Answer
advertisement

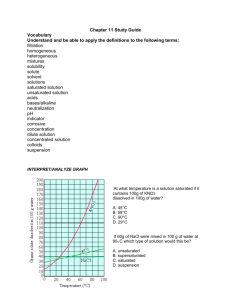
Mrs. Lee – 8th grade Physical Science Page 1 Ch 7 Practice Test – Answer Key ______ c 1) A solution is an example of a ______. a) homogeneous colloid b) heterogeneous colloid c) homogeneous mixture d) heterogeneous mixture ______ b 2) Magnesium sulfide and aluminum fluoride are ______. a) ionic compounds b) molecular compounds c) covalent electrons d) radioactive elements ______ a 3) When dissolved in water, ionic compounds ______. a) conduct electricity b) make the water cloudy c) forms double and triple bonds d) do not conduct electricity ______ d 4) When dissolved in water, molecular compounds ______. a) conduct electricity b) make the water cloudy c) form double and triple bonds d) do not conduct electricity ______ a 5) A mixture in which particles can be seen and easily separated by settling or filtration is a ______. a) suspension b) solution c) solute d) solvent ______ b 6) A neutral solution has a pH of ______. a) 3 b) 7 c) 5 d) 9 ______ c 7) A base ______. a) tastes sour c) does not react with carbonates b) corrodes metals d) turns litmus red ______ c 8) Solutes lower the freezing point of water by ______. a) stopping water molecules from moving b) forming crystals c) making it harder for water to form crystal d) making the water molecules move faster ______ b 9) A polar solvent will most likely dissolve ______. a) any solute b) polar solutes c) nonpolar solutes d) no solute ______ c 10) If you add a small amount of hydrochloric acid to 4 liters of water, what type of solution would you expect to have? a) concentrated solution b) basic solution c) dilute solution d) saturated solution ______ d 11) The pH scale ______. a) is numbered from 1 to 12 b) measures the concentration of bases c) measures the concentration of acids d) measures the concentration of hydrogen ions Ch 7 Practice Test – Answer Key 1 Mrs. Lee – 8th grade Physical Science Page 2 Ch 7 Practice Test – Answer Key ______ a 12) Neutralization ______. a) is a reaction between an acid and a base b) occurs when acid is dissolved in water c) forms an acid and a base d) does not change the pH of a solution ______ c 13) Ammonia (NH3) reacts with water to form hydroxide ions. The resulting solution ______. a) turns blue litmus paper red b) causes red litmus paper to remain red c) turns red litmus paper blue ______ b (concentrated) 14) If you add solute to a dilute solution, the solution becomes a more dilute solution. a) True b) False ______ a 15) Acids are described as being corrosive, because they dissolve some metals. a) True b) False ______ b (indicator) 16) A suspension is a substance that turns different colors in an acid or a base. a) True b) False ______ b (colloid) 17) A salt is a mixture containing small, undissolved particles that do not settle out. a) True b) False ______ a 18) When a base reacts with an acid, water and a salt form. a) True b) False ______ a 19) When acids react with carbonate compounds, carbon dioxide forms. a) True b) False ______ b (acid) 20) A base forms hydrogen ions when it dissolves in water. a) True b) False ______ b (solvent) 21) The solute is the part of a solution present in the largest amount. a) True b) False ______ b (increase) 22) Solutes decrease the boiling point of a solvent. a) True b) False ______ c 23) A mixture in which particles can be seen and easily separated is a ______. a) solution b) solvent c) suspension Ch 7 Practice Test – Answer Key 2 Mrs. Lee – 8th grade Physical Science Page 3 Ch 7 Practice Test – Answer Key ______ c 24) Solutes lower the freezing point of water by ______. a) making the water molecules move faster b) forming salts c) making it harder for water molecules to form crystals ______ a 25) Which is a dilute solution? a) 1 gram of salt dissolved in 100 grams of water b) 100 grams of salt dissolved in 100 grams of water c) 150 grams of salt dissolved in 100 grams of water ______ a 26) At higher pressure ______. a) more gas can dissolve b) less gas can dissolve c) gases cannot dissolve at all ______ c 27) An acid ______. a) tastes bitter b) turns red litmus paper blue c) dissolves some metals ______ b 28) Both sodium hydroxide (NaOH) and potassium hydroxide (KOH) produce hydroxide ions (OH-) in water. These compounds are ______. a) acids b) bases c) salts ______ c 29) The pH scale ______. a) is numbered from 1 to 12 b) measures the concentration of salts c) measures the concentration of hydrogen ions (H+) ______ b 30) Neutralization ______. a) forms an acid and a base b) is a reaction between an acid and a base c) occurs when an acid dissolves in water ______ b 31) Which factor is least likely to affect solubility? a) pressure b) volume c) temperature ______ c 32) A solid that produces H+ ions when dissolved in water will create a solution that has ______. a) a neutral pH b) a pH higher than neutral c) a pH lower than neutral ______ a 33) A mixture containing small, undissolved particles that do not settle out is a colloid. a) True b) False ______ a 34) If you add solute to a dilute solution, the solution becomes a more concentrated solution. a) True b) False Ch 7 Practice Test – Answer Key 3 Mrs. Lee – 8th grade Physical Science Page 4 Ch 7 Practice Test – Answer Key ______ a 35) A solution that contains more solute than predicted at a given temperature is supersaturated. a) True b) False ______ b 36) The solute is the largest part of a solution. a) True b) False ______ a 37) Solubility can be used to identify an unknown substance. a) True b) False ______ a 38) A base tastes bitter, feels slippery, and turns red litmus paper blue. a) True b) False ______ b 39) A base forms hydrogen ions when it dissolves in water. a) True b) False ______ b 40) A cook would never use an acid when baking a cake. a) True b) False Use the picture below to answer the following 2 questions: ______ b 41) Inferring: The jar in the diagram has been shaken up, then allowed to sit for a day. What kind of solution is this? a) dilute solution b) saturated solution c) unsaturated solution ______ c 42) Applying Concepts: To change this solution to a supersaturated solution, you would have to ______. a) add more water b) add more sugar c) heat the solution Use the equation below to answer the following 3 questions: NH3 + H2O NH4+ + OH- Ch 7 Practice Test – Answer Key 4 Mrs. Lee – 8th grade Physical Science Page 5 Ch 7 Practice Test – Answer Key ______ b 43) Interpreting Diagrams: This equation shows what happens when ammonia (NH3) dissolves in water (H2O). From this, you can tell that ammonia is a(n) ______. a) acid b) base c) salt ______ a 44) Applying Concepts: What would be the pH of the solution formed when ammonia (NH3) dissolves in water? a) greater than 7 b) equal to 7 c) less than 7 ______ a 45) Applying Concepts: The solution formed when ammonia (NH3) dissolves in water can conduct electricity because _____. a) it contains ions b) it contains molecules c) it contains table salt Ch 7 Practice Test – Answer Key 5