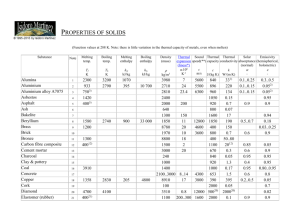
Chapter 3 Problem
advertisement

Chapter 19 Problem 1. Determine the room temperature heat capacity (in J/molK) at constant pressure for copper. The atomic weight for Cu is 63.54 g/mol and the value of its CP is 386 J/kgK. 2. The constant A in the equation Cv = AT3 is equal to 124R/5D3, where R is the gas constant and D is the Debye temperature (in K). Estimate D for some hypothetical metal, given that its specific heat is 0.78 J/molK at 11 K. Assume that the atomic weight for this metal is 63.55 g/mol. (R: 8.31 J/molK) 3. A 0.11 m rod of a metal elongates 0.2 mm on heating from 15 to 100C. Determine the value of the linear coefficient of thermal expansion for this material. 4. To what temperature (in C) must a cylindrical rod of one metal 10.001 mm in diameter and a plate of second metal having a circular hole 9.988 mm in diameter have to be heated for the rod just fit into the hole? Assume that the initial temperature is 29C and that the linear expansion coefficient values for metals one and two are 5.310-6 (C)-1 and 1710-6 (C)-1, respectively. 5. (a) Would you expect Equation 19.7 ( L k / T ) be valid for ceramic and polymeric materials? (b) Estimate the value for the Wiedermann-Franz constant L (in W/K2) at room temperature (293 K) for polytetrafluoroethylene (PTFE). Assume the values for thermal conductivity and electrical resistance to be 0.25 W/mK and 1017 m. 6. For the following pair of materials, decide which has the larger thermal conductivity. fused silica, granite □ Fused silica will have a large thermal conductivity. □ Granite will have a large thermal conductivity. □ Not enough information is known. 7. Nonsteady-state heat flow may be described by the following partial differential equation: T 2T DT 2 t x where DT is the thermal diffusivity; this expression is the thermal equivalent of Fick’s second law of diffusion. The thermal diffusivity is defined according to k DT C P In this expression, k, , and CP represent the thermal conductivity, the mass density, and the specific heat at constant pressure, respectively. Determine values of DT for polypropylene using SI units. The values for density, thermal conductivity, and specific heat capacity are 0.905 g/cm3, 0.12 W/mK, and 1925 J/kgK, respectively. 8. If a cylindrical rod of brass 150.00 mm long and 10.000 mm in diameter is heated from 10C to 130C while its ends are maintained rigid, determine its change in diameter. The value of l for brass is 20.010-6 (C)-1, and Poisson’s ratio is 0.34. 9. The two ends of a cylindrical rod of nickel 120.00 mm long and 12.000 mm in diameter are maintained rigid. If the rod is initially at 60C, to what temperature must it be cooled in order to have a 0.024-mm reduction in diameter? The values of Poisson’s ratio and l for nickel are 0.31 and 13.310-6 (C)-1, respectively. 10. Equation 19.9 (TSR = fk/El), for the thermal shock resistance of a material, is valid for relatively low rates of heat transfer. When the rate is high, then, upon cooling of a body, the maximum temperature change allowable without thermal shock, Tf, is approximately T f f E l where f is the fracture strength. Determine Tf for concrete. The values for modulus of elasticity, tensile strength, and coefficient of thermal expansion are 31 GPa, 39.3 MPa, and 11.810-6 C-1 respectively.