provided by incandescent lamps whose switches are actuated by the opening of the refrigerator door. Consider a refrigerator whose 40-W lightbulb remains on continuously as a result of a malfunction of the switch (Fig. 6–58). If the refrigerator has a coefficient of performance of 1.3 and the cost of electricity is 8 cents per kWh, determine the increase in the energy consumption of the refrigerator and its cost per year if the switch is not fixed. 6–6C What is a thermal energy reservoir? Give some examples. 6–11C Consider a pan of water being heated (a) by placing it on an electric range and (b) by placing a heating element in the water. Which method is a more efficient way of heating water? Explain. 39 6–21 An automobile engine consumes fuel at a rate of 28 L/h and delivers 60 kW of power to the wheels. If the fuel has a heating value of 44,000 kJ/kg and a density of 0.8 g/cm3, determine the efficiency of this engine. 6–28 A coal-burning steam power plant produces a net power of 300 MW with an overall thermal efficiency of 32 percent. The actual gravimetric air–fuel ratio in the furnace is calculated to be 12 kg air/kg fuel. The heating value of the coal is 28,000 kJ/kg. Determine (a) the amount of coal consumed during a 24-hour period and (b) the rate of air flowing through the furnace. 6–32C A heat pump is a device that absorbs energy from the cold outdoor air and transfers it to the warmer indoors. Is this a violation of the second law of thermodynamics? Explain. 6–47 Determine the COP of a heat pump that supplies energy to a house at a rate of 8000 kJ/h for each kW of electric power it draws. Also, determine the rate of energy absorption from the outdoor air. 6–51 A heat pump is used to maintain a house at a constant temperature of 23°C. The house is losing heat to the outside air through the walls and the windows at a rate of 60,000 kJ/h while the energy generated within the house from people, lights, and appliances amounts to 4000 kJ/h. For a COP of 2.5, determine the required power input to the heat pump. 6–55 Refrigerant-134a enters the evaporator coils placed at the back of the freezer 40 section of a household refrigerator at 120 kPa with a quality of 20 percent and leaves at 120 kPa and _20°C. If the compressor consumes 450 W of power and the COP the refrigerator is 1.2, determine (a) the mass flow rate of the refrigerant and (b) the rate of heat rejected to the kitchen air. 6–59C Why are engineers interested in reversible processes even though they can never be achieved? 6–71 A Carnot heat engine operates between a source at 1000 K and a sink at 300 K. If the heat engine is supplied with heat at a rate of 800 kJ/min, determine (a) the thermal efficiency and (b) the power output of this heat engine. 6–90 A Carnot refrigerator operates in a room in which the temperature is 25°C. The refrigerator consumes 500 W of power when operating and has a COP of 4.5. Determine (a) the rate of heat removal from the refrigerated space and (b) the temperature of the refrigerated space. 6–95 A heat pump is used to maintain a house at 22°C by extracting heat from the outside air on a day when the outside air temperature is 2°C. The house is estimated to lose heat at a rate of 110,000 kJ/h, and the heat pump consumes 5 kW of electric power when running. Is this heat pump powerful enough to do the job? 41 CH7 Entropy (系上公布 Entropy) EXAMPLE 7–1 Entropy Change during an Isothermal Process A piston–cylinder device contains a liquid–vapor mixture of water at 300 K. During a constant-pressure process, 750 kJ of heat is transferred to the water. As a result, part of the liquid in the cylinder vaporizes. Determine the entropy change of the water during this process. EXAMPLE 7–2 Entropy Generation during Heat Transfer Processes A heat source at 800 K loses 2000 kJ of heat to a sink at (a) 500 K and (b) 750 K. Determine which heat transfer process is more irreversible. EXAMPLE 7–3 Entropy Change of a Substance in a Tank A rigid tank contains 5 kg of refrigerant-134a initially at 20°C and 140 kPa. The refrigerant is now cooled while being stirred until its pressure drops to 100 kPa. Determine the entropy change of the refrigerant during this process. 