Supplementary Information (doc 136K)
advertisement

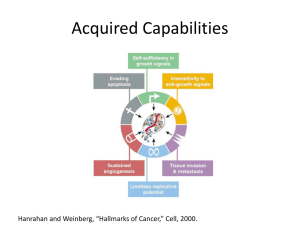
Supplemental Methods Immunohistochemistry Colon carcinoma and normal colon tissue sections (5 μm) were stained with a combination of mouse anti–human CD31 (1:100, DAKO) and mouse-anti-human CD34 (1:100, Monosan) or with mouse anti-human HMGB1 (1:50, Sigma) essentially as described previously (van Beijnum et al., 2006) Sections of the CAMs were excised, paraformaldehyde-fixed and paraffin embedded. CAM tumors were excised and stored in Zn-fixative prior to paraffin embedding. Tissue sections (5μm) were pretreated with xylene and rehydrated in a series of ethanol. Endogenous peroxidase activity was blocked using H2O2, and sections were boiled in citrate buffer (10mM, pH 6.0) for antigen retrieval. CAM sections were incubated with mouse anti-human HMGB1 (1:50, Sigma) and primary antibodies were detected with biotinylated rabbit anti-mouse (1:100, DAKO) in combination with streptavidin-HRP (1:100, DAKO). Antibody binding was detected using DAB. CAM tumors were stained with mouse anti-human α-smooth muscle actin (1:200, DAKO), in combination with HRPconjugated rabbit anti-mouse Envision staining system (DAKO). Flow cytometry Flow cytometry for the detection of HMGB1 in isolated EC was performed by double labeling of EC in single-cell suspensions form human tumor and normal colon samples (van Beijnum et al., 2008) using a combination of PE-labeled mouse anti-human CD31 (1:25, DAKO) and rabbit anti-human HMGB1 (1:50, kind gift of Dr R.G. Roeder, New York, NY). Cultured EC were trypsinized and stained with mouse anti-human ICAM1 (1:100, Monosan), Cells were fixated with 1% PFA and permeabilized (for HMGB1 detection) with 0.1% Triton X-100 to allow intracellular staining. FITC-labeled secondary antibodies were used for fluorescent detection. All antibody incubations were performed in PBS/0.1% BSA. Cells were analyzed on a FACSCalibur (BD Biosciences) and data were analyzed using CellQuest software (BD Biosciences). In silico analysis of HMGB1 expression in (colorectal) cancers The Oncomine database contains a repository of microarray data covering a broad panel of human cancers (www.oncomine.org). HMGB1 expression values were retrieved from 24 studies that matched the following criteria: Analysis type, Cancer vs. Normal Analysis and Multicancer Analysis; Cancer type, Colorectal cancer; Data type, mRNA. This enabled visualization of HMGB1 mRNA levels in colorectal cancer and normal tissues. Using the In silico Transcriptomics (IST) tool, available from www.genesapiens.org, correlations between HMGB1 and VEGFA based on expression data of >300 gene expression studies concerning cancer were retrieved and plotted. The Human Protein Atlas (http://www.proteinatlas.org) was mined for examples of colorectal and ovarian tumors, as well as normal colon and normal ovary with different intensities of HMGB1 expression. For comparison, sections showing vascular staining for von Willebrand Factor (vWF) are also shown. Proliferation assay Proliferation was measured using the 3H-thymidine incorporation assay. Cells were plated in gelatin-coated 96-well plates at 5000 (HUVEC) or 15000 (RF24) cells per well and allowed to adhere for 4 hours. Where applicable, compounds were added and cells were grown for 3 days. Cultures