electronic supplementary material sediments, sec 2 • physical and
advertisement

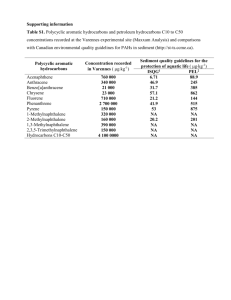
ELECTRONIC SUPPLEMENTARY MATERIAL SEDIMENTS, SEC 2 • PHYSICAL AND BIOGEOCHEMICAL PROCESSES • RESEARCH ARTICLE Investigating particle concentration effects of polycyclic aromatic hydrocarbon (PAH) sorption on sediment considering the freely dissolved concentrations of PAHs Jianwei Dong • Xinghui Xia • Yawei Zhai J. W. Dong • X. H. Xia () • Y. W. Zhai State Key Laboratory of Water Environment Simulation / School of Environment, Beijing Normal University, Beijing, 100875, P. R. China () Corresponding author: Xinghui Xia Tel.: +86 10 58805314 Fax: +86 10 58805314 e-mail: xiaxh@bnu.edu.cn 1 Contents 1 GC/MS analysis of PAHs References Tables Table S1 Physico-chemical properties of polycyclic aromatic hydrocarbons (PAHs) Table S2 Effects of SPS concentration on DOC concentration Figures Figure S1 The loss of d10-pyrene on LDPE strips vs time Figure S2 Dissolved pyrene concentration in water phase vs time during sorption experiments (sediment = 10 g/L) Figure S3 Sorption quantity of pyrene on Yellow River sediment under different SPS concentrations in the presence and absence of other PAHs Figure S4 Pyrene quantity bound with third phase for Yellow River sediment under different SPS concentrations in the presence and absence of other PAHs 2 1 GC/MS analysis of PAHs PAHs were analyzed by a gas chromatography-mass spectrometry (GC-MS) system equipped with an Agilent 5975 C mass-selective detector (MSD) (HP-5 MS, 5% phenyl methyl siloxane 35 m×0.25 mm diameter, and 0.25 μm film thickness). The injection port temperature was maintained at 280°C. Highly pure Helium (99.9999%) was chosen as the carrier gas, and kept at a constant flow rate of 1.0 ml min-1. Sample extracts (1.0 µl) were injected in the splitless mode. The GC oven temperature was held at 80°C for 2 min, then ramped at 15°C min-1 to 230°C and held for 2 min. The temperature was then programmed to increase to 290°C at 8°C min-1 and held for 2 min. Ionization was performed in electron impact (EI) mode. The ion source and quadrupole analyzer temperatures were fixed at 230 and 150°C, respectively. For identification of each target chemical, full scan mode from 35 to 550 amu was firstly performed and identification based on both retention time and characteristic ions. The quantification of each individual PAH was performed using the pre-established method in our previous work (Liu et al. 2010). Calculation of freely dissolved concentration of PAHs in water phase based on their concentrations and their deuterated compound concentrations in LDPE was referred to our previous study (Wang et al. 2011). 3 References Adams RG, Lohmann R, Fernandez LA, MacFarlane JK, Gschwend PM (2007) Polyethylene devices: Passive samplers for measuring dissolved hydrophobic organic compounds in aquatic environments. Environ Sci technol 41:1317-1323 Liu S, Xia X, Yang L, Shen M, Liu R (2010) Polycyclic aromatic hydrocarbons in urban soils of different land uses in Beijing, China: Distribution, sources and their correlation with the city's urbanization history. J Hazard Mater 177:1085-1092 Schwarzenbach RP, Gschwend PM, Imboden DM, Wiley J (2003) Environmental organic chemistry. 