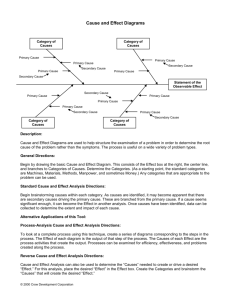
Educational objectives for Cheg 302, Separations/Equilibrium Stage
advertisement

Detailed Course Learning Outcomes
CHEG 200: Chemical Engineering Principles
Michael Hanyak, Lecture/Recitation/Laboratory
Professional Development
Value critical thinking by using a rigorous, problem solving methodology.
Value organization by keeping a course notebook during the semester.
Value documentation by keeping a laboratory notebook during the semester.
Value presentation by writing logical and concise homework solutions.
Value health and safety constraints by solving problems involving these issues.
Value self-learning by simulating a process flowsheet using the HYSYS software.
Value teamwork by participating in cooperative learning activities in the classroom.
Describe how your consulting company, called BEEF, Inc., solves complex problems.
Identify your job function as a sophomore chemical engineer in this company.
Classify all of the various job functions in this fictitious consulting company.
Problem-Solving Methodology
Identify the fundamental process operation in a well-defined problem.
Sketch a conceptual model that includes a diagram, givens, finds, and additional data.
Develop a mathematical model and list its assumptions and degrees of freedom.
Create a mathematical algorithm that satisfies the degrees of freedom.
Solve the mathematical algorithm to obtain the numerical answers.
Formulate heuristic observations by reviewing the answers, algorithm, and models.
Document formally the solution to the well-defined problem.
Apply above methodology to problems that involve material conservation, phase equilibrium, and/or
energy conservation.
Process Streams
Identify the process state of a multi-component stream or material;
that is, its temperature, pressure, flow rate (or amount), and composition.
Convert relative temperature or pressure to its absolute value and vice versa.
Express flow rate (or amount) in terms of mass, moles, and volume.
Express composition in terms of mass, mole, and volume fractions and
in terms of mass or molar concentrations.
Describe how mass and molar quantities are interrelated through molecular weight.
Describe how mass and volume quantities are interrelated through density.
Describe the interrelationship between mass and mole fractions.
Describe the interrelationship between mass and volume fractions.
Describe the interrelationship between mole fraction and molar concentration.
Describe the interrelationship between mass fraction and mass concentration.
Express mixture molecular weight in terms of component molecular weights.
Express mixture density in terms of pure-component densities or concentrations.
Classify above quantities as intensive and extensive properties.
Locate pure-component molecular weights in the literature.
Express density as a function of temperature, pressure, and composition.
Express the interrelationship between mass and molar density and specific volume.
Locate pure-component densities or specific gravities in the literature.
106728931
Page 1 of 5
7/24/2003
Detailed Course Learning Outcomes
CHEG 200: Chemical Engineering Principles
Michael Hanyak, Lecture/Recitation/Laboratory
Analyze problems that use the interrelation between flow rate (or amount) quantities and between
composition quantities.
Engineering Calculations
Identify the mks, fps, and cgs units commonly used for various quantities.
Convert units from one system to another system using conversion factors.
Determine the correct number of significant figures in a series of calculations.
Calculate the sample mean and standard deviation for a set of measured values.
Explain and use the concept of dimensional homogeneity for algebraic equations.
Use one-way and two-way linear interpolation on tabular data.
Plot experimental data either on regular, semi-log, or log-log paper.
Verify a math model with experimental data using a straight line fit by visual inspection; by the
method of least squares, and by using EXCEL.
Solve a set of linear algebraic equations using Gaussian elimination; using EXCEL.
Solve nonlinear equations using Newton's method, secant method, or successive substitution (direct
iteration).
Process Units
Describe typical process units such as mixer, splitter, reactors, and separators.
Classify their type as continuous, batch, or semi-batch (or semi-continuous).
Identify their operation as steady state or unsteady state.
Sketch their operation as a diagram and label the process state of all streams or materials.
Illustrate a process stream on a dry basis by two fictitious streams on the diagram (one for water and
one for the other chemical components).
Material Balances
Select an appropriate form for the material balances, either mass, mole, or atom.
Write general forms of the total material balance and component or atom balances.
Apply assumptions, wherever appropriate, to simplify these general forms.
Write mixture equations (i.e., total amount equals the sum of component amounts).
Select a linear independent set from the balances and mixture equations.
Write composition equations (i.e., component amount equals total amount times component mass or
molar composition).
Write conversion and/or excess equations for a main chemical reaction.
Write yield and/or selectivity equations for multiple reactions.
Write additional equations based on information given in a problem statement.
Develop mathematical models using the above balances and equations.
Analyze material balance problems for single and multiple process units.
Analyze material balance problems that involve recycles and purges.
Select a basis for an amount or flow rate and be able to do scaling when appropriate.
PVT Relationships
Sketch the PT and PVT diagrams for a typical pure component.
Identify the single phases and equilibrium phases in these diagrams.
Trace a multi-step process in both diagrams.
106728931
Page 2 of 5
7/24/2003
Detailed Course Learning Outcomes
CHEG 200: Chemical Engineering Principles
Michael Hanyak, Lecture/Recitation/Laboratory
Label each diagram with tabular data from the literature, such as melting point, boiling point, critical
point, saturated liquid and vapor molar or specific volumes.
Compare the tabular data in the steam tables to the PT and PVT diagrams.
Determine degrees of freedom for an equilibrium system using the Gibbs phase rule.
Apply the Antoine equation to both diagrams for vapor-liquid equilibrium.
Apply the ideal gas law to relate temperature, pressure, and molar volume.
Apply a cubic equation of state to relate temperature, pressure, and molar volume.
