Equilibrium Practice Test # 2 ANSWER KEY
advertisement
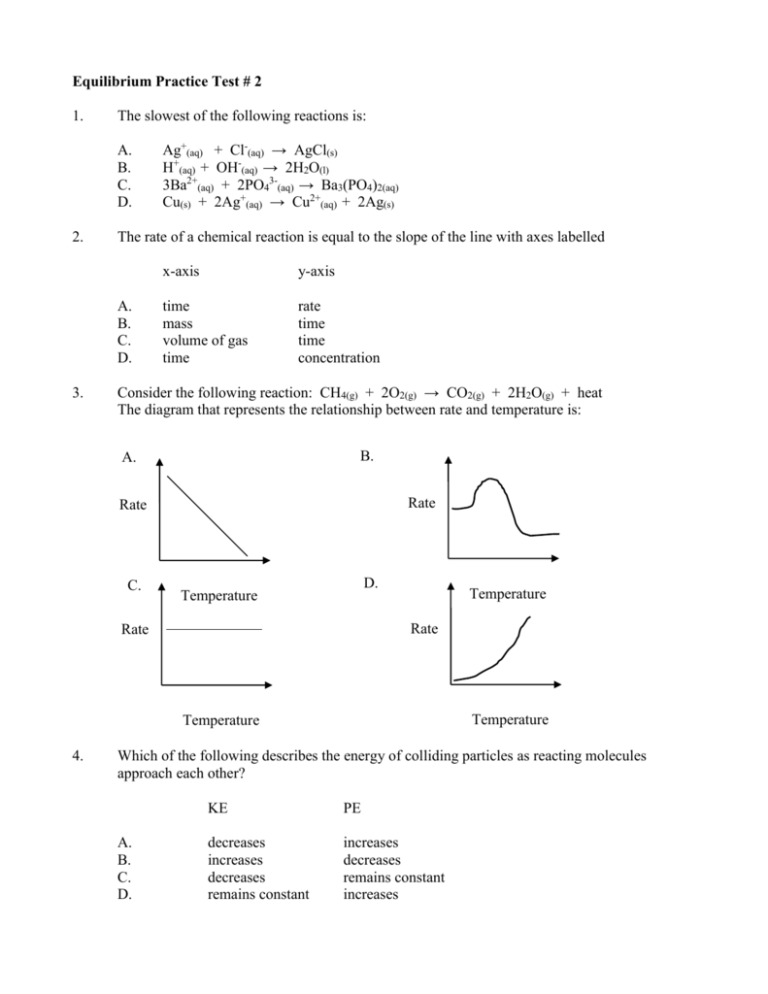
Equilibrium Practice Test # 2 1. The slowest of the following reactions is: A. B. C. D. 2. The rate of a chemical reaction is equal to the slope of the line with axes labelled A. B. C. D. 3. Ag+(aq) + Cl-(aq) → AgCl(s) H+(aq) + OH-(aq) → 2H2O(l) 3Ba2+(aq) + 2PO43-(aq) → Ba3(PO4)2(aq) Cu(s) + 2Ag+(aq) → Cu2+(aq) + 2Ag(s) x-axis y-axis time mass volume of gas time rate time time concentration Consider the following reaction: CH4(g) + 2O2(g) → CO2(g) + 2H2O(g) + heat The diagram that represents the relationship between rate and temperature is: B. A. Rate Rate C. D. Temperature Temperature Rate Rate Temperature Temperature 4. Which of the following describes the energy of colliding particles as reacting molecules approach each other? A. B. C. D. KE PE decreases increases decreases remains constant increases decreases remains constant increases 5. The average kinetic energy per molecule can be increased by A. B. 6. increasing temperature increasing reactant concentration increasing [H2] decreasing the volume C. D. finely powdering the C(s) decreasing the temperature Consider the following equilibrium: H2(g) + I2(g) ⇄ 2HI(g) At equilibrium [H2] = 0.00220 M, [I2] = 0.00220 M, and [HI] = 0.0156 M The value of the Keq is A. B. 8. C. D. Consider the following reaction: C(s) + 2H2(g) ⇄ CH4(g) ΔH = -74.8 kJ Which of the following will cause an increase in the value of the Keq? A. B. 7. adding a catalyst increasing pressure 3.10 x 10-4 1.99 x 10-2 C. D. 5.03 x 101 3.22 x 103 Consider the rate diagram for the following reaction: 2HI(g) ⇄ H2(g) + I2(g) forward Rate reverse Time t1 Which of the following occurs at t1? A. B. 9. addition of H2 addition of HI C. D. addition of a catalyst a decrease in volume Chemical equilibrium is said to be dynamic because A. the reaction proceeds quickly B. the mass of the reactants is decreasing C. the macroscopic properties are constant D. both forward and reverse rates are occurring 10. 11. Which equation has the largest value of Keq? A. N2(g) + O2(g) ⇄ 2NO(g) ΔH = 21 kJ B. C2H6(g) ⇄ 2C(s) + 3H2(g) ΔH + 83 kJ C. H2(g) + 1/2O2(g) ⇄ H2O(g) ΔH = -240 kJ D. Ca(s) + 3H2O(l) ⇄ Ca(OH)2(aq) + H2(g) ΔH = -240 kJ The value of the Keq can be changed by A. B. 12. 0.10 0.30 C. D. 3.3 10 C.right because the Trial Keq > Keq D.right because the Trial Keq < Keq Given the following system: 2CrO42-(aq) + 2H+(aq) ⇄ Cr2O72-(aq) + H2O(l) Which of the following chemicals, when added to the above system at equilibrium, would result in a decrease in [Cr2O72-]? A. B. 15. changing the reactant concentration changing the volume of the container Consider the following equilibrium: PCl3(g) + Cl2(g) ⇄ PCl5(g) Keq = 2.30 A 1.0 L container is filled with 0.05 mole PCl5, 1.0 mole PCl3, and 1.0 mole Cl2. The system proceeds to the A.left because the Trial Keq > Keq B.left because the Trial Keq < Keq 14. C. D. Consider the following equilibrium: PCl3(g) + Cl2(g) ⇄ PCl5(g) When 0.40 mole of PCl3 and 0.40 mole of Cl2 are placed in a 1.00 L container and allowed to reach equilibrium, 0.244 mole of PCl5 are present. From this information, the value of the Keq is A, B. 13. adding a catalyst changing the temperature NaOH HNO3 C. D. What is the Keq expression for the following equilibrium? 