Paper
advertisement

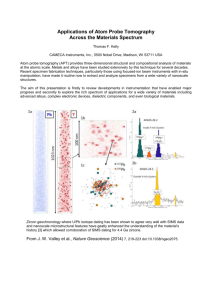
EFFECT OF Y ADDITION ON MG-ZN RAPIDLY SOLIDIFIED ALLOYS Xuefeng Guo*, and Dan Shechtman** * Department of Materials Science and Engineering, Xi'an University of Technology, Xi'an 710048, Shaan Xi, People Republic of China. ** Department of Materials Engineering, Technion, Haifa 32000, Israel 1. Introduction The development of new materials is usually driven by a need for improved properties. In recent years the need of weight reduction in the transportation and aircraft industries brought new-life to strong Mg alloys for use in structural applications [1-3], since magnesium alloys, the lightest structural materials [4, 5] have met many of the requirements. This includes high specific strengths [1-9], machinability [10, 11], good casting qualities [12] and high damping capacity [13]. It has been established that melt spinning is a convenient way to rapidly solidify (RS) alloys [14], and melt spun ribbons were used in many studies to investigate both the microstructure and mechanical properties of a wide variety of alloys [14]. RS enables to modify and make novel metastable microstructures, which can not be made by conventional casting. These microstructures are characterized by [14]: (1) Substantial extensions of the solid solubility, beyond equilibrium limits, (2) A more uniform microstructure, with less segregation and finer distribution of impurities and defects, (3) Formation of new metastable phases including crystalline, quasicrystalline or amorphous phases, and (4) Refined grain sizes. These microstructural changes provide opportunities to produce a wide variety of novel materials that exhibit improved mechanical properties and chemical stability. Using melt spinning to develop high-strength (ultimate tensile strength higher than 500 MPa [6]) magnesium alloys by microstructural refinement or even glass formation has been the focus of many research programmers [1-3, 5-9, 15-21]. Although a vast effort was made to develop other technologies [22-25], one of the best ways to get high mechanical properties and improved corrosion resistance is RS processing. Interesting achievements [6] are shown in Table 1-1. Strengthening 267 mechanisms including Orowan processes [26], deposition strengthening [26] , and fine-grain strengthening [6, 26] all contributing to the high strength, were used to explain the novel results obtained. Reviewing these studies, one can conclude that a future trend for development of Mg could be RS of alloys containing RE and Y [1-3, 5, 9, 11,15, 18-19,21,26-29] , since additions of RE and Y increase the mechanical properties both at room temperature and at elevated temperatures [4, 11], and simultaneously improve their corrosion resistance [15]. New magnesium alloys, such as Mg-Gd-Nd-Zr, Mg-Dy-Nd-Zr and Mg-Gd-YZr alloys [30], Mg-Sc[31], Mg-Sc-Mn[31] and Mg-Y-RE[11, 27-29] have been thoroughly studied, and in particular, Mg-Y-RE (WE series). Y and RE elements are considered the most useful additives to magnesium systems. Such alloys can work at temperatures above 250 oC [32-33]. Table 1-1. High strength magnesium alloys produced by of RS and extrusion Alloy composition, wt% s/ MPa Mg-18Al-10Ca b/ MPa /% E/ GPa 720 Mg-20Al-15Ca 600 Mg-21Y-10Cu 724 Mg-8Al-5Ga3Zn 518 646 1.2 Mg-9Al-6.5Ca 538 567 3.2 Mg-9Al-5Ca 496 565 Mg-9Al-1Zn1.5Si 468 541 Mg-6Zn-4Ce0.4Zr 710 3.4 506 2 Mg-5Al-5Zn5Nd EA55RS 476 513 5.0 164 2.21 330 48 1.91 168 1.78 1.78 1.82 46 124 (b/)/ MPam3Mg-1 400 50 46 455 1.81 (s/)/ MPam3Mg-1 330 5.4 Mg-11Si-4Al H/ kgmm-2 1.81 41 554 / Mg m-3 270 340 300 320 280 320 260 300 1.88 300 1.8 250 280 1.94 250 260 s tensile yield strength; b ultimate tensile strength; elongation to fracture; E Yong’s modulus; density; H hardness. 268 390 However, the effects of alloying additives of RE elements and Y in Mg-Zn base alloys, especially in the initial stages of the processing, i.e. solidification, remain unclear. It is important to understand the alloying process because Mg-Zn base alloys have excellent mechanical properties at room temperature, and when Y is added, also at elevated temperatures. In addition, thermal-shock and micro-shrinkage of Mg-Zn based alloys can be overcome by RS process. Therefore, the aim of this study is to optimize the performance of such alloying elements by developing a clear understanding of their effect on hardening and strengthening mechanisms. This article describes part of a study performed by the authors at the Technion during 2001-2002 when XG. was on Sabbatical at the Technion from Xian University. 