42 EXAMPLE 7–4 Entropy Change during a Constant-Pressure Process A piston–cylinder device initially contains 3 lbm of liquid water at 20 psia and 70°F. The water is now heated at constant pressure by the addition of 3450 Btu of heat. Determine the entropy change of the water during this process. EXAMPLE 7–5 Isentropic Expansion of Steam in a Turbine Steam enters an adiabatic turbine at 5 MPa and 450°C and leaves at a pressure of 1.4 MPa. Determine the work output of the turbine per unit mass of steam if the process is reversible. EXAMPLE 7–6 The T-S Diagram of the Carnot Cycle Show the Carnot cycle on a T-S diagram and indicate the areas that represent the heat supplied QH, heat rejected QL, and the net work output Wnet,out on this diagram. EXAMPLE 7–7 Effect of Density of a Liquid on Entropy Liquid methane is commonly used in various cryogenic applications. The critical temperature of methane is 191 K (or _82°C), and thus methane must be maintained below 191 K to keep it in liquid phase. The properties of liquid methane at various temperatures and pressures are given in Table 7–1. Determine the entropy change of liquid methane as it undergoes a process from 110 K and 1 MPa to 120 K and 5 MPa (a) using tabulated properties and (b) approximating liquid methane as an 43 incompressible substance. What is the error involved in the latter case? EXAMPLE 7–8 Economics of Replacing a Valve by a Turbine A cryogenic manufacturing facility handles liquid methane at 115 K and 5 MPa at a rate of 0.280 m3/s . A process requires dropping the pressure of liquid methane to 1 MPa, which is done by throttling the liquid methane by passing it through a flow resistance such as a valve. A recently hired engineer proposes to replace the throttling valve by a turbine in order to produce power while dropping the pressure to 1 MPa. Using data from Table 7–1, determine the maximum amount of power that can be produced by such a turbine. Also, determine how much this turbine will save the facility from electricity usage costs per year if the turbine operates continuously (8760 h/yr) and the facility pays $0.075/kWh for electricity. EXAMPLE 7–9 Entropy Change of an Ideal Gas Air is compressed from an initial state of 100 kPa and 17°C to a final state of 600 kPa and 57°C. Determine the entropy change of air during this compression process by using (a) property values from the air table and (b) average specific heats. 44 EXAMPLE 7–10 Isentropic Compression of Air in a Car Engine Air is compressed in a car engine from 22°C and 95 kPa in a reversible and adiabatic manner. If the compression ratio V1/V2 of this engine is 8, determine the final temperature of the air. EXAMPLE 7–11 Isentropic Compression of an Ideal Gas Helium gas is compressed by an adiabatic compressor from an initial state of 14 psia and 50°F to a final temperature of 320°F in a reversible manner. Determine the exit pressure of helium. EXAMPLE 7–12 Compressing a Substance in the Liquid versus Gas Phases Determine the compressor work input required to compress steam isentropically from 100 kPa to 1 MPa, assuming that the steam exists as (a) saturated liquid and (b) saturated vapor at the inlet state. EXAMPLE 7–13 Work Input for Various Compression Processes Air is compressed steadily by a reversible compressor from an inlet state of 100 kPa and 300 K to an exit pressure of 900 kPa. Determine the compressor work per unit mass for (a) isentropic compression with k=1.4, (b) polytropic compression with n = 1.3, (c) isothermal compression, and (d) ideal twostage compression with intercooling with a polytropic exponent of 1.3. EXAMPLE 7–14 Isentropic Efficiency of a Steam Turbine Steam enters an adiabatic turbine steadily at 3 MPa and 400°C and leaves at 50 kPa 45 and 100°C. If the power output of the turbine is 2 MW, determine (a) the isentropic efficiency of the turbine and (b) the mass flow rate of the steam flowing through the turbine. EXAMPLE 7–15 Effect of Efficiency on Compressor Power Input Air is compressed by an adiabatic compressor from 100 kPa and 12°C to a pressure of 800 kPa at a steady rate of 0.2 kg/s. If the isentropic efficiency of the compressor is 80 percent, determine (a) the exit temperature of air and (b) the required power input to the compressor. EXAMPLE 7–16 Effect of Efficiency on Nozzle Exit Velocity Air at 200 kPa and 950 K enters an adiabatic nozzle at low velocity and is discharged at a pressure of 80 kPa. If the isentropic efficiency of the nozzle is 92 percent, determine (a) the maximum possible exit velocity, (b) the exit temperature, and (c) the actual exit velocity of the air. Assume constant specific heats for air. EXAMPLE 7–17 Entropy Generation in a Wall Consider steady heat transfer through a 5 m ×7 m brick wall of a house of thickness 30 cm. On a day when the temperature