Nuclear objectives w definition
advertisement

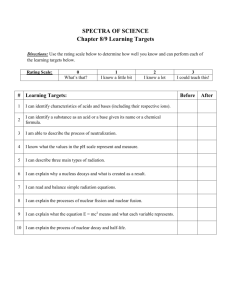
Chemistry Objectives Identify the two forces acting inside the nucleus and describe the role they play in radioactivity. Define radioactivity. List and describe the three naturally occurring forms of radioactive decay. Complete nuclear reactions. Describe the difference in mass from one side of an equation to the other and where that matter goes. Distinguish between nuclear fission and nuclear fusion. What is a chain reaction? What role do the neutrons play in the process? What does the term critical mass refer to in a chain reaction? Define the term ½ life as it relates to radioactive decay. Perform calculations related to whole ½ lives. Given ½ life, a period of time and an initial amount determine a final amount or given initial and final amounts determine the time that has passed. Given a diagram label the parts of a nuclear power plant and describe what each part does. Write a persuasive essay either pro nuclear energy or opposed to nuclear energy. o o o o What is the issue with waste? What is a spent fuel rod? What is nuclear proliferation? What is a moderator? Strong force: Nuclear force that holds the nucleus together. Weak Force: The nuclear force that pulls the nucleus apart and is responsible for radiation. Radioactivity: the process by which unstable nuclei emit or absorb particle in order to become stable nuclei. What is nuclear binding energy? The reduction in mass of a nuclei compared to the sum of its parts. Using e=mc2 you can calculate the energy that is holding the nucleus together. Alpha Decay: () Loss of a helium nucleus. Makes the nucleus lighter. Beta Decay (): Loss of an electron by the nucleus. Converts a neutron into a proton. Gamma Decay (): Light energy that accompanies radioactive decay. E=mc2 How does this equation relate to nuclear reactions? Fission: Splitting of a large nucleus into smaller ones. Fusion: Lighter nuclei coming together to make a larger one. Biological effects: What types of cells are most affected by radiation? Provide at least 2 examples. Rapidly reproducing cells that are common in reproductive organs and developing fetuses. Radiation has the ability to breaks bonds in DNA, why is that a unique and significant health risk? This can lead to chromosomal damage and possible birth defects. What is a rem? A Rad? How much energy the radiation has. A RBE? (Relative biological effectiveness) how dangerous is this type of radiation. What is a radioactive tracer and how is it used in medicine? How does it differ from radiation treatment? 1. 10.0g of a substance with a half-life of 2 years will decay into 1.25 g in how long? 2. A 48g sample is changed into 6 g in 8 minutes. What is its half-life? 3. A substance has a half-life of 3 years. If you have 36.0g how much will you have in 12 years? 4. You have 48g of a radioactive substance you need to deliver 6 grams If the ½ life is 10 minutes how much time do you have? 5. What fraction of a substance is left after 4 half lives?