3Fe(s) + 4H2O(g) ⇄ Fe3O4(s) + 4H2(g) A. B. C. D. Keq = [H2]4 Keq = [H2] [H2O] Keq = [H2]4 [H2O]4 Keq = [Fe2O3][H2]4 [Fe][H2O]4 Na2CrO4 Na2Cr2O7 16. Consider the following equilibrium: 2O3(g) ⇄ 3O2(g) Keq = 65 Initially 0.10 mole of O3 and 0.10 mole of O2 are placed in a 1.0 L container, Which of the following describes the changes in concentrations as the reaction proceeds towards equilibrium? A. B. C. D. 17. [O2] decreases decreases increases increases decreases increases decreases increases Consider the following equilibrium: 2CrO42-(aq) + 2H+(aq) ⇄ Cr2O72-(aq) + H2O(l) yellow orange An unknown solution is added to an orange equilibrium system until the sample turns yellow. The solution could be A. B. 18. [O3] KNO3 NaOH C. D. NH4NO3 CH3COOH Consider the following equilibrium: CH3COOH(aq) ⇄ CH3COO-(aq) + H+(aq) + heat A stress was applied at time t1 and the data plotted on the following graph: The stress imposed at time t1 is the result of [H+] time A. B. t1 the addition of HCl decreasing the temperature C. D. the addition of NaCH3COO increasing the volume of the container 19. Consider the following potential energy diagram for an equilibrium system: P.E. Progress of the reaction When the temperature of the system is increased, the equilibrium shifts to the A. B. 20. C. D. right and the Keq increases right and the Keq decreases Addition of a catalyst to an equilibrium system A. B. C. D. 21. left and the Keq increases left and the Keq decreases increases the value of the Keq increases the yield of the product has no effect on the rates of the reaction increases the rates of formation of both reactants and products Ammonia, NH3, is produced by the following reaction: N2(g) + 3H2(g) 2NH3(g) + energy Which of the following would result in the highest concentration of ammonia at equilibrium? A. B. C. D. 22. increasing the temperature and increasing the pressure decreasing the temperature and increasing the pressure increasing the temperature and decreasing the pressure decreasing the temperature and decreasing the pressure Consider the following equilibrium: 2NO2(g) N2O4(g) Keq = 1.15 The equilibrium concentration of NO2 is 0.05mol/L. Calculate the equilibrium concentration of N2O4(g). A. B. 0.22 mol/L 0.29 mol/L C. D. 0.43 mol/L 0.58 mol/L 23. Consider the following equilibrium: H2(g) + I2 2HI(g) Keq = 50.0 What is the value Keq for the reaction rewritten as: 2HI(g) H2(g) + I2(g) A. B. 24. Keq = ? -50.0 0.0200 C. D. 25.0 50.0 Consider the following equilibrium: 2NOCl(g) 2NO(g) + Cl2(g) A flask is filled with NOCl, NO, and Cl2(g). Initially there were a total of 5.0 moles of gases present. When equilibrium is reached, there are a total of 8.0 moles of gases present. Which of the following explains the observation? A. B. C. D. 25. Consider the following equilibrium: 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) + energy Which of the following will cause the equilibrium to shift to the left? A. B. 26. adding H2O(g) removing some NO(g) C. D. increasing the volume decreasing the temperature A catalyst is added to a system already at equilibrium. How are the forward and reverse reaction rates affected by the addition of the catalyst. A. B. C. D. 27. The reaction shifted left because the Trial Keq > Keq The reaction shifted left because the Trial Keq < Keq The reaction shifted right because the Trial Keq > Keq The reaction shifted right because the Trial Keq < Keq Forward Rate Reverse Rate increases increases constant constant increases constant decreases constant Consider the following equilibrium: 2NOBr(g) 