of the outdoors is 0℃, the house is maintained at 27℃. The temperatures of the inner and outer surfaces of the brick wall are measured to be 20℃ and 5℃, respectively, and the rate of heat transfer through the wall is 1035 W. Determine the rate of entropy generation in the wall, and the rate of total entropy generation associated with this heat transfer process. EXAMPLE 7–18 Entropy Generation during a Throttling Process 46 Steam at 7 MPa and 450℃is throttled in a valve to a pressure of 3 MPa during a steady-flow process. Determine the entropy generated during this process and check if the increase of entropy principle is satisfied. EXAMPLE 7–19 Entropy Generated when a Hot Block Is Dropped in a Lake A 50-kg block of iron casting at 500 K is thrown into a large lake that is at a temperature of 285 K. The iron block eventually reaches thermal equilibrium with the lake water. Assuming an average specific heat of 0.45 kJ/kg · K for the iron, determine (a) the entropy change of the iron block, (b) the entropy change of the lake water, and (c) the entropy generated during this process. EXAMPLE 7–20 Entropy Generation in a Mixing Chamber Water at 20 psia and 50 F enters a mixing chamber at a rate of 300 lbm/min where it is mixed steadily with steam entering at 20 psia and 240 F. The mixture leaves the chamber at 20 psia and 130 F, and heat is lost to the surrounding air at 70 F at a rate of 180 Btu/min. Neglecting the changes in kinetic and potential energies, determine the rate of entropy generation during this process. CH8 EXERGY: A MEASURE OF WORK POTENTIAL (系上公布 EXERGY: A MEASURE OF WORK POTENTIAL) EXAMPLE 8–1 Maximum Power Generation by a Wind Turbine A wind turbine with a 12-m-diameter rotor, as shown in Fig. 8–6, is to be installed at a location where the wind is blowing steadily at an average velocity of 10 m/s. Determine the maximum power that can be generated by the wind turbine. 47 EXAMPLE 8–2 Exergy Transfer from a Furnace Consider a large furnace that can transfer heat at a temperature of 2000 R at a steady rate of 3000 Btu/s. Determine the rate of exergy flow associated with this heat transfer. Assume an environment temperature of 77°F. EXAMPLE 8–3 The Rate of Irreversibility of a Heat Engine A heat engine receives heat from a source at 1200 K at a rate of 500 kJ/s and rejects the waste heat to a medium at 300 K (Fig. 8–11). The power output of the heat engine is 180 kW. Determine the reversible power and the irreversibility rate for this process. 48 EXAMPLE 8–4 Irreversibility during the Cooling of an Iron Block A 500-kg iron block shown in Fig. 8–12 is initially at 200°C and is allowed to cool to 27°C by transferring heat to the surrounding air at 27°C. Determine the reversible work and the irreversibility for this process. EXAMPLE 8–5 Heating Potential of a Hot Iron Block The iron block discussed in Example 8–4 is to be used to maintain a house at 27°C when the outdoor temperature is 5°C. Determine the maximum amount of heat that can be supplied to the house as the iron cools to 27°C. EXAMPLE 8–6 Second-Law Efficiency of Resistance Heaters A dealer advertises that he has just received a shipment of electric resistance heaters for residential buildings that have an efficiency of 100 percent (Fig. 8–19). Assuming an indoor temperature of 21°C and outdoor temperature of 10°C, determine the 49 second-law efficiency of these heaters. EXAMPLE 8–7 Work Potential of Compressed Air in a Tank A 200 m3 rigid tank contains compressed air at 1 MPa and 300 K. Determine how much work can be obtained from this air if the environment conditions are 100 kPa and 300 K. EXAMPLE 8–8 Exergy Change during a Compression Process Refrigerant-134a is to be compressed from 0.14 MPa and 10°C to 0.8 MPa and 50°C steadily by a compressor. Taking the environment conditions to be 20°C and 95 kPa, determine the exergy change of the refrigerant during this process and the minimum work input that needs to be supplied to the compressor per unit mass of the refrigerant. EXAMPLE 8–9 General Exergy Balance for Closed Systems Starting with energy and entropy balances, derive the general exergy balance relation for a closed system (Eq. 8–41). EXAMPLE 8–10 Exergy Destruction during Heat Conduction Consider steady heat transfer through a 5 m ×6 m brick wall of a house of thickness 30 cm. On a day when the temperature of the outdoors is 0°C, the house is maintained 50 at 27°C. The temperatures of the inner and outer surfaces of the brick wall are measured to be 20°C and 