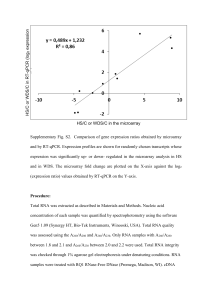
Supplementary Methods (doc 34K)
advertisement

Supplementary Methods Patients and Healthy donors Fresh and thawed samples from AML and ALL patients (Supplementary Table 1) were obtained with informed consent and stored at the HIMIP collection. According to French law, the HIMIP collection is registered with the Ministry of Higher Education and Research (DC 2008-307 collection 1) and a transfer agreement (AC 2008-129) was obtained after approval by the ethics committee Comité de Protection des Personnes Sud-Ouest et Outremer II. Clinical and biological annotation of the samples was declared to the National Committee on Data Processing and Liberties Comité National Informatique et Libertés. Bone marrow was obtained from healthy individuals after orthopedic trauma surgery. Blood samples were obtained from donors with their informed consent and provided by the Etablissement Français du Sang. CD3+, CD19+ and CD33+ cells were obtained with the Miltenyi® cell separation protocol (Miltenyi Biotech) by using CD3, CD19 and CD33 whole blood microbeads according to the manufacturer's instructions. Only samples with a 94% cell purity were selected. High-throughput quantitative PCR The Fluidigm BioMark System consists of a real-time PCR instrument using nanoliter volumes, Dynamic Array™ Integrated Fluidic Circuits (IFCs) for multiplexing the samples and reagents, and an IFC loader to load the Dynamic Array IFCs. Pre-amplification of cDNA from sorted controls and tumor samples was performed by pooling all primers (Supplementary Table 2) at a final concentration of 0.2µM. 2.47µl of TaqMan® PreAmp Master Mix kit (Applied Biosystems) and 1.23µl of primer mix were added to 1.3µl of cDNA (prepared as described in the “RNA preparation and cDNA synthesis” section, 5ng/µl) diluted at 1:100. Amplifications were carried out at 95°C for 10 min, followed by 14 PCR cycles at 95°C (15s) and 60 °C (4 min). Each pre-amplification mixture was diluted 1:5 in Tris-EDTA (TE) buffer. A ‘samples’ plate (BioMark®) was prepared by mixing in each well 3.73µl Taqman gene expression master mix, 0.37µl DNA binding dye sample loading reagent (PN 100-0388), 0.37µl Evagreen, 1.12µl TE and 1.9µl cDNA. A plate “assays” (BioMark®) was prepared by loading into each well a mixture containing 3.38µl assay loading reagent, 1.7µl TE and 1.9µl of each primer couple at 20µM. 5µl from each well in plate “samples” and plate “assays” was loaded onto the chip. Amplifications were carried out at 95°C for 10min, followed by 40 PCR cycles at 95°C (15s) and 60°C (4min) on the BioMark System. Data was analyzed as for qPCR using 5S rRNA as the housekeeping gene (GAPDH and ACTIN were also tested). Non-supervised clustering was realized using dChip software. Rows (corresponding to RNA of interest) were standardized by respective RNA means of expression and distant metric system applied is correlation. Sample clustering was carried out by the centroid method. The unpaired t-test was used for statistical analyses. Culture of APL blast Mononuclear cells from bone marrow or blood samples were prepared by Ficoll-Hypaque density gradient purification (PAA) and cultured in Iscove's Modified Dulbecco's Medium (Lonza) supplemented with 15% fetal calf serum, 1% penistreptomycine, 100ng/mL DNase (MP biotech), 10ng/mL human recombinant IL3 (R&D Systems), 10ng/mL human recombinant IL6 (R&D Systems) and 100ng/mL human recombinant SCF (Invitrogen) at 2 x 106/mL. APL patients cultured for in vitro differentiation assay or siRNA targeting of 14q(II-1) snoRNA were different from the original cohort of patients used for microarrays and highthroughput RT-qPCR since the original samples were no longer available. This therefore expanded the number of APL patients from eight to thirteen (all sharing the same phenotype i.e expressing the DLK1-DIO3 snoRNAs) and confirmed the specificity of the signature.