humidity flow
advertisement

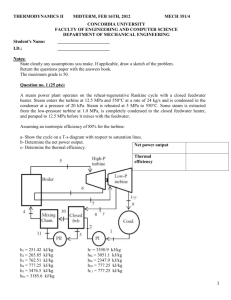
FACULTY OF ENGINEERING AND COMPUTER SCIENCE DEPARTMENT OF MECHANICAL AND INDUSTRIAL ENGINEERING COURSE NUMBER Thermodynamics II EXAMINATION Final SECTION MECH 351 DATE TIME & PLACE April 24, 2012 9:00 – 12:00 PROFESSORS Room: All Sections # OF PAGES 18 LAB INSTRUCTOR I. Hassan and L. Kadem MATERIALS ALLOWED X NO YES (PLEASE SPECIFY) CALCULATORS ALLOWED NO x YES (non programmable) SPECIAL INSTRUCTIONS: Answer the following four questions. State clearly any assumptions you make. Draw a clear sketch of the problem. Return the Exam paper with the answers’ book. Name: _______________________________ I.D.: ______________________ Surname, given names 1 Question no. 1 (30 Points) Consider a cogeneration power plant modified with regeneration. Steam enters the turbine at 6 MPa and 450°C and expands to a pressure of 0.4 MPa. At this pressure, 60 percent of the steam is extracted from the turbine, and the remainder expands to 10 kPa. Part of the extracted steam is used to heat the feedwater in a feedwater heater. The rest of the extracted steam is used for process heating and leaves the process heater as a saturated liquid at 0.4 MPa. It is subsequently mixed with feedwater heater, and the mixture is pumped to the boiler pressure. Assuming the turbine and the pumps to be isentropic, a- Show the cycle on a T-s diagram with respect to saturation lines. b- Determine the mass flow rate of steam through the boiler for a net power output of 15 MW. c- Compute the utilization factor. Question no. 2 (25Marks): A stationary gas-turbine power plant, as shown in figure, operates on an ideal regenerative Brayton q regen,act cycle ( = 75 percent, ) with air as the working fluid. Air enters the compressor (state q regen,max 1) at 100 kPa and 290 K and the turbine (state 3) at 610 kPa and 1100 K. The compressor and turbine isentropic efficiencies are 84 and 100%, respectively. (a) Plot the T-s diagram of the cycle. (b) Determine the thermal efficiency of the cycle. (c) Determine the net work delivered by this plant. Assume constant specific heats with temperature, Cv = 0.718 kJ/kg K and Cp= 1.005 kJ/kg K. 2 Question no. 3 (20 Marks) Atmospheric air enters an air-conditioning system at 30oC and 70 percent relative humidity with a volume flow rate of 4 m3/min and is cooled to 20oC at a pressure of 1 atm, as shown in the figure. The system uses refrigerant-134a as the cooling fluid that enters the cooling section at 350kPa with a quality of 20 percent and leaves as a saturated vapor. a- Sketch the process on the Psychrometric chart. b- What is the heat transfer from the air to the cooling coils, in kW? c- If any water is condensed from the air, how much water will be condensed from the atmospheric air per min? d- Determine the mass flow ate of the refrigerant, in kg/min. 3 Question no. 4 (25 Marks) Benzene gas (C6H6) at 25°C is burned during a steady-flow combustion process with 95 percent theoretical air that enters the combustion chamber at 25°C and 1 atm. All the hydrogen in the fuel burns to H2O, but part of the carbon burns to CO. If the products leave at 1000 K, determine, a- The mole fraction of the CO in the products. b- The heat transfer from the combustion chamber during this process. 4 s THERMODYNAMICS DATA SHEET R = 8.315 kJ/kmole°K 1 bar = 100kPa 1 gm/cm3 = 103 kg/m3 Molar masses and specific heats: Air: M = 29 kg/kmol Cp= 1.005kJ/kg K Enthalpy of Formation: 5 Note: Cp = Cv + R 6 7 8 9 10 11 12 13 14 15 . 16 Formula Sheet – Thermodynamics II T2 V1 T1 V2 K 1 T P ; 2 2 T1 P1 K 1 K MEP= K P V W W C ; 2 1 ; where K= P ; P s ;T a ; Wa Ws CV P1 V2 W Vmax Vmin ; cycle Wnet Qin Ni m ; Average molar mass M m = m = yi M i ; gas constant: Nm Nm P V N R Rm u Ru 8.314 KJ /( Kmol.K ) ; PV ZNRuT ; (Zm yi Zi ) ; i i i yi ; Mm Pm Vm Nm Mole number: Ni mi ; Mole fraction: yi Mi Dalton’s law: Pm Pi (Tm .Vm ) ; Amagat’s law: Vm Vi (Tm .Pm ) ; U m = mfiU i , U m = yi U i (same for h and s); Cv,m mfiCv,i ; C v,m yi C v,i , same for Cpm Real gasses: h (h2 h1 ) ideal Ru Tcr ( Z h 2 Z h1 ) ; u (h2 h1 ) Ru (Z 2T2 Z1T1 ) ; s (s2 s1 )ideal R(Zs2 Zs1 ) ; . Air conditioning energy balance: Q Air fuel ratio: AF . in . . . . . ; m=NM ; m fuel . out . m air . W in mi hi Q W out me h e , m m a Pvi Ptotal mair,act N Ni ; Percentage of theoretical air: or air ,act Ntotal mair,th N air,th Enthalpy of combustion : hc=H prod H react ; HHV LHV (mhfg )H2O ; First law analysis of reacting systems: Q W N p (h f h h) p Nr (h f h h)r Entropy generation of reacting systems: S gen N p s p N r s r yP N p s o i T , Po Ru ln i m Po yP N r s o i T , Po Ru ln i m Po Pr od 17 react Summary of Final Answers Question No 1: Required Numerical Final Answer (1) Mass flow rate (2) Utilization factor Question No 2: Required Thermal efficiency Work in kW Numerical Final Answer Question No 3: Required Numerical Final Answer Heat Transfer Condensed water flow rate Refrigerant mass flow rate Question No 4: Required Numerical Final Answer (1) The mole fraction of CO (2) The heat transfer rate from the combustion chamber. 18