APL Materials Kai SI 24 Oct Revised RVK

Supporting Information
Graphene-wrapped Sulfur/Metal Organic Framework (MOF)-Derived
Microporous Carbon Composite for Lithium Sulfur Batteries
Renjie Chen 1,3
‡
*, Teng Zhao 1,3
‡
, Tian Tian 2 , Shuai Cao 3 , Paul R. Coxon 3 , Kai Xi 3 *,
David Fairen-Jimenez 2 , R. Vasant Kumar 3 and Anthony K. Cheetham 3
1 Beijing Key Laboratory of Environmental Science and Engineering, School of Chemical
Engineering and Environment, Beijing Institute of Technology, Beijing, 100081, China.
2 Department of Chemical Engineering and Biotechnology, Pembroke Street, University of
Cambridge, Cambridge CB2 3RA, United Kingdom.
3 Department of Materials Science and Metallurgy, University of Cambridge, Cambridge, CB3
0FS, United Kingdom.
__________________
*Author to whom correspondence should be addressed. Email: R.C. chenrj@bit.edu.cn
& K.X. kx210@cam.ac.uk
†
Electronic supplementary information (ESI) available.
‡
These authors contributed equally to this work.
EXPERIMENTAL DETAILS
Chemical synthesis of Graphene Oxide (GO). GO was synthesized according to a modified Hummer’s method.
1
Natural graphite powder (1 g, TianJin Fu Chen Chemical
Reagents Factory, China) with an average particle size of 25 µm was added to a solution of H
2
SO
4
(34 mL) and NaNO
3
(0.75 g) and vigorously stirred in an ice-water bath for 20 min. KMnO
4
(5 g) was gradually added, and then the mixture was reacted for 2 h at room temperature. The reaction was terminated by addition of distilled water
(50 mL) and 30% H
2
O
2
(5 mL). The mixture was washed with 1:10 HCl/water and then deionized water until the pH of the suspension was between 6 and 7. The brown graphite oxide suspension was sonicated for 6 h, and then centrifuged at 2000 rpm for
20 min to afford a supernatant that was a stable exfoliated GO solution.
Fabrication of C
ZIF8-D and S/
CZIF8-D composite. C
ZIF8-D
was derived from one-step pyrolysis of ZIF-8, which was rapidly synthesized in aqueous system
2
. In a typical synthesis, 1.17 g of zinc nitrate (3.95 mmol) in 8 distilled water was added into a solution of 2-methylimidazole (22.70 g, 276.50 mmol) in 80 g DI water (1:70 molar ratio) and the mixture was stirred at room temperature for 5 min. The precipitate was subsequently collected by centrifugation, repeatedly washed with ethanol and finally dried at 70 o
C for 24h. The prepared ZIF-8 precursor was carbonized at 900 o
C for 3h under N
2
atmosphere to produce microporous carbon C
ZIF-8-D
. Then, elemental S and
C
ZIF8-D
(70:30 w/w) were mixed by mechanical ball milling at 300r/min for 5 h. The mixture was sealed in a polytetrafluoroethylene container and heated to 155 °C in an oven for 24 h under the protection of argon gas.
Assembly of hierarchical sandwich-type GS-S/C
ZIF8-D composites. S/C
ZIF8-D composite (1 g) was dispersed in water/ethanol (1:1, 100 mL) and then sonicated for 30 min. Exfoliated GO solution (3 mg/mL) was added to give a GO: S/C
ZIF8-D weight ratio of 1:9. The mixture was ultrasonically treated for 1 h. The resulting homogeneous black solution was heated in an oil bath at 95 °C under magnetic stirring for 48 h. The final product was collected by vacuum filtration with a membrane filter (47 mm in diameter,
0.2 µm pore size) and dried in a vacuum oven at 60 °C for 24 h.
Material characterization. X-ray diffraction measurements were performed on individual sulfur cathode components and the as-prepared cathode itself using a diffractometer (Rigaku) with a Cu Kα radiation source (λ=0.154 nm) at a scan rate of
2 ° min -1 . Raman spectra were measured on a Raman spectrometer (Renishaw-1000) with an excitation laser beam wavelength of 532 nm. XPS measurements were collected at room temperature with PHI-1600 ESCA spectrometer and monochromatic
AlKa (1486.6 eV) X-ray source. The morphology of the composites was determined by scanning electron microscopy (FEI Quanta 250) and high-resolution transmission
electron microscopy (JEOL-2010, 200 kV). Elemental analysis was performed by using an Elementar Vario Miro Cube (Germany).