2NO(g) + Br2(g) Keq = 0.064 At equilibrium, a 1.00 L flask contains 0.030 mole NOBr and 0.030 mole NO. How many moles of Br2 are present? A. B. 0.0019 0.064 C. D. 0.030 0.47 28. Which of the following does not apply to all chemical equilibrium systems? A. B. C. D. 29. The relationship between Ea and reaction rate is best represented as A. C. 30. B. D. The relationship between Keq and temperature for an exothermic reaction is best represented as A. C. 31. They are closed. The macroscopic properties are constant Forward and reverse rates are equal There are equal concentrations of reactants and products B. D. The relationship between reaction rate and temperature is best represented by A. C. B. D. 32. The relationship between Ea and temperature is best represented by A. B. D. C. 32. Methanol, CH3OH, can be produced by the following: CO(g) + 2H2(g) CH3OH(g) + energy The conditions necessary to maximize the equilibrium yield of CH3OH are A.low temperature and low pressure B.high temperature and low pressure 33. low temperature and high pressure high temperature and high pressure Consider the following equilibrium: 2NO(g) + O2(g) 2NO2(g) + energy When the volume of the container is increased, the equilibrium shifts to the A. B. C. D. 34. C. D. left and the Keq decreases right and the Keq increases left and the Keq remains constant right and the Keq remains comstant Consider the following reaction: C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) H = -2202 kJ Which of the following applies to the forward reaction? A. B. C. D. Entropy Enthalpy increases increases decreases decreases increases decreases increases decreases Subjective 1. Consider the following equilibrium: N2H4(g) + 2O2(g) 2NO2(g) + 2H2O(g) More oxygen is added to the above equilibrium. After the system re-establishes equilibrium, identify the substance(s), if any, that have a net 2. a) increase in concentration b) decrease in concentration Given the following equilibrium: H2(g) + I2(g) 2HI(g) Initially, 0.200 mole H2 and 0.200 mole I2 were placed into a 1.0 L container. At equilibrium, the [I2] is 0.040 M. Calculate the Keq. 3. Consider the following equilibrium: 2CrO42-(aq) + 2H+(aq) ⇄ Cr2O72-(aq) + H2O(l) yellow orange When HCl is added, the solution turns orange. Explain why this colour change occurs. 4. Consider the following equilibrium system: N2(g) + 3H2(g) 2NH3(g) + energy A 1.00 L container is filled with 7.0 mole NH3 and the system proceeds to equilibrium as indicated by the graph. 8.0 M 6.0 4.0 2.0 NH3 a) Draw and label the graph for N2 and H2. Fill in an ICE chart if you are not sure how to do this. N2(g) + 3H2(g) 2NH3(g) I C E b) Calculate the Keq for the above reaction. 5. Consider the following equilibrium 2NO(g) + O2(g) 2NO2(g) Keq = 1.5 0.800 mole NO, 0.600 moles O2, and 0.400 moles NO2 are placed in a vessel that 2.0 L. Show by calculation that the reaction is not at equilibrium? What will happen to [O2] as equilibrium is approached? 