5°C, respectively, and the rate of heat transfer through the wall is 1035 W. Determine the rate of exergy destruction in the wall, and the rate of total exergy destruction associated with this heat transfer process. EXAMPLE 8–11 Exergy Destruction during Expansion of Steam A piston–cylinder device contains 0.05 kg of steam at 1 MPa and 300°C. Steam now expands to a final state of 200 kPa and 150°C, doing work. Heat losses from the system to the surroundings are estimated to be 2 kJ during this process. Assuming the surroundings to be at T0=25°C and P0=100 kPa, determine (a) the exergy of the steam at the initial and the final states, (b) the exergy change of the steam, (c) the exergy destroyed, and (d) the second-law efficiency for the process. EXAMPLE 8–12 Exergy Destroyed during Stirring of a Gas An insulated rigid tank contains 2 lbm of air at 20 psia and 70°F. A paddle wheel inside the tank is now rotated by an external power source until the temperature in the tank rises to 130°F (Fig. 8–38). If the surrounding air is at T0=70°F, determine (a) the exergy destroyed and (b) the reversible work for this process. EXAMPLE 8–13 Dropping a Hot Iron Block into Water A 5-kg block initially at 350°C is quenched in an insulated tank that contains 100 kg of water at 30°C (Fig. 8–40). Assuming the water that vaporizes during the process condenses back in the tank and the surroundings are at 20°C and 100 kPa, determine (a) the final equilibrium temperature, (b) the exergy of the combined system at the initial and the final states, and (c) the wasted work potential during this process. 51 EXAMPLE 8–14 Exergy Destruction during Heat Transfer to a Gas A frictionless piston–cylinder device, shown in Fig. 8–41, initially contains 0.01 m3 of argon gas at 400 K and 350 kPa. Heat is now transferred to the argon from a furnace at 1200 K, and the argon expands isothermally until its volume is doubled. No heat transfer takes place between the argon and the surrounding atmospheric air, which is at T0=300 K and P0=100 kPa. Determine (a) the useful work output, (b) the exergy destroyed, and (c) the reversible work for this process. EXAMPLE 8–15 Second-Law Analysis of a Steam Turbine Steam enters a turbine steadily at 3 MPa and 450°C at a rate of 8 kg/s and exits at 0.2 MPa and 150°C, (Fig. 8–45). The steam is losing heat to the surrounding air at 100 kPa and 25°C at a rate of 300 kW, and the kinetic and potential energy changes are negligible. Determine (a) the actual power output, (b) the maximum possible power output, (c) the second-law efficiency, (d) the exergy destroyed, and (e) the exergy of the steam at the inlet conditions. 52 EXAMPLE 8–16 Exergy Destroyed during Mixing of Fluid Streams Water at 20 psia and 50°F enters a mixing chamber at a rate of 300 lbm/min, where it is mixed steadily with steam entering at 20 psia and 240°F. The mixture leaves the chamber at 20 psia and 130 °F, and heat is being lost to the surrounding air at T0=70 °F at a rate of 180 Btu/min (Fig. 8–46). Neglecting the changes in kinetic and potential energies, determine the reversible power and the rate of exergy destruction for this process. EXAMPLE 8–17 Charging a Compressed Air Storage System A 200 m3 rigid tank initially contains atmospheric air at 100 kPa and 300K and is to be used as a storage vessel for compressed air at 1 MPa and 300K (Fig. 8–48). 53 Compressed air is to be supplied by a compressor that takes in atmospheric air at P0= 100 kPa and T0=300 K. Determine the minimum work requirement for this process. 8–31 The radiator of a steam heating system has a volume of 20 L and is filled with superheated water vapor at 200 kPa and 200°C. At this moment both the inlet and the exit valves to the radiator are closed. After a while it is observed that the temperature of the steam drops to 80°C as a result of heat transfer to the room air, which is at 21°C. Assuming the surroundings to be at 0°C, determine (a) the amount of heat transfer to the room and (b) the maximum amount of heat that can be supplied to the room if this heat from the radiator is supplied to a heat engine that is driving a heat pump. Assume the heat engine operates between the radiator and the surroundings. 8–36 An insulated piston–cylinder device contains 2 L of saturated liquid water at a constant pressure of 150 kPa. An electric resistance heater inside the cylinder is 54 turned on, and electrical work is done on the water in the amount of 2200 kJ. Assuming the surroundings to be at 25°C and 100 kPa, determine (a) the minimum work with which this process could be accomplished and (b) the exergy destroyed during this process. 