Porosity of ZIF8 derived microporous carbon was measured by N
2
sorption isotherm at 77 K on a Micromeritics ASAP 2020 instrument. Samples were outgassed in vacuum (~5μbar) for at least 5 hours at 453 K before starting the sorption measurements. The surface areas were estimated using the
Brunauer–Emmett–Teller (BET) equation for the relative pressure range (P/P o
) of
0.002 to 0.3. The saturation pressure, P o
, corresponds to ca. 103.4 kPa. The t
HJ
-plot micropore volume is the calculated total pore volume with dimensions < 20 Å, using the Harkins and Jura method. Non-local density functional theory (NLDFT) calculation, based on the carbon slit pores geometry, was used to generate data on the pore size distribution of ZIF8 derived microporous carbon.
Electrochemical Measurements. GS-S/C
ZIF8-D
composites and S/C
ZIF8-D
composite cathode slurries were produced by mixing 70% composite, 20% carbon black, and 10% polyvinylidene fluoride binder in N-methyl-2-pyrrolidinone. The mixtures were ball milled for 4 h to form homogeneous slurries. After stirring, each slurry was coated onto aluminum foil using a roll press. The coated electrodes were dried in a vacuum oven at
60 °C for 24 h. The electrodes were cut into disks with a diameter of 11 mm.
Two-electrode coin cells (CR2025) with Li foil as the counter electrode were assembled in an argon-filled glovebox for electrochemical experiments. The electrolyte used was 1.0 M bis-(trifluoromethane)sulfonimide lithium in a 1:1 v/v mixture of dimethoxyethaneand 1,3-dioxolane. The cells were discharged and charged from
1.0–3.0 V at current densities of S at room temperature using an electrochemical station
(LAND, Wuhan) to test their cycle life.
Supporting Data
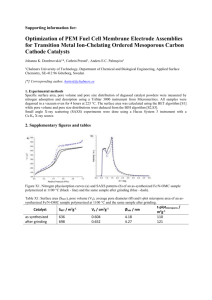
5-10 layers
Fig. S1 HR-TEM image of fringes of graphene
ZIF-8 Nano crystal
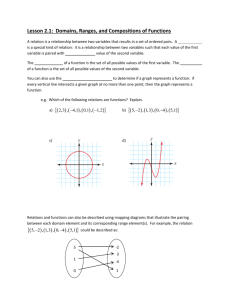
10 20 30
2 deg
40 50 60
Fig. S2. XRD pattern of the ZIF-8 nano crystals.
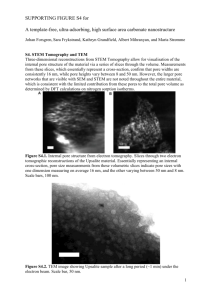
500
(a)
5.0
(b)
2.5
250
0.0
0
0.0
0.2
0.4
0.6
0.8
Relative pressure
(
P/P0
)
1.0
6 10 20 40 60 100
Pore size (A)
200 400 800
Fig S3. (a) N
2
adsorption-desorption isotherms of the C
ZIF8-D
(adsorption: ●; desorption:
○); (b) the pore size distribution calculated from the desorption branches.
Table S1 Textural characteristics of porous carbon from ZIF-8
C
ZIF8-D
BET surface area
(m 2 g -1 )
969
Total pore volume*
(cm 3 g -1 )
0.40
Micropore Mesopore Macropore volume
(cm 3 g -1 ) volume
(cm 3 g -1 ) volume
(cm 3 g -1 )
0.39 0 0
Surface area of ZIF-8
(m 2 g -1 )
1630 3
Note: * Pores with dimensions up to 999 Å by NLDFT, assuming carbon slit pore geometry.
Table S2. Percentage composition (by weight) of the elements in the various composites
Composite
Elements
S C O
GS-S/C
ZIF8 -D
composite 54.96 39.92 5.12
S/C
ZIF8 -D
composite 65.88 34.12 -
1 Hummers, W. S.; Offeman, R. E. J. Am. Chem. Soc . 1958 , 80 , 1339–1339.
2 Pan, Y.; Liu Y.; Zeng G.; Zhao, L.; Lai, Z. Chem. Commun. 2011 , 47 , 2071-2073.
3
K. S. Park, Z. Ni, A. P. Cote, J. Y. Choi, R. D. Huang, F. J. Uribe-Romo, H. K. Chae, M. O'Keeffe and O. M. Yaghi, Proc. Natl.
Acad. Sci. U. S. A.
, 2006, 103 , 10186-10191.