Solutions of nonelectrolytes are nonconductors
advertisement

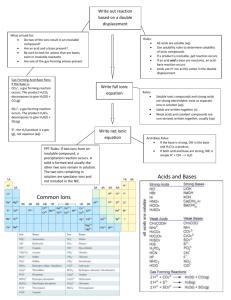
CHEMISTRY 4 CHAPTER 9 CHEMICAL CHANGE 1 ELECTROLYTES AND SOLUTION CONDUCTIVITY Strong electrolytes. Strong electrolyte is a substance whose solution is a good conductor. They are completely dissociated into ions. The solute is present entirely as ions. Weak electrolytes Weak electrolytes conduct electricity, but poorly. Most acids like acetic acid are weak electrolytes and are considered weak acids. Ammonia and its derivatives are weak electrolytes and are considered weak bases Nonelectrolytes. Solutions of nonelectrolytes are nonconductors: Sugar 2 SOLUTION OF IONIC COMPOUNDS When ionic compounds are dissolved in water, the ions become separated from one another by water molecules. The ions are not formed when an ionic solid dissolves but become free to move apart in the presence of water. Each ion has a number of water molecules closely associated with it. We say that each ion is hydrated. NaCl (s) Na+(aq.) NaNO3(s) Na+(aq.) MgCl2(s) Mg2+(aq.) + Cl-(aq.) + NO3-(aq.) + 2 Cl-(aq.) 3 STRONG AND WEAK ACID Hydrogen chloride HCl, which is a molecular compound, when dissolved in water forms chloride ions and hydronium ions. Solution of HCl in water is called hydrochloric acid. All molecules of HCl are converted to ions and we say that hydrochloric acid is a strong acid. HCl + H2O H3O+(aq.) + Cl-(aq.) To simplify sometime we write the dissociation of HCl as HCl H+(aq.) + Cl-(aq.) Only seven commonly encountered acids are strong acids: HCl, HBr, HI, HNO3, H2SO4, HClO4, HClO3. Most acids like acetic acid are weak electrolytes and are considered weak acids. A weak acid is only slightly ionized in solution. Because of the low concentration of ions, weak acids are poor conductors of electricity. 1 4 NET IONIC EQUATIONS Let us consider the full formula equation (conventional equation) of the reaction 2 NaCl(aq.) + Pb(NO3)2(aq.) 2 NaNO3 (aq.) + PbCl2(s) Let us replace the formulas of strong electrolytes by their ions. This produces the full ionic equation 2 Cl-(aq.) + 2Na+(aq.) + Pb2+(aq.) + 2NO3 -(aq.) + 2 Na+(aq.) + 2 NO3- (aq.) + PbCl2(s) Ions that are present at the scene of the reaction but experience no chemical changes are called spectators ions. Because spectator ions remain unchanged, we can remove them. This cancellation leaves the net ionic equation Pb+2 (aq.) + 2 Cl- (aq.) PbCl2(s) When writing a net ionic equation be sure that the equation is balanced in both atoms and charge. 5 SINGLE-REPLACEMENT OXIDATION-REDUCTION REACTION Let us consider the reaction between zinc and hydrochloric acid. The full formula equation is: ZnCl2(aq.) + H2(g) Zn(s) + 2 HCl(aq.) The net ionic equation is Zn(s) + 2 H+(aq.) Zn+2(aq.) + H2(g) In this reaction, electrons are transferred from zinc(s) to hydrogen ions (hydronium ions) Electron-transfer reaction is called oxidation-reduction (redox) reaction. The zinc atoms, which lost electrons, are said to have been oxidized, and hydrogen ions, the receivers of electrons , have been reduced. Let us consider the reaction between zinc and copper sulfate. The conventional equation is: Zn(s) + CuSO4 (aq.) ZnSO4(aq.) + Cu(s) The net ionic equation is Zn(s) + Cu+2(aq.) Zn+2 (aq.) + Cu(s) 2 To predict whether or not a redox reaction will take place we can use the following : activity series Li > K > Ba > Ca > Na > Mg > Al > Zn > Fe > Ni > Sn > Pb > H2> Cu > Ag > Au 6 OXIDATION-REDUCTION REACTIONS OF SOME ORGANIC COMPOUNDS When organic compounds react with oxygen to form water and carbon dioxide, we say they are oxidized. Electrons are transferred from organic compounds to oxygen. Example 1 Reaction of methane and oxygen CH4 (g) + 2O2(g) CO2(g) + 2H2O(l) Example 2 The burning of octane in gasoline 25O2(g) 16CO2(g) + 18H2O (g) + energy 2C8H18 (l) + Example 3 The burning of sugar in blood C6H12O6 (aq) + 6O2(g) 6CO2(g) + 6H2O (l) + energy 7 DOUBLE-REPLACEMENT PRECIPITATION REACTION The reaction occurs when the cation from one reactant combines with the anion from the another to form a precipitate. NaCl(aq.) + AgNO3(aq.) NaNO3 (aq.) + AgCl(s) Ag+ (aq.) + Cl- (aq.) AgCl(s) Solubility rules: The following compounds are insoluble: Chlorides, bromides, iodides of Ag+, Hg2+2, Pb+2 Sulfates of Ba+2, Sr+2, Ca+2, Pb+2 , Ag+, Hg2+2, Hg+2 Most carbonates, phosphates, hydroxides, sulfides except salt of alkali metals and NH4+ 8 DOUBLE-REPLACEMENT MOLECULE-FORMATION REACTION Water and weak acids are the most common kinds of molecular products of reaction Example: HCl(aq.) + NaOH(aq.) H2O(l) + NaCl(aq.) H+(aq.) + OH-(aq.) H2O(l) 3 + NaCH3 COO(aq.) HCl(aq.) HCH3COO-(aq.) CH3 COO-(aq.) H+(aq.) + + NaCl(aq.) HCH3COO-(aq.) 9 DOUBLE REPLACEMENT REACTION THAT FORM UNSTABLE PRODUCTS Ion combination may produce an unstable product that will further decompose into a gas Examples + CO3-2 (aq.) H2CO3 (aq.) CO2 (g) + H2O(l) 2 H+ (aq.) + SO3-2(aq.) H2SO3(aq.) SO2 (g) + H2O(l) 2 H+(aq.) NH4+ (aq.) + OH-(aq.) NH3 (aq.) + H2O(l) 10 DOUBLE REPLACEMENT REACTION WITH UNDISSOLVED REACTANTS Hydrochloric acid can dissolve aluminum hydroxide 3 HCl (aq.) + 3 H+(aq.) + Al(OH)3 (s) 3 H2O (l) + AlCl3 (aq.) Al(OH)3 (s) 3 H2O (l) + Al3+ (aq.) 4