Acids/Bases and Cells
advertisement

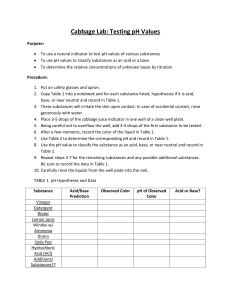
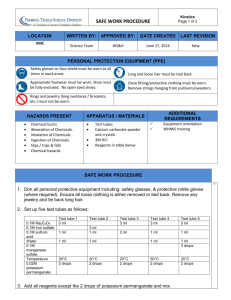
Acids, Bases, and Cells BACKGROUND: Cells must deal with many environmental and internal changes. These changes can affect the processes of cells. Cells need to maintain Homeostasis (a balance) between the inside and outside of themselves. One condition that cells must deal with constantly is pH. pH is a measure of the hydrogen (H) ion concentration of a solution. pH measures how acidic or basic something is. The pH scale is below. Changes in pH can change the structure of proteins, which are the workhorses of cells. These changes affect the structure and then affect function. Most cells maintain a pH close to neutral. Biochemical activities of living tissues frequently tend to change the pH. Yet life depends upon maintaining a pH that will not inactivate the enzymes or other cellular molecules from working in that tissue. Enzymes, which are a very important type of protein, are essential for the many chemical reactions in a cell. Buffers are solutions that resist changes in pH. PURPOSES: How are living cells affected by changes in pH? What are buffers and how do they affect a living cell? Materials: pH probe and Lab Quest 0.1M HCl 0.1M NaOH Commercial Buffer solution, pH 7 Liver and potato homogenates 2 50 mL beakers PROCEDURE/DATA- HCl- adding Acid READ AND CREATE ONE DATA TABLE FOR THE DATA BELOW Pour 25 ml of tap water into a 50 mL beaker. Use the pH probe and LabQuest to measure pH after every 5 drops of .1 M HCl until a total of 30 drops is reached. Swirl in between each set of 5 drops. Copy data into your notebook. Rinse the beaker and pour into it 25 mL of liver homogenate. Use the pH probe and LabQuest to measure pH after every 5 drops of .1 M HCl until a total of 30 drops is reached. Swirl in between each set of 5 drops. Copy data into your notebook. Rinse the beaker and pour into it 25 mL of potato homogenate. Use the pH probe and LabQuest to measure pH after every 5 drops of .1 M HCl until a total of 30 drops is reached. Swirl in between each set of 5 drops. Copy data into your notebook. Rinse the beaker and pour into it 25 mL of commericial buffer Use the pH probe and LabQuest to measure pH after every 5 drops of .1 M HCl until a total of 30 drops is reached. Swirl in between each set of 5 drops. Copy data into your notebook. REPEAT THE ABOVE USING NaOH- Adding Base CREATE TABLE FOR THIS DATA