8–49 An ordinary egg can be approximated as a 5.5cm diameter sphere. The egg is initially at a uniform temperature of 8°C and is dropped into boiling water at 97°C. Taking the properties of egg to be ρ=1020 kg/m3 and Cp=3.32 kJ/kg · °C, determine how much heat is transferred to the egg by the time the average temperature of the egg rises to 70°C and the amount of exergy destruction associated with this heat transfer process. Take T0=25°C. 55 CH12 THERMODYNAMIC PROPERTY RELATIONS (系上公布 THERMODYNAMIC PROPERTY RELATIONS) EXAMPLE 12–1 Approximating Differential Quantities by Differences The Cp of ideal gases depends on temperature only, and it is expressed as Cp(T )= dh(T )/dT. Determine the cp of air at 300 K, using the enthalpy data from Table A–17, and compare it to the value listed in Table A–2b. EXAMPLE 12–2 Total Differential versus Partial Differential Consider air at 300 K and 0.86 m3/kg. The state of air changes to 302 K and 0.87 m3/kg as a result of some disturbance. Using Eq. 12–3, estimate the change in the pressure of air. EXAMPLE 12–3 Verification of Cyclic and Reciprocity Relations Using the ideal-gas equation of state, verify (a) the cyclic relation and (b) the reciprocity relation at constant P. EXAMPLE 12–4 Verification of the Maxwell Relations Verify the validity of the last Maxwell relation (Eq. 12–19) for steam at 250°C and 300 kPa. 56 EXAMPLE 12–5 Evaluating the hfg of a Substance from the P-v-T Data Using the Clapeyron equation, estimate the value of the enthalpy of vaporization of refrigerant-134a at 20°C, and compare it with the tabulated value. EXAMPLE 12–6 Extrapolating Tabular Data with the Clapeyron Equation Estimate the saturation pressure of refrigerant-134a at - 50°F, using the data available in the refrigerant tables. EXAMPLE 12–7 Internal Energy Change of a van der Waals Gas Derive a relation for the internal energy change as a gas that obeys the van der Waals equation of state. Assume that in the range of interest Cv varies according to the relationCv =C1 +C2T, where C1 andC2 are constants. EXAMPLE 12–8 Internal Energy as a Function of Temperature Alone Show that the internal energy of (a) an ideal gas and (b) an incompressible substance is a function of temperature only, u=u(T). EXAMPLE 12–9 The Specific Heat Difference of an Ideal Gas Show thatCp-Cv =R for an ideal gas. EXAMPLE 12–10 Joule-Thomson Coefficient of an Ideal Gas Show that the Joule-Thomson coefficient of an ideal gas is zero. EXAMPLE 12–11 The _h and _s of Oxygen at High Pressures Determine the enthalpy change and the entropy change of oxygen per unit mole as it undergoes a change of state from 220 K and 5 MPa to 300 K and 10 MPa (a) by 57 assuming ideal-gas behavior and (b) by accounting for the deviation from ideal-gas behavior. 12–14 Consider an ideal gas at 400 K and 100 kPa. As a result of some disturbance, the conditions of the gas change to 404 K and 96 kPa. Estimate the change in the specific volume of the gas using (a) Eq. 12–3 and (b) the ideal-gas relation at each state. 12–15 Using the equation of state P(v - a) = RT, verify (a) the cyclic relation and (b) the reciprocity relation at constant v. 12–16 Verify the validity of the last Maxwell relation (Eq. 12–19) for refrigerant-134a at 80°C and 1.2 MPa. 12–18E Verify the validity of the last Maxwell relation (Eq. 12–19) for steam at 800°F and 400 psia. 12–37 Derive an expression for the specific-heat difference Cp-Cv for (a) an ideal gas, (b) a van der Waals gas, and (c) an incompressible substance. 58 12–44C The pressure of a fluid always decreases during an adiabatic throttling process. Is this also the case for the temperature? 12–54C What is the enthalpy departure? 12–56C Why is the generalized enthalpy departure chart prepared by using PR and TR as the parameters instead of P and T? 12–57 Determine the enthalpy of nitrogen, in kJ/kg, at 175 K and 8 MPa using (a) data from the ideal-gas nitrogen table and (b) the generalized enthalpy departure chart. Compare your results to the actual value of 125.5 kJ/kg. 59 60
 0
0
advertisement
Related documents
Download
advertisement
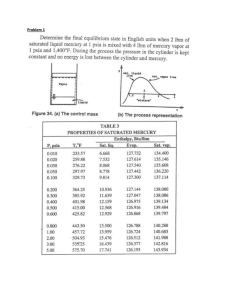
Add this document to collection(s)
You can add this document to your study collection(s)
Sign in Available only to authorized usersAdd this document to saved
You can add this document to your saved list
Sign in Available only to authorized users