6. Consider the following equilibrium: SO3(g) + NO(g) NO2(g) + SO2(g) Keq = 0.500 Exactly 0.100 mole SO3 and 0.100 mole NO were placed in a 1.00 L flask and allowed to go to equilibrium. Calculate the equilibrium concentration of SO2. Equilibrium Practice Test # 2 ANSWER KEY 1. The slowest of the following reactions is: A. B. C. D. 2. The rate of a chemical reaction is equal to the slope of the line with axes labelled A. B. C. D. 3. Ag+(aq) + Cl-(aq) → AgCl(s) H+(aq) + OH-(aq) → 2H2O(l) 3Ba2+(aq) + 2PO43-(aq) → Ba3(PO4)2(aq) Cu(s) + 2Ag+(aq) → Cu2+(aq) + 2Ag(s) x-axis y-axis time mass volume of gas time rate time time concentration Consider the following reaction: CH4(g) + 2O2(g) → CO2(g) + 2H2O(g) + heat The diagram that represents the relationship between rate and temperature is: B. A. Rate Rate C. D. Temperature Temperature Rate Rate Temperature Temperature 4. Which of the following describes the energy of colliding particles as reacting molecules approach each other? A. B. C. KE PE decreases increases decreases increases decreases remains constant D. 5. increasing [H2] decreasing the volume finely powdering the C(s) decreasing the temperature Consider the following equilibrium: H2(g) + I2(g) ⇄ 2HI(g) At equilibrium [H2] = 0.00220 M, [I2] = 0.00220 M, and [HI] = 0.0156 M The value of the Keq is A. B. C. D. 8. adding a catalyst increasing pressure increasing temperature increasing reactant concentration Consider the following reaction: C(s) + 2H2(g) ⇄ CH4(g) ΔH = -74.8 kJ Which of the following will cause an increase in the value of the Keq? A. B. C. D. 7. increases The average kinetic energy per molecule can be increased by A. B. C. D. 6. remains constant 3.10 1.99 5.03 3.22 x x x x 10-4 10-2 101 103 Consider the rate diagram for the following reaction: 2HI(g) ⇄ H2(g) + I2(g) forward Rate reverse Time t1 Which of the following occurs at t1? A. B. C. D. 9. addition of H2 addition of HI addition of a catalyst a decrease in volume Chemical equilibrium is said to be dynamic because A. B. C. D. 10. 11. 12. Which equation has the largest value of Keq? A. N2(g) + O2(g) ⇄ 2NO(g) ΔH = 21 kJ B. C2H6(g) ⇄ 2C(s) + 3H2(g) ΔH + 83 kJ C. H2(g) + 1/2O2(g) ⇄ H2O(g) ΔH = -240 kJ D. Ca(s) + 3H2O(l) ⇄ Ca(OH)2(aq) + H2(g) ΔH = -240 kJ The value of the Keq can be changed by A. adding a catalyst B. changing the temperature C. D. changing the reactant concentration changing the volume of the container Consider the following equilibrium: PCl3(g) + Cl2(g) ⇄ PCl5(g) When 0.40 mole of PCl3 and 0.40 mole of Cl2 are placed in a 1.00 L container and allowed to reach equilibrium, 0.244 mole of PCl5 are present. From this information, the value of the Keq is A, B. C. D. 13. the reaction proceeds quickly the mass of the reactants is decreasing the macroscopic properties are constant both forward and reverse rates are occurring 0.10 0.30 3.3 10 Consider the following equilibrium: PCl3(g) + Cl2(g) ⇄ PCl5(g) Keq = 2.30 A 1.0 L container is filled with 0.05 mole PCl5, 1.0 mole PCl3, and 1.0 mole Cl2. The system proceeds to the A. B. C. D. left because the Trial Keq > Keq left because the Trial Keq < Keq right because the Trial Keq > Keq right because the Trial Keq < Keq 14. Given the following system: 2CrO42-(aq) + 2H+(aq) ⇄ Cr2O72-(aq) + H2O(l) Which of the following chemicals, when added to the above system at equilibrium, would result in a decrease in [Cr2O72-]? A. NaOH B. HNO3 C. D. 15. Na2CrO4 Na2Cr2O7 What is the Keq expression for the following equilibrium? 