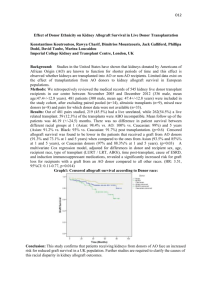
Receive report of suspected allograft
advertisement

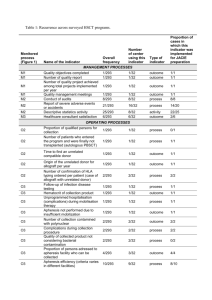
Begin: Receive report of suspected allograft-transmitted infection Adverse Reaction form/Complaint database entry (note: w/ every action rectangle this applies but this symbol is not repeated Enter into appropriate adverse reaction/complaint database and contact Responsible Persons May be assisted by QA personnel Communicated to Responsible Person of Regulatory Affairs/Quality Assurance Department Communicated to Medical Director In-house Donor Record Return Graft Authorization form Quarantine Graft form/database designation Review report information, assign eventtracking code, check for other complaints involving same donor, ensure any returned grafts are quarantined; quarantine inventory not yet distributed or released, verify w/reporting entity that complaint was received and investigation in process, review donor chart (A.) Review report information and in-house donor record (B.) Communicate with recipient’s physician (or reporting person) Request specific recipient information/medical records (C.) In-house Donor Record Other complaint report(s) exist(s) for this donor Review information received Determine product type affected by adverse report An allograft designated as a Biologic or a Medical Device (351 product) To insert on both sides: Medical Director Review (prior to reporting) A conventional tissue allograft (361 product or HCT/P) Delayed or no response to requests* Delayed or no response to requests* Determination whether or not this is a “possible” allograft-transmitted infection. (reasonable possibility) MUST be done w/in 15 days of initial report, regardless of delays* All known information is reviewed MedWatch entry/form. Determine Reporting Requirements MDR Scheme; International Communications** Determine Reporting Requirements International reporting if distributed there: Health Canada (see form); EU Directive – to competent authority of Member State Voluntary market withdrawal (or recall) may be instituted Determine if “probable” allograft transmitted infection MedWatch entry/form Yes Canada Vigilance Reporting Entry/Form (EUSTITE is developing a form); see Directive 2006/86/EC, Annex III Enter into appropriate adverse event database and initiate Responsible Person actions No Review decision history, produce summary report Action Items for Medical Director MedWatch entry/form Actions for Responsible Person of Regulatory Affairs/Quality Assurance Department **EU (MDV), Canada (TPP), Australia (TPA), Japan (ARR) Canada Vigilance Reporting Entry/Form Institute formal recall Immediately Contact Appropriate Agencies (D.) This should appear earlier Investigation by federal authority Inspect/test returned/quarantined grafts CAPA (Corrective and Preventive Action) In-process review/impact Communicate with original reporting entity Delayed or no response to requests Initiate Field Alert: Investigate recipients of other allografts from same donor Request specific recipient information/medical records and/or samples Test recipient samples Review decision history, produce summary report All known information is reviewed Determine if “proven” allograft transmitted infection Create and Issue response/report letters Close Report Close Report A. Pre-processing, inprocessing, postprocessing culture results. Reagent, equipment, and material records. Environmental monitoring test results D. Recovery Agency(ies), other Processing Establishment(s), federal authority (e.g., FDA, CDC), state authority (e.g., NYDOH, Florida AHCA), AATB (Accreditation Policies, part V), appropriate state/county health dept., International competent authority, if applicable** B. Donor: Medical history, clinical course, cause of death, autopsy, all cultures, lab work, trauma/physical assessment. Recipient: current clinical course, lab work/cultures, operative report C. Clinical course, symptoms, all cultures/tests, operative report, how graft culture was obtained at implant, viral testing history Glossary of Acronyms and Terms FDA = United States Food and Drug Administration CDC = Centers for Disease Control and Prevention NYDOH = New York State Department of Health Florida AHCA = State of Florida Agency for Health Care Administration AATB = American Association of Tissue Banks **EU Authorities (MDV – Medical Device Vigilance), Canada (TPP- Therapeutic Products Program), Australia (TGA – Therapeutic Goods Act), Japan (ARR – Adverse Reaction Reporting).