Counting particles 2010 April 26
advertisement

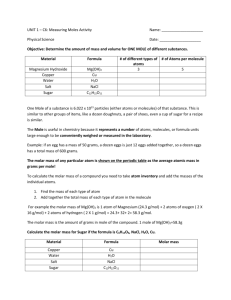
What is the mass? How many moles? How many?Circle your answers 1. How many aluminum atoms are found in a 10.00g aluminum can? This sample contains more than 6.02 x 1023 atoms / less than 6.02 x 1023 atoms / 6.02 x 1023 silicon atoms. 2. What is the mass of 1.00 x 1012 ( 1 trillion ) atoms of gold? The mass of the sample is greater than 197.0g / less than 197.0 g/ equal to 197.0g 3. How many lithium atoms are in 6.941 gram sample of Lithium? This sample contains more than 6.02 x 1023 atoms / less than 6.02 x 1023 atoms / 6.02 x 1023 lithium atoms. 4. How many moles of titanium (Ti) atoms are found in a 100 g sample of titanium? This sample contains more than 1 mole of Ti atoms / less than 1 mole of Ti atoms / 1 mole of Ti atoms 5. How many moles of Lead (Pb) atoms are found in a sample containing 8.03 x 1023 atoms of lead? This sample contains more than 1 mole of atoms / less than 1 mole of atoms / 1 mole of atoms 6. What is the mass of .875 moles of iron atoms? The mass of the sample is greater than 55.8g / less than 55.8 g / equal to 55.8 g Factor label conversions – Circle the factor that can be used to answer the question and calculate your answer 7. How many moles of aluminum atoms are found in a 10 g sample of aluminum? 10g Al ( 1 mole of Al ) (27.0g Al ) (27.0 g Al ) ( 1 mole of Al ) (6.02 x 1023Al) ( 1 mole of Al) ___________ ( 1 mole of Al_) (6.02 x 1023Al) (6.02 x 1023 Al) (27.0g Si ) ( 1 mole of Na) (6.02 x 1023 Na) (6.02 x 1023 Na ) ( 23.0 g Na ) (23.0 g Na ) (6.02 x 1023Na ) ( 1 mole of Hg__) (6.02 x 1023 Hg) (6.02 x 1023 Hg ) (200.6 g Hg ) (200.6 g Hg___ ) (6.02 x 1023 Hg ) (27.0 g Al ) (6.02 x 1023 Al) 8. What is the mass of .875 mole of sodium? _______________ .875 mole ( 1 mole of (23.0 g Na Na ) ) (23.0 g Na ) ( 1 mole of Na ) (6.02 x 1023Na ) ( 1 mole of Na) 9. How many mercury atoms are in 100.0 gram sample of mercury? _________ 100.0 g Hg ( 1 mole of Hg ) (200.6 g Hg ) (200.6 g Hg ) (6.02 x 1023Hg) ( 1 mole of Hg ) ( 1 mole of Hg) 10. How many titanium (Ti ) atoms are found in a 100 g sample of titanium?___________ 100g Ti ( 1 mole of Ti ) ( 47.9 g Ti ) ( 47.9 g Ti ) ( 1 mole of Ti ) (6.02 x 1023 Ti) ( 1 mole of Ti ) ( 1 mole of Ti__) (6.02 x 1023 Ti ) (6.02 x 1023 Ti ) (47.9 g Ti ) (47.9 g Ti ) 23 (6.02 x 10 Ti ) 11. How many moles of Osmium (Os) atoms are found in a sample containing 8.02 x 1023 atoms of osmium? ____________ 8.02 x 1023 Os ( 1 mole of Os ) (190.2 g Os ) (190.2 g Os ) ( 1 mole of Os ) (6.02 x 1023Os) ( 1 mole of Os ) ( 1 mole of_Os) (6.02 x 1023 Os ) (6.02 x 1023 Os ) ( 190.2 g Os ) ( 1 mole of Cu ) (6.02 x 1023 Cu ) (6.02 x 1023 Cu) ( 63.5 g Cu ) (63.5 g Cu ) (6.02 x 1023 Cu ) (190.2 g Os___) (6.02 x 1023 Os ) 12. What is the mass of 1.0 x 106 copper atoms? _____________ 1.0 x106 Cu ( 1 mole of Cu ) (63.5 g Cu ) (63.5 g Cu ) ( 1 mole of Cu ) (6.02 x 1023 Cu ) ( 1 mole of Cu ) What is the formula of Aluminium sulfide given the following calculations? 13. a) How many moles of aluminium are found in a compound containing 10.8 g Al? b) How many moles of sulfur are found in a compound containing 19.23 g S? c) How many times more sulfur atoms are found in aluminum sulfide than aluminium atoms? d) What is the formula of Aluminium sulfide? 14. Which sample of gold would you rather have? Why? a) 64.0 g gold (Au) b) 6.40 x 1012 gold (Au) c) 0.640 moles of gold (Au)