were pulsed with 0.3µCi of 3H-thymidine for 6 hours. Subsequently, cells were lysed by freeze-thawing and lysates were transferred to nitrocellulose filters. Filters were submerged in scintillation fluid and radioactivity was measured using a liquid scintillation counter (Wallac LSC) (van Beijnum et al., 2006). Sprouting assay Bovine capillary endothelial cells (BCE) were seeded on cytodex-3 gelatin-coated microcarrier beads (500 cells/bead) in suspension. After 24 hours, beads (25/well) were embedded in collagen gel (2.4 mg/ml) in 96-well flat-bottom plates. BCE were allowed to sprout into the collagen gel for 24 hrs after which images were taken. Sprouting was quantified by counting intersections of sprouts with a concentric ring grid overlay on the image (van Beijnum et al., 2006). Collagen gels and overlay medium were supplemented with compounds as indicated. From freshly excised human colorectal tumor tissue, a blood vessel was prepared free, cut in 1mm sections and embedded in collagen gel as described above, in the presence or absence of mouse anti-human HMGB1 antibodies (10 μg/ml), and allowed to sprout for 5 days. Sprout length was quantified in ImageJ. Apoptosis assay HUVEC were plated in gelatin-coated 96-well plates at 5000 cells per well and allowed to adhere for 4 hours. Where applicable, compounds were added and cells were grown for 3 days. Cells were harvested by trypsinization and incubated with propidium iodide (PI) (20 µM) in buffer containing 2.5 µM citric acid, 45 µM Na2HPO4 and 0.1% Triton-X100, pH7.4 for 20 minutes at 37˚C. Cells were analyzed with a FACSCalibur (BD Biosciences) in the FL2 channel. Apoptotic cells were defined as those that showed subG1 DNA staining. Western blot analysis Cells were cultured in serum-free medium for 16 hours prior to stimulation with rHMGB1. Nuclear and cytoplasmic extracts were prepared using NE-PER reagent (Pierce) according to the manufacturers’ instructions. Protein (25 µg) was loaded on a standard SDS-PAGE gel and blotted to nitrocellulose. Blots were incubated with antiHMGB1 antibody (1:1000; Sigma) or anti-actin antibody (1:50000; MP Biomedicals) followed by biotinylated rabbit-anti-mouse antibody (1:1000; DAKO) in combination with streptavidin-HRP (1:1000; DAKO) and DAB. Blots were scanned and band intensities were quantified using QuantityOne software (Bio-Rad). HMGB1 ELISA RF24 cells were seeded at 15000 cells/well as described above. Cells were grown for 48 hours, after which cells were treated with rHMGB1 as indicated in serum free medium for 16 hours. Cells were washed and fresh serum free medium was added. Media were collected after 8 hours and measured using a commercially available ELISA kit for the detection of HMGB1 in serum or culture medium (Shino-Test). Wells without cells were taken along as a control. NF-B, MMP2 and ITGB1 activity measurement The proinflammatory adhesion molecules ICAM1, VCAM1 and E-selectin (SELE), are under control of NF-B. Active p65 NF-B subunit was detected using the EZ-detect NF-B p65 ELISA assay kit (Pierce), according to the manufacturer’s instructions. The activity of MMP1 and MMP2 was measured to assess whether the increased mRNA expression also contributed to more active MMPs that can facilitate matrix invasion. In fact, HMGB1 itself may mediate MMP activation by