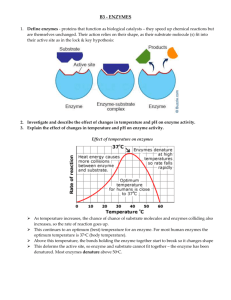
enzymes
advertisement

Enzyme notes Inhibitors Inhibitors are chemicals which reduce the rate of an enzyme-catalysed reaction- they do so by altering the shape of the active site. There are various categories of inhibitor, They can be classified into reversible and non-reversible (permanent). Reversible inhibitors only bind temporarily to an enzyme- so their effect is non permanent; Whereas non-reversible bind permanently to the enzyme leaving it unable to carry out further catalysis. Reversible inhibitors can also be competitive or non-competitive . Competitive inhibitors have a molecular shape which is similar to the substrate. They can fit into the active site of the enzyme, preventing the substrate from occupying it and reducing the number of enzyme-substrate complexes. They are sometimes referred to as active-site directed inhibitors. Therefore they compete for the active site with the substrate, but this is only temporary. The degree of inhibition depends on the concentration of the substrate and inhibitor. When you increase the concentration of the substrate this reduces the effect of the competitive inhibitor. Non-Competitive inhibitors do not become attached to the active site, but bind to other parts of the enzyme, altering the overall shape of the enzyme molecule, incl the active site. These are also called non-active site directed inhibitors. Competitive inhibitors are ALWAYS reversible Non- competitive inhibitors may be reversible or permanent Non-reversible inhibitors are always NON-COMPETITIVE- they leave the enzyme permanently damaged. De-naturation can occur due to non-reversible inhibitors. An e.g of a non-reversible enzyme inhibitor is the ion cyanide (CN-). This toxic substance combines with enzymes involved in cellular respiration, preventing energy production by cells, rapidly resulting in death. (Also see diagram pg 110) Commerci al use of Michelle Murphy 13~46 Biology AS notes enzymes Enzymes synthesised by bacterial and fungal cells have been used for years in baking and brewing. However the process of catalysis by enzymes within whole cells is inefficient, because the products may be used up by the cells, or contaminated with cells debris any by-products. Enzymes separated from the cells which produce them are more useful as they retain their catalytic powers while giving greater efficiency of substrate conversion and greater yield of product. Enzymes have large catalysis powers when compared with inorganic catalysts and they catalyse their reactions in aqueous (water) solutions at low temps. The reactions which they catalyse are specific. Uses of Enzymes Food manufacture Production of pharmaceuticals and chemicals -See table on pg 108 for table of commercial usesEnymes in detergents Biological detergents contain a number of enzymes which digest protein based stains such as blood, gravy, grass and starchy stains. They can work with temps of 40oc, which saves energy and prevents damage to fabrics. Addition of a cellulase to a washing powder breaks down micorfibrils that form on cotton during wear, this enhances the colour of washed clothes. Pectinases and cellulases in food + drink manufacture The primary cell wall of plant cells consists of cellulose but contains small amounts of other polysaccharides as hemicelluloses and pectin. Hemicelluloses like cellulose are composed of beta glucose but with side chains of other sugars. Pectins are made of galactose and galacturonic acid which forms a salt with calcium ionscalcium pectate. This pectate attracts water molecules forming a gel which acts as cement between the cellulose microfibrils bonded to the cellulose by the hemicellulose molecules. The ability of pectin to form a gel is used in the manufacture of jam. A group of enzymes called pectinases will break down pectin into its monosaccharide building blocks. Suspecting are used to clarify (clear) wine and vinegar and to prevent conc. Juices forming gels. Also celluloses break down cellulose to shorter chains of sugar and beta glucose.. They can clarify orange juice. Cellulases are used to break down cellulose in rye grass and straw to produce animal feed. Enzymes as analytical reagents Enzymes can be used to identify one type of molecule from a mixture of different types. They are very sensitive and can be used as analytical reagents, measuring the level of substances even when they are present in very low concentrations. Urea assay The enzyme urease has been used for analysing body fluids for the waster product urea. Urease hydrolyses the urea in the blood/urea sample into carbon dioxide and ammonia. The ammonia formed can be measured by the colour change it produces when reacted with an Michelle Murphy 13~46 Biology AS notes indicator. Glucose assay Two enzymes can be used for determining the concentration of glucose in blood or urine. Glucose oxidase catalyses the oxidation of glucose to gluconic acid and hydrogen peroxide. The hydrogen peroxide then oxidises a colourless chromogen to its coloured form by the removal of hydrogen. A chromogen is a substance which develops a colour. The second reaction is catalysed by another enzyme called peroxidase. The amount of the coloured substance produced is a direct measure of the concentration of the glucose present at the start and can be measured in a colorimeter. This is often used by people with diabetes to test their blood for glucose. Immobilisation of enzymes Enzymes which are isolated from the cells or tissues are much more useful if they are not in solution but instead immobolisied by being attached to or trapped within an an insoluble material. Advantages of dissolved enzymes: The enzymes are more stable at high temps They are more resistant to changes in pH They re less likely to be degraded by organic solvents The products are uncontaminated by enzymes and can be collected more easily The enzyme can be retained and reused Use of columns of immobilised enzymes allows automation of industrial processes Such processes often need high temps and extremes of pH and organic solvents to work efficiently. Costs can be minimised if the enzyme is not lost with the product but trapped in the immobilised form to be used for further reactions. 5 basic methods of immobilisation. Absorption onto a material such as porous gas Covalent bonding to a solid as cellulose Cross-linking between enzyme molecules using reagents such as glutaradehyde Entrapment within the internal structure of a polymer such as collagen or alginate Encapsulation within a selectively permeable membrane such as nylon (SEE DIAGRAMS PAGE 112) Advantages + Disadvantages Name of property Advantages Disadvantages Covalent bonding Commonly used Expensive- the enzyme forms stable cov. Links with the supporting matrix but it must be ensured that the active site on each molecule is exposed. Entrapment Commonly used. Since the enzyme is trapped, rather than bound its catalytic properties are not affected. Absorption Simple and cheap- the enzyme Not efficient. Michelle Murphy 13~46 Biology AS notes can be washed from the absorptive material An example of commercially used immobilisation enzyme is lastase, it is used to hydrolyse the sugar lactose present in glucose and galactose. Immobilised lasctase can be used to produce lactose-reduced milk and milk products for those who are lactose intolerant. (FOR OVERALL SUMMARY SEE PAGE 113) Michelle Murphy 13~46 Biology AS notes