3Fe(s) + 4H2O(g) ⇄ Fe3O4(s) + 4H2(g) Keq = [H2]4 Keq = [H2] [H2O] Keq = [H2]4 [H2O]4 Keq = [Fe2O3][H2]4 [Fe][H2O]4 A. B. C. D. 16. Consider the following equilibrium: 2O3(g) ⇄ 3O2(g) Keq = 65 Initially 0.10 mole of O3 and 0.10 mole of O2 are placed in a 1.0 L container, Which of the following describes the changes in concentrations as the reaction proceeds towards equilibrium? A. B. decreases increases decreases increases Consider the following equilibrium: 2CrO42-(aq) + 2H+(aq) ⇄ Cr2O72-(aq) + H2O(l) yellow orange An unknown solution is added to an orange equilibrium system until the sample turns yellow. The solution could be KNO3 NaOH C. D. 18. decreases increases increases A. B. [O2] decreases C. D. 17. [O3] NH4NO3 CH3COOH Consider the following equilibrium: CH3COOH(aq) ⇄ CH3COO-(aq) + H+(aq) + heat A stress was applied at time t1 and the data plotted on the following graph: The stress imposed at time t1 is the result of [H+] time t1 A. B. C. D. 19. the addition of HCl decreasing the temperature the addition of NaCH3COO increasing the volume of the container Consider the following potential energy diagram for an equilibrium system: P.E. Progress of the reaction When the temperature of the system is increased, the equilibrium shifts to the A. B. C. D. 20. left and the Keq increases left and the Keq decreases right and the Keq increases right and the Keq decreases Addition of a catalyst to an equilibrium system A. B. C. increases the value of the Keq increases the yield of the product has no effect on the rates of the reaction D. increases the rates of formation of both reactants and products 21. Ammonia, NH3, is produced by the following reaction: N2(g) + 3H2(g) 2NH3(g) + energy Which of the following would result in the highest concentration of ammonia at equilibrium? A. increasing the temperature and increasing the pressure B. decreasing the temperature and increasing the pressure C. D. 22. increasing the temperature and decreasing the pressure decreasing the temperature and decreasing the pressure Consider the following equilibrium: 2NO2(g) N2O4(g) Keq = 1.15 The equilibrium concentration of NO2 is 0.05 mol/L. Calculate the equilibrium concentration of N2O4(g). 23. A. 0.22 mol/L B. 0.29 mol/L C. D. 0.43 mol/L 0.58 mol/L Consider the following equilibrium: H2(g) + I2 2HI(g) Keq = 50.0 What is the value Keq for the reaction rewritten as: 2HI(g) H2(g) + I2(g) A. B. -50.0 0.0200 C. D. 24. Keq = ? 25.0 50.0 Consider the following equilibrium: 2NOCl(g) 2NO(g) + Cl2(g) A flask is filled with NOCl, NO, and Cl2(g). Initially there were a total of 5.0 moles of gases present. When equilibrium is reached, there are a total of 8.0 moles of gases present. Which of the following explains the observation? A. B. C. The reaction shifted left because the Trial Keq > Keq The reaction shifted left because the Trial Keq < Keq The reaction shifted right because the Trial Keq > Keq D. The reaction shifted right because the Trial Keq < Keq 25. Consider the following equilibrium: 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) + energy Which of the following will cause the equilibrium to shift to the left? A. adding H2O(g) B. C. D. 26. A catalyst is added to a system already at equilibrium. How are the forward and reverse reaction rates affected by the addition of the catalyst. A. B. C. D. 27. increases increases constant constant increases constant decreases constant 0.0019 0.064 0.030 0.47 They are closed. The macroscopic properties are constant Forward and reverse rates are equal There are equal concentrations of reactants and products The relationship between Ea and reaction rate is best represented as A. C. 30. Reverse Rate Which of the following does not apply to all chemical equilibrium systems? A. B. C. D. 29. Forward Rate Consider the following equilibrium: 2NOBr(g) 2NO(g) + Br2(g) Keq = 0.064 At equilibrium, a 1.00 L flask