promoting plasmin production on the cell surface through interaction with urokinase-type or tissue-type plasminogen activator. MMP2 activity was determined with an immunocapture assay (QuickZyme Biosciences). EC were trypsinized and stained with an antibody against activated ITGB1(Chavakis et al., 2007) (1:50, BD Biosciences), and analysed by flow cytometry as described above. siRNA transfection RF24 (1*104) were seeded in 96-well tissue culture plates that were coated with gelatin and where siRNA (Qiagen; 20 nM) and transfection reagent (HiPerfect; Qiagen, 0.75 µl) were complexed for 20 minutes. Cells were processed for downstream analysis 48-72 hr later. Alternatively, EC were grown for 3 days after transfection with oligodeoxynucleotides (ODN) (Biognostik; 5µM). Transfection and cloning of HMGB1 expression constructs HMGB1 open reading frame was amplified from cDNA (Fw primer: 5’GCGCACTGGGCGACTCTGTG-3’, Rev primer: 5’-ACTGCGCTAGAACCAACTTATTC-3’) and cloned into pcDNA3.1+-Hygro (pHMGB1) using HindIII and XhoI. RF24 were transfected using the Amaxa nucleofector system as described (Thijssen et al., 2010). Cells were processed for analysis three days after transfection. Supplemental Table 1: qPCR primer sequences Gene Forward Reverse ACTB CATTCCAAATATGAGATGCATT CCTGTGTGGACTTGGGAGAG ANG1 AGCTACCACCAACAACAGTG GCAAAGATTGACAAGGTTGTGG ANG2 TGCCACGGTGAATAATTCAG TTCTTCTTTAGCAACAGTGGG ANG3 AACAGCGCGCTCGAGAAG GCTTCGCCTTCTTGCTGA B2M TCCATCCGACATTGAAGTTG CGGCAGGCATACTCATCTT bFGF AGGAGTGTGTGCTAACCGTTAC ACTCATCCGTAACACATTTAGAA EGFR TGCCCATGAGAAATTTACAGG ATGTTGCTGAGAAAGTCACTGC FGFR1 ATCACGGCTCTCCTCCAGTG AAGACCAGTCTGTCCCGAGG FGFR3 ATGATCATGCGGGAGTGCTG AGTACTGCTCGAAAGGCGCC HMGB1 TTCATTTCTCTTTCATAACGGG TCTAAGAAGTGCTCAGAGAGGTG ICAM1 GGCCGGCCAGCTTATACAC TAGACACTTGAGCTCGGGCA ITGA2 GGAACGGGACTTTCGCAT GGTACTTCGGCTTTCTCATCA ITGB1 GAAGGGTTGCCCTCCAGA GCTTGAGCTTCTCTGCTGTT MMP1 CCAAATGGGCTTGAAGCT GTAGCACATTCTGTCCCTAA MMP2 AGATGCCTGGAATGCCAT GGTTCTCCAGCTTCAGGTAAT MMP9 TACTGTGCCTTTGAGTCCG TTGTCGGCGATAAGGAAG NRP1 CCCGAGAGAGCCACTCATG GTCATCACATTCATCCACCAA NRP2 CCGGTCGTTTGGGCTGGA AACTGCAACTTTGATTTTCCG PDGFRA TTCATTGAAATCAAACCCACC TTTCCTGAATCTTTTCCACATC PDGFRB AGAGCTGCCCATGAACGAG CGAATCTCAAACTTCTCTTCCC PlGF ACGTCTGAGAAGATGCCGGTC ACCACTTCCACCTCTGACGA PPIA CTCGAATAAGTTTGACTTGTGTTT CTAGGCATGGGAGGGAACA RAGE AACTCTAGCCCTGGCCCTGG TTCTGGGGCCTTCCTCTCCT SELE CCCGAAGGGTTTGGTGAG CAAATCCCTCCTCACAGCTG Tie1 CCCCGCTGGTCTCGTTCTC CACAATGGTCGACCAGTCC Tie2 TTGAAGTGGAGAGAAGGTCTG GTTGACTCTAGCTCGGACCAC TLR2 ATTCCCCAGCGCTTCTGCA AATCCTTCCCGCTGAGCCTC TLR4 TTGAAAGTGTGTGTGTCCGC CAAGCATAAGGGATAAGGGG VCAM1 TCAGATTGGAGACTCAGTCATGT ACTCCTCACCTTCCCGCTC VEGFA AAGGAGGAGGGCAGAATCAT CAGAAGGAGAGCAGAAGTCC VEGFR1 CCAGCAGCGAAAGCTTTGCG CTCCTTGTAGAAACCGTCAG VEGFR2 ATGACATTTTGATCATGGAGC CCCAGATGCCGTGCATGAG Supplemental Table 2: Species-specific qPCR primer sequences Gene Forward Reverse ACTB CATTCCAAATATGAGATGCATT CCTGTGTGGACTTGGGAGAG B2M TGTAGACGGCTTCGCTGC AGGAGTGTGTGCTAACCGTTAC bFGF AGGAGTGTGTGCTAACCGTTAC ACTCATCCGTAACACATTTAGAA CD31 ACTCCAGTGGCATGAAAACT ACAGAGCAGAGAAAAGTGGTC EGFR AGAGCTGCCAATGAAACG-G AAGTACTGTGAGAGGCTTCCT HMGB1 AAGAATATTCTTCCGTAACTGG TTCTCAACTCTCCAAGACTGG PDGFRA GTTCATTCATCTAGAGCCTCAAT TTTCCTGGACTCTGTTTGAACT PDGFRB CATGGCGGTGAACAGCAA TCTGTGTGGGCTCCAGGCT PPIA AAGGAGGGGATGAACGTG AGCTGCCCGCAGTTGGA VCAM TGATGTGGTCACAACCCTTAA