2nd ed. John Wiley & Sons, USA Wang F, Bu Q, Xia X, Shen M (2011) Contrasting effects of black carbon amendments on PAH bioaccumulation by Chironomus plumosus larvae in two distinct sediments: Role of water absorption and particle ingestion. Environ Pollut 159:1905-1913 4 Table S1 Physico-chemical properties of polycyclic aromatic hydrocarbons (PAHs) Molecular Mi ρi Tm Tb formula (g•mol-1) (g•cm-3) (°C ) (°C ) Phenanthrene C14H10 178.2 0.98 101.0 Pyrene C16H10 202.2 1.27 Chrysene C18H12 228.3 1.28 Compound name Log(Pi* Pa-1) -logCiwsat -logKiaw logKiow Log KPEW 339.0 -1.66 5.20 2.85 4.57 4.3 156.0 403.0 -3.09 6.16 3.32 5.13 5.0 255.0 448.0 -6.22 8.05 4.56 5.81 5.7 -1 Notes:Mi (molar mass), ρi (density), Tm (melting point), Tb (boiling point), Pi* (vapor pressure), CSat iW (aqueous solubility, mol L ), Kiaw (air-water partition constant), Kiow (octanol-water partition constant), Kia (acidity constant of some environmentally relevant organic chemicals density (20°C), all other data are given for 25°C), Sat fusG / RT CSat , (Schwarzenbach et al. 2003); Log KPEW from Adams et al. 2007 iW ( L) C W (S) e 5 Table S2 Effects of SPS concentration on DOC concentration DOC concentration (mg L-1) River sediment 2 g L-1 SPS 5 g L-1 SPS 10 g L-1 SPS Yellow River 1.343±0.031 1.383±0.043 1.695±0.001 Haihe River 1.770±0.021 2.027±0.064 2.400±0.082 6 The loss of d10-Pyrene on LDPE (%) 120 100 80 60 40 20 0 0 5 10 15 20 25 30 35 Time (d) The loss of d10-Pyrene on LDPE (%) 70 60 50 40 30 20 10 0 0 5 10 15 20 25 Time (h) Fig. S1 The loss of d10-pyrene on LDPE strips vs time 7 30 Pyrene concentration in water phase (µg/L) 1 0.8 0.6 0.4 0.2 0 0 100 200 300 400 500 600 700 800 Time (h) Fig. S2 Dissolved pyrene concentration in water phase vs time during sorption experiments (sediment = 10 g L-1) 8 Sorption quantity of pyrene on SPS (µg) 14 12 2 g L-1 in the absence of other PAHs 10 in the presence of other PAHs 8 6 4 2 0 0.1 0.2 0.5 1 2 5 10 5 10 5 10 Initial pyrene concentration (µg L -1 ) Sorption quantity of pyrene on SPS (µg) 14 12 5 g L-1 in the absence of other PAHs 10 in the presence of other PAHs 8 6 4 2 0 0.1 0.2 0.5 1 2 Initial pyrene concentration (µg L-1 ) Sorption quantity of pyrene on SPS (µg) 14 10 g L-1 12 in the absence of other PAHs 10 in the presence of other PAHs 8 6 4 2 0 0.1 0.2 0.5 1 2 Initial pyrene concentration (µg L-1 ) Fig. S3 Sorption quantity of pyrene on Yellow River sediment under different SPS concentrations in the presence and absence of other PAHs 9 Quantity bound with third phase (µg) 8 2 g L-1 7 in the absence of other PAHs 6 in the presence of other PAHs 5 4 3 2 1 0 0.1 0.2 0.5 1 2 5 10 5 10 5 10 Quantity bound with third phase (µg) Initial pyrene concentration (µg L -1 ) 8 7 5 g L-1 in the absence of other PAHs 6 in the presence of other PAHs 5 4 3 2 1 0 0.1 0.2 0.5 1 2 Quantity bound with third phase (µg) Initial pyrene concentration (µg L-1 ) 8 7 10 g L-1 in the absence of other PAHs 6 in the presence of other PAHs 5 4 3 2 1 0 0.1 0.2 0.5 1 2 Initial pyrene concentration (µg L-1 ) Fig. S4 Pyrene quantity bound with third phase for Yellow River sediment under different SPS concentrations in the presence and absence of other PAHs 10