2. Experimental 2.1 Chill Casting Based on the commercial alloys ZK31 and ZK60 [34] which contain 3%Zn (in weight per cent hereafter, unless indicated otherwise) and 6%Zn respectively, two groups of Mg-Zn based alloys containing different levels of Y, Ce and Zr were prepared by chill casting and melt spinning, as listed in Table 2-1. The raw materials for making the alloys were Mg ingots of 99.95% purity, Zn bars of 99.95% purity, and Mg47%Y, Mg-90%Ce and Mg-30 %Zr master alloys. Initial melting of billets, 700 gr each, were carried out in low carbon steel crucibles in a resistance furnace under protection of S6F and CO2 gas mixture, to avoid excessive oxidation. Melting temperatures were between 760 oC and 800 oC for the different alloys. Ingots were made by chill casting into copper molds 60 mm in diameter and 150 mm high. Both melting and casting were processed. Each BCI was then cut into several pieces, weighing ~70 g, each and remelted in carbon crucible covered by pre-heated salt-mixture of 50%KCl and 50%NaCl. The remelted alloys were made into small ingots (SCI) by chill casting into low carbon steel molds of 22 mm in diameter and 110 mm high. 269 Table 2-1 Experimental alloys Grou p Designat ion A1 I A2* A3* B1 B2 B3 B4 B5 II B6 Cast state CS RS CS CS CS RS CS RS CS RS CS RS CS RS CS RS CS B7 RS CS B8* RS B9* RS EDS analyzed composition at. % wt. % Mg-1Zn0.4Y Mg-2.7Zn1.3Y Mg-1.4Zn0.3Y Mg-3.6Zn1Y Mg-1.7Zn0.3Y0.14Zr Mg-4.5Zn1Y0.5Zr Mg-1.2Zn0.4Ce0.14Zr Mg-3Zn2Ce0.5Zr Mg-2.1Zn Mg-5.4Zn Mg-2.3Zn Mg-6Zn Mg-2.5Zn0.3Y Mg-6.2Zn1.3Y Mg-2.4Zn0.3Y Mg-6Zn1Y Mg-2.1Zn1Y Mg-5.4Zn3.5Y Mg-2.5Zn1Y Mg-6.3Zn3.2Y Mg-2Zn1.4Y Mg-5Zn4.7Y Mg-2.2Zn1.3Y Mg-5.4Zn4.3Y Mg-2.6Zn0.4Y0.2Ce Mg-6.3Zn1.5Y1Ce Mg-2.7Zn0.4Y0.2Ce Mg-6.5Zn1.4Y0.7Ce Mg-1.9Zn0.4Y0.2Ce Mg-4.8Zn1.5Y1Ce Mg-1.9Zn0.4Y0.2Ce Mg-4.8Zn1.5Y1Ce MgMg-5Zn1.3Y0.4Ce0.8Zr 2.0Zn0.4Y0.1Ce0.2Zr MgMg-5.3Zn1.5Y0.4Ce0.7Zr 2.1Zn0.4Y0.1Ce0.2Zr MgMg-5.5Zn0.7Y0.6Ce0.6Zr 2.1Zn0.2Y0.1Ce0.2Zr MgMg-6.0Zn1.0Y0.6Ce0.6Zr 2.2Zn0.3Y0.1Ce0.2Zr MgMg-5.5Zn1.7Y1Ce0.6Zr 2.2Zn0.5Y0.2Ce0.2Zr *Samples for mechanical property tests were made from those alloys, CS means chill casting, 2.2 RS Rapidly solidified ribbons, 3 mm wide and 80 μm thick were produced by single-roller melt spinning under low-pressure argon environment from SCI. Melt-spinning temperature was between 700 oC and 730 oC according to alloy composition. The estimated cooling rate was no less than 106 oC/s. 270 2.3 Extrusion Two extrusion procedures were made, of the RS, comminuted and canned ribbons and of the regular cast billets. In preparation for the extrusion of the RS alloys, ribbons were comminuted and canned in cylinders 30 mm in diameter. The canned powder was degassed in a vacuum chamber for 1 h, under ~ 10-5 Pa, heated to 150oC and pressed to form pre-extrusion bulk. The preextrusion bulk was then extruded at 300 oC via a die to form a 12 mm diameter round rod. The extrusion processed was performed at a constant strain rate of 0.5 min-1. Prior to extrusion the as-cast ingot it was machined to a cylinder 30 mm in diameter and 82 mm in length, and then extruded at 300 oC. at strain rate of 0.5 min-1. 2.4 Hear Treatment Heat treatments were performed on coupons cut from both extruded and as-cast bars. The coupons were encapsulated in quartz tubes that had been backfilled with high purity argon after evacuation. As-cast samples were heat-treated for 100 h at 300 3 oC, extruded samples were heat-treated for 1 h at 100 oC. The temperature was monitored by high precision equipment. 2.5 Microstructural Analysis The alloys at different stages were analyzed by XRD with CuKα radiation operating at 40 KV and 40 mA on a Philips X-ray diffractometer. For microstructural study, ribbon samples were mounted edgeon (along longitudinal cross section) in cold-setting resin. All the samples at different experiment stage were abraded to 1000 grit, polished to 1µm, etched with 5% of HNO3 in ethanol, observed with an optical microscope, then coated with carbon and analyzed by a SEM equipped with EDS. Transmission electron microscopy (TEM) specimens were made from cross-section of the ribbons. These were mounted in cold-setting resin within copper tubes. The tubes were then cut, polished to a thickness less than 0.08 mm, and ion beam thinned to final thickness on a Gatan-691 with beam energy 5.0 KeV, Characterization was performed in a Philips CM20 TEM operating at 200 kV. 271 In order to analyze critical phase transforming temperatures, specimens were tested on a Perkin-Elmer DTA 1700 high temperature differential thermal analyzer (DTA) system with heating rate of 15 oC min-1 under flowing argon. 