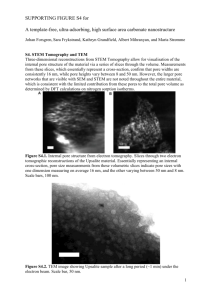
fr260704poster skirda.finaldoc
advertisement

1 DEPENDENCE OF SELF-DIFFUSION COEFFICIENT ON GEOMETRICAL PARAMETERS OF POROUS MEDIA *V.А. Sevriugin, **V.V. Loskutov, *V.D. Skirda *Kazan State University 420000, Kazan, Kremlyovskaya st., 18. **Mari State Pedagogical Institute 424002, Yoshkar-Ola, Communisticheskaya st., 44. The self-diffusion of liquids in porous media is a general problem of molecular physics with a wide range of practical applications. In [1,2] analytical expressions for the effective self-diffusion coefficient D (t ) of a liquid in a closed pore have been reported for the shorttime regime. It has been shown there that ratio of surface area to pore volume S V D(t ) t 0.5 . With and is defined by the t increase D(t ) changes in 1 accordance to D(t ) t , which corresponds to a regime of completely restricted particle diffusion. However, theoretical approaches describing diffusion in porous media in the long-time regime , taking into account geometry, pore connectivity and the sizes of molecules of liquids are have not been presented, though there are some empirical expressions. The dependence of the selfdiffusion coefficient Ds on the pore observable in a regime t for cyclohexane and cyclodecane in porous Vycor glass were described by [3]: t Ds 1 Do 1 1 d where Do - self-diffusion coefficient of a bulk liquid, (1) - a "permeability" of the medium, d – an average pore diameter. In [4] it was remarked that dependence (1) fails for experimental data at small d values. In the same way, attempts have been made to take into account the size of molecules in expression (1) by replacing d by d a , where a is an molecular effective diameter. 2 The aim of this work is to find the dependence of the molecule selfdiffusion coefficient in a liquid in a porous medium on the geometrical characteristics of the porous medium in a regime t . Let the molecule of a liquid in the porous media diffuse during a long 2 time t. The value L represents the sum of the squares of diffusion transitions 2i of the molecule in all pores: L2 i 2i . Then: L2 k 2 ( R) 6 Ds ( R)t (2) where k – an average number of diffusion transitions of the molecule from pore to pore during the time t , Ds (R ) – the measured self-diffusion 2 coefficient, ( R ) – an average square diffusion replacement of molecules for the time between two consecutive inputs to the neighboring pore. In (2) the average number of transitions k of molecules is defined by the average value of the "life"-time ( R ) of molecules in pore: k t (R) . In accordance with (2) it is possible to write for the self- diffusion coefficient 2 ( R ) Ds ( R) . 6 ( R ) (3) Let there be no specific interactions between molecules of a liquid in a pore and molecules of a material of the porous media, and the translational mobility of molecules of a liquid in the medium will be characterized by the 2 same characteristic sizes of length o and time o as in the bulk liquid. Generally, for any porous medium with any geometry, it is possible to consider the characteristic time determining the frequency of "interactions" of molecules of a liquid with the walls of the pore. Thus the average life-time ( R ) of molecules in a pore can be defined as R n R , where n R – an average number of "interactions" of a molecule with walls for a time from the moment a molecule enters the pore to the moment it exits. The number of "interactions" of any molecule with the walls of a pore for some interval of time t is a random variable and, it is assumed, follows the Poisson distribution Pn t e t n! where Pn – the probability for any molecule to interact with pore walls n times during t and 1 . The probability Pn is a function of the geometry of 3 the pore: ( Pn Pn ( R, t ) . The average t ): n R,t will be defined as n R, t nPn R, t . n 0 There are two possibilities for the molecule in the pore to interact with a boundary surface under diffusion, namely: to appear on an element of a boundary surface and to pass into the next pore, or to appear on an element of a surface S and to remain in the same pore (where – total effective area of channels). Two different probabilities correspond to "interaction" with a surface Pst and "interaction" with the channel P . It is necessary to note that both probabilities are functions of the pore size. Then the expression for an average number of "interactions" with boundary surfaces for a macroscopically long time t (R) is: n0 n0 n0 n R, k nPn R, k nPn R, k Pst R, k nPn R, k P R where k t (R) . At k 1 (4) expression (4) may be written in the time- independent form: n0 n0 n R,1 nPn R,1Pst R nPn R,1P R . In this expression the second sum is a constant close to unity. Then it is possible to write the differential equation for n R in the form: n R [ nPn R Pst R ] R R n0 P R n Pn1 R Pn R Pst R Pn R st . R n 0 For high n values probabilities therefore: Pn1 R and Pn R in (5) are equal and P R P R n R nPn R st n R st R R R n 0 and (5) 4 n R no exp Pst R . The value (6) no 1 in (6) is easily defined from the condition Pst R =0. Then, R according n R to (3) and taking into account that , we obtain the expression for the self-diffusion coefficient of a liquid in the pore: where 2 ( R) Ds ( R) exp[ Pst R ] Do exp[ Pst R ] , 6 Do – a self-diffusion coefficient of a bulk liquid. Ds/D0 1 exp(-11.04d0/d) 0,000 0,005 0,010 0,015 0,020 4 1/d, 10 A -1 Figure 1. Dependence of the relative self-diffusion coefficient Ds / D0 for benzene in porous media on the glass sphere diameter: straight line – theoretical values, symbol – experimental data. 5 Pst is the probability for a molecule to appear near the wall at the distance of the effective diameter of a molecule d 0 . Hence, this probability is equal to the ratio of the number of molecules in the layers near the walls to the total number of molecules in the pore or, otherwise, to the ratio of the volume of layers near the walls to the pore volume, that is: Pst do S / V . Finally, it is possible to write an expression for the self-diffusion coefficient of molecules of a liquid in a pore in long-time regime t as: 6Sdo Ds Do exp V (7) Figure 1 shows the experimentally observed dependences of the selfdiffusion coefficient diameter for benzene in porous media, formatted by glass spheres (the sphere diameters are: 44-53 mkm, 53-63 mkm, 63-74 mkm, 74-88 mkm). The diffusion measurement were made by the PFG-NMR method with a proton resonance frequency of 60 MHz. For this system the dependence of the area of a pore surface and its volume on the diameter of a glass sphere can be calculated: S=0.90675d2; V=0.08215d3; S/V=11.04 and (7) can write: Ds 11,04d 0 exp D0 d where d 0 =4.96 (8) A – is an effective diameter of benzene molecule. Fig.1 illustrates the theoretical values (8) Ds / D0 (straight line) and experimental data for glass spheres of different sizes. Acknowledgments This work was performed within the framework of projects: CRDF REC 007; CRDF RPO 1331; 6 References 1. Mitra P.P., Sen P.N., Schwartz L.M. (1993) Phys. Rev. 47, N.14, P. 8565. 2. Fatkullin N.F. (1992) ЖЭТФ. 101, № 5, P. 1561, 3. Mitzithras A., J.H. Strange. (1994) Magn. Res. Imagina. 12, N.2, P. 261. 4. Skirda M.V., Skirda V.D. (1997) Structure and dynamics of molecular systems./ Yoshkar-Ола, Moscow, Kazan,1. P. 186.