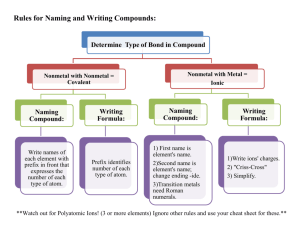
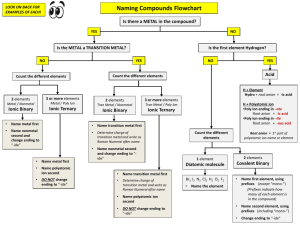
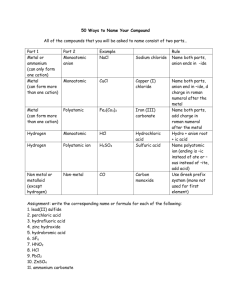
Rules for Naming Ionic Compounds
I. Four different types of ionic compounds
A. Ionic compounds metal and nonmetal
1. Metal of Family IA, IIA, IIIA, Zn, Cd, Ag and a nonmetal
a. Name the metal and then name the nonmetal changing the ending to the -ide suffix.
b. Zinc, Cadmium and Silver always have the same charges so they follow the same rule as
above. (Zn2+, Cd2+, Ag+)
c. Examples:
NaCl
K2S
Mg3N2
ZnO
=
=
=
=
sodium chloride
potassium sulfide
magnesium nitride
zinc oxide
2. Metal of Families IVA, VA and Transition metals and a nonmetal
a. These metals can have more than one charge. A roman numeral is added to the name to
indicate the positive charge of the metal. The nonmetal is named changing the ending
to the -ide suffix.
b. Examples:
FeO
= iron (II) oxide
Fe2O3 = iron (III) oxide
CuCl2 = copper (II) chloride
Nonmetal Root names
bor-
carb-
nitr-
ox-
silic-
phospharsen-
sulfselentellur-
hydrfluorchlorbromiodastat-
B. Ionic compounds metal and polyatomic ion.
Polyatomic ion - tightly bound group of atoms that behaves as a unit and carries a charge that is
usually negative.
3. Metal of Family IA, IIA, IIIA, Zn, Cd, Ag and a polyatomic ion
a. Name the metal and then name the polyatomic ion
b. Examples:
Al2(CO3)3
Na2SO4
AgClO3
(NH4)2SO4
=
=
=
=
aluminum carbonate
sodium sulfate
silver chlorate
ammonium sulphate
NH4+
Ammonium
Ion is the
exception
4. Metal of Families IVA, VA and Transition metals and a nonmetal
a. Name the metal, use a Roman numeral to indicate the positive charge of the metal and
name the polyatomic ion.
b. Examples:
Cu(NO3)2 = copper (II) nitrate
MnSO3
= manganese (II) sulfite
SnCrO4 = tin (II) dichromate
Rules for Naming Covalent Compounds
Naming Compounds Containing Two Nonmetals: when two nonmetals are combined in a compound,
it is important that the name indicate the number of atoms of each element that are present
because more than one compound can exist containing the same two nonmetals. For example,
nitrogen and oxygen form a number of different compounds with chemical formulas such as:
NO, NO2, N2O4, etc. It is essential that the names of these compounds be different. A system is
used in which Greek number prefixes are employed to indicate how many atom of each element are
present. You will need to learn the following number prefixes if you do not already know them.
Greek Prefixes:
1
(mono)
2
di-
3
tri-
4
5
tetra- penta-
6
hexa-
7
hepta-
8
octa-
9
nona-
10
deca-
If there is only one atom of the first nonmetal, the prefix, mono, is not used, but in
all other cases, the number prefix is attached to the name of the nonmetal to indicate how many
atoms of each element are present. Again, because the compound contains only two elements,
the root of the name of the second element is given an -ide ending.
Writing Covalent Formulas
carbon dioxide
(mono) carbon di - ox -ide
C1 O2 CO2
(note: mono is implied for the first element, it is not called mono- )
carbon monoxide (mono) carbon
dinitrogen pentoxide
mono - ox -ide C1 O1
di - nitrogen penta - ox -ide N2 O5
CO
N2O5
Naming Covalent Compounds
Greek prefix (Nonmetal name) + Greek prefix (Nonmetal root name) -ide
CO2
1-carbon 2-ox(ygen)-ide (mono)carbon di - ox - ide
carbon dioxide
(note: mono is implied for the first element, it is not called mono- )
CO
1-carbon 1-ox(ygen)-ide (mono)carbon mono -ox -ide carbon
N2O5
2-nitrogen 5-ox(ygen)-ide di -nitrogen penta -ox -ide
Other Examples:
SO3 = sulfur trioxide
P4O10 = tetraphosphorus decaoxide
H2O = dihydrogen monoxide
monoxide
dinitrogen pentaoxide
Rules for Naming Acids
Hydrogen and another nonmetal - all acids contain hydrogen as the first element and are given acid names
when in aqueous solution which is denoted by (aq) following the formula.
1. If the formula is followed by (g) denoting a gaseous state, the compound is named in the same way
as Rule A-1 (See ionic naming rules page 1)
Examples: HCl (g)
= hydrogen chloride
H2Te (g) = hydrogen telluride
Note: No Greek Prefixes are used when H is the cation
2. If the formula is followed by (aq) denoting an acid solution, add a hydro- prefix to the root of the
second nonmetal name and add an -ic suffix then the word acid
H2S (aq) = hydrosulfuric acid
Examples:
HCl (aq) = hydrochloric acid
NOTE: The acid with only two different atoms has a longer name.
Hydrogen and a polyatomic ion - all acids contain hydrogen as the first element and are given acid names
.
1.
Polyatomic ions ending in -ite
a. Add an -ous suffix to the polyatomic ion name and add the word acid.
b. Examples:
HNO2 = (hydrogen nitrite) = nitrous acid
H2SO3 = (hydrogen sulfite) = sulfurous acid
HClO = (hydrogen hypochlorite) = hypochlorous acid
c. Mnemonic – Last n-ite, I saw a m-ous-e.
2. Polyatomic ions ending in -ate
a. Add an -ic suffix to the polyatomic ion name and add the word acid.
b. Examples:
H2CO3 = (hydrogen carbonate) = carbonic acid
H3PO4 = (hydrogen phosphate) = phosphoric acid
HBrO4 = (hydrogen perbromate) = perbromic acid
c. Mnemonic – I -ate a bug -ic-k
NOTE: The acid with three or more different atoms has a shorter name.
 0
0