TCCAAATTTGTTAGTGAATGTGC VEGFA GACCTGTAAATGTTCCTGCAA AGAAATCAGGCTCCAGAAACA VEGFR1 TCGACACTATCTTCACAGCGG GCTTCTGCAGTTTGGGCT VEGFR2 TCACGCCTTACAGACACCCT AGGGAGATGTTACGGAGAATG ACTB TTCCTATGTGGGCGACGAG TCCTCGGGAGCCACACG B2M TCCATCCGACATTGAAGTTG ACACGGCAGGCATACTCAT bFGF CGTAAGTGCAAACCGCTTT CGTAAGTGCAAACCGCTTT CD31 TTCCCACGCCAAAATGTTA CACAGCACATTGCAGCACA EGFR TGCCCATGAGAAATTTACAGG ATGTTGCTGAGAAAGTCACTGC HMGB1 ACATCCAAAATCTTGATCAGTTA AGGACAGACTTTCAAAATGTTT PDGFRA TTCATTGAAATCAAACCCACC TTTCCTGAATCTTTTCCACATC PDGFRB AGAGCTGCCCATGAACGAG CGAATCTCAAACTTCTCTTCCC PPIA AGCATGTGGTGTTTGGCAAA TCGAGTTGTCCACAGTCAGC VCAM TAACGGGGAGCTACAGCC CAGCCTGGTTAATTCCTTCAC VEGFA CCATCGACAGAACAGTCCTT CGAATCCAATTCCAAGAGGG VEGFR1 CAATGCCATACTGACAGGAA CAGAGCTTCCTGAATTAAACTT VEGFR2 ACAGCCTCTGCCAATCCATG AAGGATGCATTCTTAAGCTCC Chicken Human Supplemental Figure legends Supplementary Figure 1: HMGB1 expression in human tumors Proteinatlas.org was mined for examples of human colorectal tumors and normal colon with different levels of overall HMGB1 expression. Endothelial expression of HMGB1 is highlighted in the boxed sections using arrows. For comparison, endothelial von Willebrand factor (vWF) staining is also shown. Endothelial expression of HMGB1 is also evident in human ovarian cancer and normal ovary tissue samples. Supplementary Figure 2: Expression of and reactivity to HMGB1 in different endothelial cells (a) RF24 and HUVEC were treated with bFGF (20 ng/ml) or rHMGB1 ( 50 an 500 ng/ml) derived from different sources (see materials and methods), and was assessed using a wound healing assay. (b) qPCR was used to determine expression of HMGB1 in primary (HUVEC) and immortalized (RF24) endothelial cells. In addition, HMGB1 expression levels were determined in A2780 human ovarian carcinoma and LS174T human colorectal carcinoma cell lines. Supplementary Figure 3: HMGB1 expression correlates with VEGFA expression in clinical samples The In silico Transcriptomics (IST) data repository was queried for expression of HMGB1 in gastrointestinal tumor types. Correlation of HMGB1 with VEGFA was significant and positive for CRC and pancreas tumors (left), as well as in renal cancer (right). Supplementary Figure 4: rHMGB1 activates primary endothelial cells HUVEC were treated for 3 days with bFGF (20 ng/ml) or rHMGB1 (500 ng/ml; Sigma). (a) The amount of active MMP2 was measured in HUVEC using ELISA in the conditioned medium of the cells (left). The amount of activated form of ITGB1 was determined by flow cytometry, and normalized for total ITGB1 expression (right). (b) HUVEC were treated as described above, and expression of genes was profiled by qPCR. (c) HUVEC were treated as described above and NF-B subunit p65 activity was determined by target sequence binding ELISA. (d) NF-B target genes ICAM, VCAM and E-selectin (SELE) were determined by qPCR. (e) Relative cytoplasmic HMGB1 content in HUVEC treated with bFGF or rHMGB1 was determined as described. *P<0.05; **P<0.01 Mann-Whitney U-test. Supplementary Figure 5: rHMGB1 stimulates angiogenesis in ovo CAMs were treated with bFGF (20 ng/ml) or rHMGB1 (500 ng/ml; Sigma) or control saline solution from EDD10-14. CAMs were treated daily and 100 μl solution was applied topically. CAMs were photographed after injection of a suspension of vegetable oil with zincoxide powder to provide contrast. An