Chemical Bonding Lab
advertisement

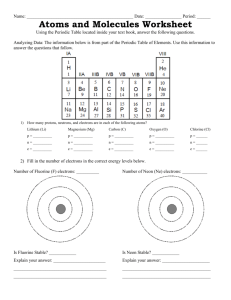
Chemical Bonding Lab Each ball in your kit represents an atom. Each stick represents a single bond. The spring represents a double bond. This assignment will help you decide how atoms bond together. Transferring vs. Sharing Electrons: An atom transfers electrons when one atom wants to gain electrons while another atom wants to lose electrons. Sharing electrons occurs when both atoms need to gain electrons, so they each share one pair of electrons (single bond) or two pairs of electrons (double bond). Single vs. Double Bonds: A single bond forms when an atom transfers an electron to another atom, or when two atoms share ONE pair of electrons. A double bond forms when atoms share TWO pairs of electrons. Part 1: WRITE ON YOUR OWN PAPER! 1. On your paper, draw out an oxygen atom. Include the correct number of valence electrons. 2. How many valence electrons are missing from an oxygen atom for it to have a full outer energy level? 3. Draw out another oxygen atom next to your first oxygen atom (from #1). You see that both oxygen atoms need valence electrons to complete their outer energy level. 4. Since both atoms want to gain electrons, would atoms be transferred or shared between the atoms? 5. Would a single or double bond be formed between the oxygen atoms? 6. With your molecule kit, oxygen atoms are represented by yellow balls. Create a model of an oxygen molecule (O2): (Hint: Look up top to see how to represent single vs. double bonds…which one should you choose?) Come show me your oxygen molecule when you have made it, and have me initial your papers. 7. Draw the ball and stick model (sketch your model you just created!) on your notebook paper. Part 2: WRITE ON YOUR OWN PAPER! 1. 2. 3. 4. 5. 6. 7. On your paper, draw out a lithium atom. Include the correct number of valence electrons. How many valence electrons does lithium have? Do you think lithium wants to gain or lose electrons? Why? On your paper, draw out a fluorine atom. Include the correct number of valence electrons. How many valence electrons does fluorine have? Do you think fluorine wants to gain or lose electrons? Why? Based on your above observations, do you think lithium and fluorine would have a transfer of electrons or a sharing of electrons between them? Why? 8. Would a single or a double bond be formed? 9. In your molecule kits, lithium is represented by red balls, and fluorine is represented by green balls. Create a model of a lithium fluoride molecule (LiF), then come show it to me. 10. Draw the ball and stick model of lithium fluoride on your notebook paper! Part 3: WRITE ON YOUR OWN PAPER! 1. 2. 3. 4. On your paper, draw two hydrogen atoms side by side. How many valence electrons does each hydrogen atom have? Does a hydrogen atom want to gain or lose electrons? Based on your above observations, do you think two hydrogen atoms would have a transfer of electrons or a sharing of electrons between them? Why? 5. Would a single or a double bond be formed? 6. In your molecule kits, hydrogen is represented by orange balls. Create a model of a hydrogen molecule (H2), and then come show it to me. 7. Draw the ball and stick model of a hydrogen molecule on your notebook paper! ***Clean up: Make sure you put all balls, sticks, and springs back into their black boxes. Bring the black boxes up to the front, then turn in your papers into the basket.