contains 0.030 mole NOBr and 0.030 mole NO. How many moles of Br2 are present? A. B. C. D. 28. removing some NO(g) increasing the volume decreasing the temperature B. D. The relationship between Keq and temperature for an exothermic reaction is best represented as A. B. 31. The relationship between reaction rate and temperature is best represented by A. C. 32. D. The relationship between Ea and temperature is best represented by A. C. 32. B. B. D. Methanol, CH3OH, can be produced by the following: CO(g) + 2H2(g) CH3OH(g) + energy The conditions necessary to maximize the equilibrium yield of CH3OH are A. B. C. low temperature and low pressure high temperature and low pressure low temperature and high pressure D. 33. Consider the following equilibrium: 2NO(g) + O2(g) 2NO2(g) + energy When the volume of the container is increased, the equilibrium shifts to the A. B. C. D. 34. left and the Keq decreases right and the Keq increases left and the Keq remains constant right and the Keq remains comstant Consider the following reaction: C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) H = -2202 kJ Which of the following applies to the forward reaction? A. B. high temperature and high pressure Entropy Enthalpy increases increases increases C. D. decreases decreases decreases increases decreases Subjective 1. Consider the following equilibrium: N2H4(g) + 2O2(g) 2NO2(g) + 2H2O(g) More oxygen is added to the above equilibrium. After the system re-establishes equilibrium, identify the substance(s), if any, that have a net 2. a) increase in concentration NO b) decrease in concentration N2H4 Given the following equilibrium: H2 O H2(g) + I2(g) O2 2HI(g) Initially, 0.200 mole H2 and 0.200 mole I2 were placed into a 1.0 L container. At equilibrium, the [I2] is 0.040 M. Calculate the Keq. H2(g) I + 0.200 M I2(g) 0.200 M 2HI(g) 0 3. C 0.160 M 0.160 M 0.320 M E 0.040 M 0.040 M 0.320 M Keq = (0.320)2 (040)2 = 64 Consider the following equilibrium: 2CrO42-(aq) + 2H+(aq) ⇄ Cr2O72-(aq) + H2O(l) yellow orange When HCl is added, the solution turns orange. Explain why this colour change occurs. Adding HCl increases the [H+] and shifts the system right turning orange. 4. Consider the following equilibrium system: N2(g) + 3H2(g) 2NH3(g) + energy A 1.00 L container is filled with 7.0 mole NH3 and the system proceeds to equilibrium as indicated by the graph. H2 8.0 M 6.0 4.0 N2 2.0 NH3 a) Draw and label the graph for N2 and H2. Fill in an ICE chart if you are not sure how to do this. N2(g) I C 0 3.0 + 3H2(g) 0 9.0 2NH3(g) 7.0 6.0 E b) 3.0 1.0 Calculate the Keq for the above reaction. Keq 5. 9.0 = (1.0)2 = (3.0)(9.0)3 0.00046 Consider the following equilibrium 2NO(g) + O2(g) 2NO2(g) Keq = 1.5 0.800 mole NO, 0.600 moles O2, and 0.400 moles NO2 are placed in a vessel that 2.0 L. Show by calculation that the reaction is not at equilibrium? What will happen to [O2] as equilibrium is approached? Trial Keq (0.200)2 (0.400)2(0.300) = Not at equilibrium 6. = Shifts Right 0.833 < Keq = 1.5 [O2] will decrease. Consider the following equilibrium: SO3(g) + NO(g) NO2(g) + SO2(g) Keq = 0.500 Exactly 0.100 mole SO3 and 0.100 mole NO were placed in a 1.00 L flask and allowed to go to equilibrium. Calculate the equilibrium concentration of SO2. SO3(g) + NO(g) NO2(g) + SO2(g) I 0.100 0.100 0 0 C x x x x E 0.100 – x 0.100 – x x x x2 (0.100 – x)2 = 0.500 x 0.100 – x = 0.7071 1.7071x = 0.07071 x = 0.0414 M [SO2] = 0.0414 M