Use the theory of corresponding states and the compressibility factor charts.
Use property packages in HYSYS to compare and contrast equations of state.
Analyze material balance problems that incorporate PT and PVT relationships.
Vapor-Liquid Equilibrium
Sketch the TXY and PXY diagrams for a typical, binary vapor-liquid system.
Identify the single phases and equilibrium phases in these diagrams.
Trace a multi-step process in both diagrams.
Label each diagram with tabular data from the literature.
Apply the reverse-lever rule in the two-phase regions of these diagrams.
Sketch the TXY and PXY diagrams for a one-condensible system.
Describe vapor-liquid equilibrium using Raoult's Law.
Derive different math algorithms such as dew point and bubble point.
Use different math algorithms in HYSYS to solve phase equilibrium problems.
Analyze material balance problems that incorporate vapor-liquid equilibrium.
Energy Balances without chemical reaction
Select an appropriate form for the energy balance, either based on mass or mole.
Write the general form of the energy balance for either an open or closed system.
Apply assumptions like negligible changes in kinetic and potential energy, no reactions, adiabatic,
and no shaft work to simplify this general form.
Define the terms flow work and specific internal energy, volume, and enthalpy.
Write the specific enthalpy of any process stream (or material) in general form as a function of
temperature, pressure, and composition {i.e., H = hmix[T, P, comp]}.
Sketch the PH and PU diagrams for a typical pure component and choose its reference state.
Identify the single phases and equilibrium phases in these diagrams.
Trace a multi-step process in both diagrams.
Label each diagram with tabular data from the literature, such as melting point, boiling point, critical
point, saturated liquid and vapor molar or specific quantities.
Apply the reverse-lever rule in the two-phase regions of these diagrams.
Compare the tabular data in the steam tables to the PH and PU diagrams.
Analyze energy balance problems using a PH diagram or tabular PH or PU data.
Calculate the molar or specific enthalpy of multi-component process stream for an ideal solution (i.e.,
mixture is equal to sum of its pure components).
Select a reference state (T, P, and stable phase) for a pure component, and calculate sensible and
latent heat changes that go from the reference state to the state of the process stream.
Derive why the reference state of a pure component can be arbitrarily selected for non-reacting
system.
106728931
Page 3 of 5
7/24/2003
Detailed Course Learning Outcomes
CHEG 200: Chemical Engineering Principles
Michael Hanyak, Lecture/Recitation/Laboratory
Calculate the molar or specific enthalpy of a process stream for a non-ideal mixture.
Analyze material and energy balance problems that require the mixture enthalpies be calculated by a
set of equations.
Solve material and energy balance problems that require the use of an HXY diagram.
Energy Balances with chemical reaction
Select the reference state as the pure elements (i.e., atoms) at 25ºC, 1 atm, and stable phase of the
elements.
Calculate mixture enthalpies of process streams starting with this reference state; that is, the standard
heat of formation to create the compound from the elements.
Calculate the standard heat of a reaction and identify if it is endo- or exo-thermic.
Classify the operation of a reactor as isothermal, adiabatic, non-isothermal, or non-adiabatic.
Analyze energy balance problems using the heat-of-formation method (i.e., energy balance contains
no heat-of-reaction term and mixture enthalpy calculations begin with the standard heat of formation
of the components).
Analyze energy balance problems using the heat-of-reaction method (i.e., energy balance is written
with a heat-of-reaction term and mixture enthalpy calculations do not contain the standard heat of
formation of the components).
Derive the heat-of-reaction method starting with the heat-of-formation method.
Compare and contrast these two forms of the energy balance.
Chemical Process Simulation
Analyze the material and energy balances for individual process units like a process stream, pump,
cooler, mixer/tee, and a conversion reactor using HYSYS.
Analyze the material and energy balances for a process flowsheet using HYSYS by building the
flowsheet one process unit at a time starting with a reactor that converts methanol and toluene into
styrene monomer.
Do material and energy balance closures on the simulated process flowsheet.
Calculate the gross profit for the simulated process flowsheet to produce styrene monomer from
methanol and toluene.
Laboratory Work
Describe several ways to measure temperature and pressure.
Describe several ways to measure mass and volumetric flow rates (or amounts).
Describe several ways to measure composition of a multi-component mixture.
Operate several of these types of measuring instruments in the laboratory.
Construct steel and copper networks of piping, valves and fittings and pressure test each network.
Construct a network of compression tubing, valves and fittings, connect this network to a rotometer,
and produce a calibration curve for the flow measuring device.
Operate a membrane separator and do a material balance closure.
Operate a plate-and-frame filter press and do a material balance closure.
Operate a furnace on a spray dryer and do material and energy balance closures.
Operate a distillation column and do material and energy balance closures.
Prepare your laboratory notebook before each experiment, record experimental data in your lab
notebook, and do the necessary calculations in your lab notebook.
106728931
Page 4 of 5
7/24/2003
Detailed Course Learning Outcomes
CHEG 200: Chemical Engineering Principles
Michael Hanyak, Lecture/Recitation/Laboratory
Software Tools
Use HYSYS to begin a process simulation by opening a pre-defined simulation file.
Use HYSYS to build a fluid package and analyze an example process stream.
Use HYSYS to conduct a case study on an example heater and print the results.
Use HYSYS to create a reaction set and simulate an example conversion reactor.
Use HYSYS to manipulate an example process flow diagram (PFD) and print it.
Use HYSYS to recognize the significance of different enthalpy reference states.
Use WORD to prepare your project memo reports.
Use EXCEL to analyze your experimental data and graph your results.
Use an internet browser to locate information on chemical engineering data.
106728931
Page 5 of 5
7/24/2003