organisms laboratory
advertisement

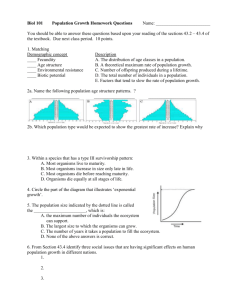
S2 OPERATING INSTRUCTIONS Date: 12.02.2016 Field of Application: Gebäude und Raumnummern angeben, Räume mit abweichender Einstufung (S1) vermerken. Project Manager:Name, Tel. - ggf. weitere/stellvertr. Proj.Man. angeben Emergency Phone Number: 0-112 Responsible for Biological Safety:Name, Tel. First Aider: Name, Tel. Company Medical Officer: Name, Tel. First Aid Case: Standort HAZARDS AND HAZARD DESCRIPTIONS BIO II Security Level S2 Genetic Operations - Genetic operations of which – at the current state of scientific knowledge and in keeping with these instructions – only low risks to human health or to the environment proceed are classified as Security Level S2. - Risk Group 2 micro-organisms as well as Security Level 2 genetically modified organisms (GVO) may cause infections and illness. - Incorporation is possible through the inhalation of aerosols, the swallowing of test specimens containing causative organisms, the penetration of causative organims into skin damage which either already exists or is due to injury, or through contact with the eyes and mucous membranes. Aerosoles may occur due to many typical laboratory activities (e.g. decantation, plating, pipetting, vortexing) and thus may cause infections in case of exposure. - Hier ggf. weitergehende arbeitsplatzbezogene Gefahren angeben! (Verwendete biologische Agenzien? Infektionsrisiken? - Krankheitssymptome?) - SAFETY PRECAUTIONS AND RULES OF CONDUCT - The handling of genetically modified organisms of risk group 2 may only be carried out in a genetic laboratory of security level 2 (or higher). - Access is only granted to those persons who are sufficiently qualified and have been authorized and instructed by the project manager. In addition to fulfilling the requirements of these operating instructions, all employees are to be annually briefed by the project manager on any changes relevant to safety matters. Female employees must be aware of the hazards particular to expectant mothers. Prior to laboratory access, a briefing on the operating instructions is obligatory and must be confirmed in writing. Further regulations (e.g. hazardous materials regulations, law on the protection of expectant and nursing mothers) may be viewed in room Raum-Nr?. - Wear a buttoned laboratory coat, sturdy footgear, safety glasses, and – if necessary – disposable gloves. Protective equipment is only to be worn in the working areas and must be taken off before leaving. - Doors and windows must be kept closed during laboratory work. - Do not eat, drink, smoke, use snuff, chew chewing gum, or wear cosmetics in the laboratory. Do not keep foodstuffs, beverages, tobacco, or cosmetics in the laboratory. - While pippeting use suitable equipment only. No oral pippeting! - When directly handling contagious material, wear disposable gloves and exchange if necessary. Avoid smear contamination (e.g. on telephones, door handles, fitting, and keyboards). Change gloves if necessary or after each work step. - Use syringes, canulas, and scalpels only when absolutely necessary. Put used canulas into the canula waste container directly after use; never put them back into their protective covering. - All operations during which aerosol formation may occur are to be carried out in a safety cabinet. Keep safety cabinets clean and tidy. After finishing work in safety cabinets, disinfect work surfaces according to hygiene regulations, clean surfaces, and turn on UV light. - Regularly check identity and purity of organisms if necessary for the evaluation of their potential danger. Check intervals are based on potential danger. The person responsible for this is Name. - Hier Verfahren beschreiben! - All work surfaces are to be disinfected after finishing operations. Adhere to hygiene regulations! - Hands are to be disinfected and then washed after finishing work and before leaving the working areas (especially after skin contact with materials containing causative organisms). After that, adhere to skin care plan. - Keep the working areas clean and tidy. Only required devices and materials should be present on work surfaces. Keep supplies only in rooms and closets made for available for that purpose. - Clean and disinfect, sterilize, or autoclave contaminated devices and instruments according to hygiene regulations. - Remove contaminated clothes (including underwear) and only reuse after disinfection. - Vermin and carriers of genetically modified organisms are to be exterminated in a suitable way. - Genetically modified organisms are to be kept in tightly closed and secure containers. - Genetically modified organisms as well as waste material containing genetically modified organisms are to be transported only in locked containers which are secured against breakage (surfaces are to be disinfected prior to transport). Containers must be labeled. - Employees dealing with security level 2 genetic operations may if necessary need to be medically examined (prior to beginning work and on a yearly basis). Vaccinations may be required. - Changes in health condition (e.g. impaired immune system) or pregnancy have to be reported. CONDUCT IN THE CASE OF DANGER - Employees are to be warned if genetically modified organisms of risk group 2 are released. Affected work areas might need to be closed off. Immediately inform the project manager as well as the person responsible for biological safety. - See to personal safety (safety glasses, disposable gloves, etc.) when cleaning up hazardous waste. In case of aerosol presence use filtration half masks (protection level FFP3 SL). - Absorb fluids with tissue papers or the like. Immediatle disinfect and the clean the contaminated area with suitable disinfectant according to hygiene regulations. Observe application time! - Windows and doors must stay closed until cleaning procedure is completed. Unauthorized personnel may not enter the area. - Autoclave all contaminated objects. FIRST AID - Open wounds are to be thoroughly cleaned or left to bleed under medical supervision. Spray with wound disinfectant (e.g. Cutasept) immediately. Follow hygiene regulations. - In the case of contact with the eyes, wash eyes with eye shower for at least 10 minutes. Consult an eye doctor. - If material containing causative organisms enters the mouth, spit it out immediately, rinse and gargle with bactericidal solution (e.g. Hexarol). - Any injuries are to be reported to the project manager and the person responsible for biological safety. Injuries are to be recorded in the accident log. - In the case of extreme contact with hazardous materials (e.g. swallowing, inhalation, incorporation into wounds) consult a doctor (university hospital, emergency surgery 0-597-4367) or call an ambulance. Mention agents used. - Contact first aider and company medical officer. PROPER WASTE DISPOSAL GVO - Waste material containing genetically modified organisms is to be labeled ("GVO") and preferably to be autoclaved prior to leaving the laboratory. Otherwise, it is to be deactivated with disinfectant. An autoclave is located in Standort?.