HW10 Potential Energy Attachment
advertisement

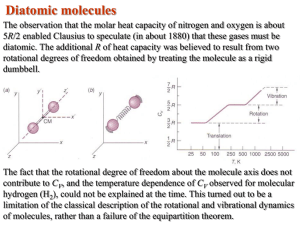
CM HW10: Potential Energy and Conservative Forces PE HW1. A particle is constrained to move in one dimension along the x axis and is acted upon by a force given by F(x) = −k/x3 i where k is a constant with units appropriate to the SI system. Find the potential energy function U(x), if U is arbitrarily defined to be zero at x = 2.0 m so that U(2.0 m) = 0. Ans. U(x) = −k/2x2 + k/8 PE HW 2. If U = 3x2 + 2xy + 4y2z, what is the force F? PE HW 3. A 75-gram Frisbee is thrown from a point 1.1-meter above the ground with a speed of 12 m/s. When it has reached a height of 2.1 m its speed is 10.5 m/s. What was the reduction in Emechanical of the Frisbee−Earth system because of air drag? 0.535 J PE HW 4. A 3.5 kg block is accelerated from rest by a compressed spring of spring constant 640 N/m over a frictionless surface. The block leaves the spring at the spring’s relaxed length and then travels over a wooden horizontal floor with a coefficient of kinetic friction µk, = 0.25. The frictional force stops the block in distance d = 7.8 meters. a. What is the increase in the thermal energy of the block−floor system? 67 J b. The maximum kinetic energy of the block? c. The original compression distance of the spring? 46 cm Potential Energy PE HW 5. The potential energy of the two atoms in a diatomic (two-atom) molecule can be written a b U ( r ) 6 12 , where r is the distance between the two atoms and a and b are r r positive constants. a. At what values of r is U(r) a minimum? A maximum? r = (2b/a)1/6 (min), r = 0 (max) b. At what values of r is U(r) = 0? c. Sketch U(r) as a function of r from r = 0 to r at a value large enough for all the features in (a) and (b) to show. d. Let F be the force one atom exerts on the other. Determine F as a function of r. e. For what value of r is F = 0 (the equilibrium position)? f. The binding energy of a two-particle system is defined as the energy required to separate the two particles from their state of lowest energy to r = . Determine the binding energy for this diatomic molecule. a2/4b