150715195347suschemEfomatAbstractsubmissiontemplate
advertisement

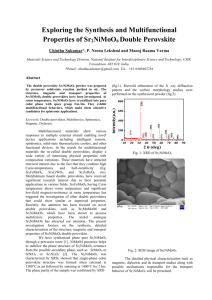
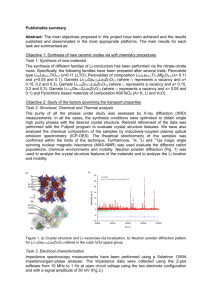
SusChemE 2015 International Conference on Sustainable Chemistry & Engineering October 8-9, 2015, Hotel Lalit, Mumbai Syngas through CO2 reforming of methane over AZr1-xNixO3 (A= Ca, Sr and Ba) perovskites Srikanth Dama, Seema Ghodke, Hanmant R. Gurav, Richa A Bobade, Satyanarayana Chilukuri* Catalysis Division, CSIR-National Chemical Laboratory, Dr. Homi Bhabha Road, Pune-411008, India E-mail addresses: sn.dama@ncl.res.in, s.ghodke@ncl.res.in, hr.gurav@ncl.res.in, ra.bobade@ncl.res.in, sv.chilukuri@ncl.res.in. 1. Introduction: Among the green house gases, CO2 contributes a major part to the global warming. If urgent steps are not taken to either control its emissions or utilize it for manufacture of chemicals, CO 2 concentration can reach upto 991 ppm by the year 2100 [1]. In this context, CO2 reforming or dry reforming of methane (DRM) is important as it converts two potent green house gases (CH 4 and CO2) to yield synthesis gas with low H2/CO ratio (~1), which is the suitable feed for Fischer-Tropsch and DME synthesis [2]. But, in DRM process, rapid deactivation of catalysts as a result of carbon deposition and sintering of metals due to high reaction temperatures are the major issues. [2]. Herein we report synthesis and characterization of Ca, Sr and Ba substituted AZr 1-xNixO3 (A = Ca, Sr and Ba) perovskites with the view to develop a catalyst that prevents formation of coke and resists sintering at high temperatures in DRM reaction. 2. Material and Methods: Ca, Sr and Ba substituted AZr1-xNixO3 (A = Ca, Sr and Ba) perovskites were prepared through modified pechini method and characterized by means of XRD, BET surface area, Raman spectroscopy, XPS and temperature programmed (TP) techniques. Catalyst evaluation through DRM was carried out at atmospheric pressure in a fixed bed down flow Incolloy HT reactor (19mm OD and 11mm ID). The product gas containing H 2, CO, CO2 and CH4 was analyzed using Chemito-1000 GC equipped with TCD and Spherocarb (1/8” OD, 8’) column. 3. Significant Results and Discussion: Figure 2: CH4 conversion vs TOS for AZr1-xNixO3 (A = Ca, Sr and Ba) Figure 1: XRD pattern of perovskite catalysts. 1 SusChemE 2015 International Conference on Sustainable Chemistry & Engineering October 8-9, 2015, Hotel Lalit, Mumbai PXRD (Figure 1) of Ni substituted AZrO3 (A = Ca, Sr and Ba) perovskite crystallized in cubic phase (Pm3m) at 1023 K. Figure 2 shows that the conversion of CH4 strongly depended on substituted element in the A site of perovskite type oxide and maximum conversion was achieved for Ca substituted zirconate (CZO) catalyst. A similar trend was observed in the case of CO 2 conversion. The stoichiometric ratio of H2 to CO, which is expected to be ‘one’ was lower indicating that the conversion of CO2 was higher than CH4, due to reverse water gas shift reaction. The perovskite structures could be retained during the dry reforming reaction and the catalyst stability could be related to the basicity of the perovskite. 4. Conclusions: Ca, Sr and Ba substituted AZr1-xNixO3 (A = Ca, Sr and Ba) family of perovskites were successfully prepared. The substitution of A site cations in the perovskite type lattice with increasing ionic radii from Ca to Ba lowered the reduction temperature of Ni and led to the reduced activity of the zirconate perovskite. The Ca substituted zirconate perovskite proved to be the most stable catalyst. Correlation between the effect of substitution of A-site cations in the perovskite lattice and the catalytic activity and stability of in situ formed Ni particles were established. References [1] M.C.J. Bradford, M. A. Vannice, Catal. Rev-Sci. Eng, 41, 1999, 1-42. [2] T. Sukonket, A. Khan, B. Saha, H. Ibrahim, S. Tantayanon, P. Kumar, R. Idem, Energy Fuels, 25, 2011, 864-877. 2