3. Microstructure of RS Alloy Ribbons with Different Y Content Y addition to magnesium alloys is the most effective way to improve creep resistance and high strength at elevated temperatures thanks to fine and thermally stable Y containing particles [35] that can impede dislocation motion. In this study we prepared Mg-Zn alloys with different Y concentrations by melt spinning and evaluated their microstructures. Following is the microstructural study. 3.1 Solidification Microstructures Fig. 3-1 shows X-ray diffraction patterns of the ribbons. Except for two or three peaks at low angles which cannot be identified, A1 consists of supersaturated magnesium solid solution (Mg); B2 and B3 consist of (Mg) and W phase (Mg3Y2Zn3) and B4 consists of (Mg), W and X phase (Mg12YZn). Fig. 3-2 shows SEM images of the ribbons. The microstructure of A1 consists of characteristic columnar grains that originate at the wheel side and grow through the cross section of the ribbon. In addition, a discontinuous white layer was found at cell boundaries, as shown in Fig. 3-2 (a) and (b). EDS analysis reveals that the white layer contains higher Zn and Y than the average composition. In the second alloy group, the images show that the microstructures consist of Mg cells, submicron particles dispersed within them and cell boundary network. The (Mg) cells appear in two different layers along the cross section. One layer that originates at the wheel side consists of directionally solidified columnar cells reaching 10 to 20 m in size. The cells’ axes are not perpendicular to the quenching surface but are tilted some degrees away from it and are perpendicular to the solidification front. The other layer that consists of fine equiaxed grains about 5 m in diameter appears above the columnar grains. The size, shape and distribution of the submicron particles are similar in the second alloy group as shown in Fig. 3-2 (c) to (h). In B4, although X-ray 272 analysis (Fig. 3-1 (d)) indicate formation of X phase is difficult to be distinguished it from W phase by SEM and EDS perhaps because of its limited quantity. The network at grain boundaries is not the general eutectic mixture, but rather an intermetallic compound. 2500 * * (Mg) ? Unknown phase Intensity 2000 1500 (a) * 1000 * * 500 ? ** * * ? ? * * * * * * * * * * 0 20 40 60 80 * 100 * 120 2[deg.] 10000 * Intensity 8000 * (Mg) W Mg3Y2Zn3 ? Unknown phase (b) 6000 4000 2000 * ?W ? ? *? W 0 20 40 * * * ** * 60 * 80 * * * * * 100 2 [deg.] Fig. 3-1. X-ray diffraction patterns of alloys (a) A1; (b) B2 273 * 120 2000 1800 * 1600 1400 Intensity (c) * (Mg) W Mg3Y2Zn3 ? Unknown phase * 1200 1000 800 600 200 * * 400 * ?W ? ? W * W ** * * * * * * * * * * 0 20 40 60 80 100 120 2[deg.] 3000 (d) * 2500 * (Mg) W Mg3Y2Zn3 X Mg12YZn ? Unknown phase * Intensity X 2000 1500 * 1000 500 X W ? ? X W * X W * * W W W * ** * *W * * * * * * * * 0 20 40 60 80 100 2[ded.] Fig. 3-1. X-ray diffraction patterns of alloys (c) B3; (d) B4 274 120 (a) (b) Wheel cooling side Wheel cooling side (c) (d) Wheel cooling side (e) Free surface (f) Free surface (g) (h) Fig. 3-2. Microstructures of RS alloys (a) and (b) A1; (c) and (d) B2; (e) and (f) B3; and (g) and (h) B4 275 Fig. 3-3 shows DTA results. Each of the A1, B2 and B3 shows only one endothermic reaction, the melting of (Mg) matrix. B4 shows two endothermic peaks, the lower temperature peak is the melting of (Mg) + W + X eutectic mixture, and the higher temperature peak is the melting of (Mg) matrix. Fig. 3-3 DTA results of the RS ribbons (a) A1, (b) B2, (c) B3, and (d) B4 3.2 Solidification Process It has been demonstrated that the RS process consists of two steps [36-38]. First, rapid solidification takes place and coarse cells, composed of fine super-saturate dendrite branches from highly undercooled melt. This stage results in rapid latent heat release and temperature recalescence. During this stage only about 20% of the total melt solidified [38]. In the second stage, the residual melt undergoes relatively slower solidification, and the fine dendrite branches reheat and possibly remelt due to the solute redistribution and heat