Percent NH4Cl in Mixture
advertisement

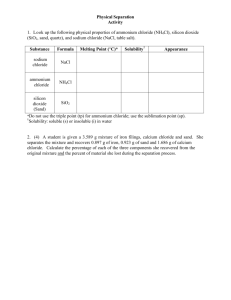
1 Determining the Percent Composition of Ammonium Chloride in a Mixture PRE-LAB ASSIGNMENTS: To be assigned by your lab instructor. CONCEPTS TO BE LEARNED IN THIS LAB: By the end of this lab, the student should understand what it means to know the percent composition of a mixture. The students should understand the concept of determining the mass of a substance by taking the difference of two other masses. Learn how to use physical properties to separate the components of a mixture. EXPERIMENTAL GOALS: The purpose of this procedure is to determine the percent composition of an unknown mixture that contains ammonium chloride, NH4Cl, and sand, (SiO2)n. INTRODUCTION: This lab is the first of three procedures which are designed to introduce the concept of reaction stoichiometry. In this procedure, the percent composition of a mixture of ammonium chloride and sand will be determined from the physical properties of the components of the mixture. Most solid substances, when heated, undergo a phase transition from the solid phase to the liquid phase, a process known as melting. However, some substances go from the solid phase directly to the gas phase, a process known as sublimation. (A familiar example is frozen carbon dioxide, or dry ice. If you put a chunk of dry ice on a tabletop, the dry ice does not melt, but the chunk gets smaller and smaller as the particles in the solid warm up and go directly into the gas phase.) Ammonium chloride sublimes when heated, which gives us a convenient way to separate ammonium chloride from sand. By knowing the weight of the sample before and after heating, the mass of the ammonium chloride in the sample can be determined. Once the weights of each component are known, the percent composition of the mixture is determined by dividing the weight of each component by the total weight of the sample. 2 Figure 1. Apparatus for the sublimation of ammonium chloride. PROCEDURE: 1. Obtain a sample of an unknown mixture of ammonium chloride and sand from the stockroom and record the sample number on the lab report (1). 2. Clean an evaporating dish, warm it over a Bunsen burner to dry, allow it to cool, and weigh it to the accuracy of the balance (3). Handle the clean dish with tongs, not your hands. Add between four and five grams of your unknown mixture to the dish, and weigh it again (2). 3. Place the evaporating dish containing the mixture on a wire gauze held by an iron ring in the fume hood (see Figure 1.) Heat carefully at first to avoid any spattering, and increase the heat after a couple of minutes. After heating strongly for 10 minutes, remove the flame and stir the contents with a glass stirring rod. Heat the sample again for another 10 minutes, or until no more white fumes are observed. Using your crucible tongs, carefully move the hot evaporating dish to a wire gauze or a clay triangle bent into the shape of a tripod, and allow the dish and its contents to cool in the hood. Do NOT put the hot dish down directly on the floor of the hood or the lab bench, because when the dish cools off, it may stick. 4. When the dish has cooled to room temperature, weigh the dish and remaining material (5). CALCULATIONS: 1. Determine the weight of the unknown mixture (4), the ammonium chloride (6), and the sand (7). 2. Calculate the percentage of each component in the mixture (8) using the formula weight % C grams C 100 total grams sample 3 LAB REPORT Determining the Percent Composition of Ammonium Chloride in a Mixture Name ________________________________ Date _________ Partner ________________________________ Section _________ Report Grade ______ 1. Unknown number ________________ 2. Mass of unknown mixture and container __________ 3. Mass of empty container __________ 4. Mass of unknown mixture __________ 5. Mass of sand and container after sublimation __________ 6. Mass of ammonium chloride __________ 7. Mass of sand __________ 8. Percent composition of sample: % ammonium chloride __________ CALCULATIONS: Mass of ammonium chloride: Mass of sand: % ammonium chloride % sand % sand __________