overlay grid was applied on the photos (5x magnification) and intersections of vessels with the grid were counted as a measure of vessel density. *P<0.05; **P<0.01 Mann-Whitney U-test. Supplementary Figure 6: HMGB1 receptor expression in endothelial cells (a, b) RAGE and TLR4 expression were profiled by qPCR in HUVEC and RF24 (a) and in endothelial cells isolated from colon tumors (TEC) and normal colon (NEC) (b). Supplementary Figure 7: Overexpression of HMGB1 stimulates EC migration (a) Immortalized HUVEC (EVLC2) were transfected with pcDNA3-HMGB1 (pHMGB1) and expression levels were measured. (b) No effect of increased HMGB1 expression was seen on proliferation of EC (middle), though migration was markedly induced (right). Supplementary Figure 8: HMGB1 antibodies inhibit angiogenesis in vitro and in vivo (a, b) HUVEC were treated for 3 days with the indicated concentration of anti-HMGB1 Ab. Proliferation was determined by 3H-labeled thymidine incorporation (a), apoptotic cells were detected by PI DNA staining and flow cytometry (b). (c) Color representation of Figure 5e. Fragments of tumor blood vessel were embedded in collagen gel and cells were allowed to sprout for 5 days in the presence or absence of anti-HMGB1 antibodies. (d) Cross sections of paraffin embedded CAMs were stained with anti-HMGB1 antibodies to determine reactivity with chick HMGB1. Clear staining of vascular structures is seen. Scale bar = 100 μm. (e) CAMs were treated with anti-HMGB1 antibodies for 4 days (EDD8-12), after which images were taken after injection of contrast as described in Figure S5, and vessel density adjacent to the tumor was quantified using the overlay grid as described. HET-CAM software was used to determine capillary branching. Clear avascular areas are seen after anti-HMGB1 treatment. Scale bar = 500 μm. (f) LS147T cell spheroids were grown in non-adhesive culture plates and placed on top of the chick chorioallantoic membrane (CAM). qPCR was used to determine expression of HMGB1 in the endothelium of control CAMs and LS174T containing CAMs (left panel). CAMs were treated with anti-HMGB1 antibodies or control antibodies applied topically for 4 consecutive days. CAMs were photographed and analyzed as described. Scale bar = 1 mm. Supplementary Figure 9: Species-specific primer validation (a) Alignment of human (hs) and chicken (gg) HMGB1 sequences. Asterisks indicate identical nucleotides. In bold are the primer sequences used for qPCR. (b, c) qPCR was performed with both primer pairs on both human cDNA and chicken cDNA. No amplification was observed when chicken primers were used in combination with human cDNA and vice versa, whereas specific products were formed with species-matching primers. (e) Primer sensitivity is comparable for both species HMGB1 primer sets. References Chavakis E, Hain A, Vinci M, Carmona G, Bianchi ME, Vajkoczy P, Zeiher AM, Chavakis T and Dimmeler S. (2007). Circ Res, 100, 204-12. Thijssen VL, Barkan B, Shoji H, Aries IM, Mathieu V, Deltour L, Hackeng TM, Kiss R, Kloog Y, Poirier F and Griffioen AW. (2010). Cancer Res, 70, 6216-24. van Beijnum JR, Dings RP, van der Linden E, Zwaans BM, Ramaekers FC, Mayo KH and Griffioen AW. (2006). Blood, 108, 2339-48. van Beijnum JR, Rousch M, Castermans K, van der Linden E and Griffioen AW. (2008). Nat Protoc, 3, 1085-91.