flux. The remelting temperature is determined by the local solidification time [38], solute redistribution and heat flux rate [38]. In the ribbons, the local solidification time is limited due to their high ratio of surface to volume. In the lower solute content A1, solute accumulation around dendrite 276 branch necks is not high enough to remelt the dendrite, therefore, coarse grains are formed during first step of solidification. In higher solute content B2, B3 and B4, on the other hand, the dendrite branches of the coarse grains are partly remelted and separated into small dendritic groups, these groups grow into the undercooled melt and eject solute to the solid/liquid front to form the final fine grains, mainly away from the wheel side (as shown in Fig. 3-2 (c) and (g)) or along the cross section of the ribbons (as shown in Fig. 3-2 (e)). From this point of view, the thickness of ribbon is also important. The thicker it is, the slower is the solidification rate, and the more serious is the remelt of the dendrite. As seen in Fig. 3-2 (e), where the microstructure consists of small grains along the whole section of the ribbon which is thicker than those in Fig. 3-2 (c) and (g). The white submicron particles and network are isolated compound rather than eutectic mixtures as shown in Fig. 3-4. According to the X-ray analysis results and equilibrium phase diagram [39], the compositions of the last solidification melts are located at (Mg) + W two-phase region in B2 and B3, and at (Mg) + W +X three-phase region in B4. However, during non-equilibrium solidification the last growing (Mg) grows on the primary (Mg) directly, resulting in the growing of isolated compounds at the last solidifying regions. (a) (b) Fig. 3-4. The compound morphologies of rapidly solidified (a) B3 and, (b) B4 3.3 Potential Effect of Dispersiods and Grain Boundary Network It is well known that refined grain microstructure of Mg alloys [40] can effectively improve their mechanical properties at room temperature. Refined grains, obtained by rapid solidification, is therefore a benefit. However, it is also known that elevated temperature strength 277 and creep resistance cannot be improved by refined grain structure because of the rapid diffusion [41] through interior of the grains and along grain boundaries. Therefore, the smaller the grain size, the greater the total grain-boundary area available for grain boundary diffusion [42] resulting in lower elevated temperature strength and creep resistance. High melting point compound dispersed uniformly within the magnesium solid solution and at grain boundaries is the best way to increase elevated temperature strength and creep resistance [35]. The increase in the volume fraction of particles within the magnesium benefits mechanical properties by impeding dislocation mobility. Moreover, these particles are thermally stable (as shown in Fig.3-3) due to their higher melting point (> 800 K) [39], and limited solubility in magnesium matrix [35]. High melting point compound network at grain boundary is broken up during the extrusion process and have no bad effect on the extruded material. 3.4 Y Contents Even with 1.3%Y content the B2 ribbon can get reasonable amount of Y containing compounds, as shown in Fig. 3-2 (c), due to two reasons. First, in the Mg-Zn-Y system the solubility of Y in matrix is limited as [42] at ambient temperature. During solidification, Y will be ejected onto the solid/liquid interface and, then accumulate around the necks of dendrite branches to form submicron compound and at cell boundaries to form a particle network. Second, the resulted compounds involve a large number of magnesium, or magnesium and zinc. On the other hand, higher Y content will result in low melting point eutectic mixture as indicated by the DTA results (Fig. 3-3 (d)) and X-ray diffraction patterns (Fig. 3-1 (d)) of B4. Based on our experimental results, we can conclude that when the alloy contains 5.5%Zn to 6%Zn, Y contents of less than 4.3% can be beneficial. Conclusion When the magnesium alloy with 5.5%Zn to 6%Zn contains also an addition of 1.3%Y-3.2%Y a reasonable amount of dispersed micro W particles are formed and improve properties. The microstructures of rapidly solidified Mg-Zn based alloys consist of cellular structure. The cell wall contains higher concentration 278 of alloying elements than the center of the cell, as expected. The ribbon solidifies under thermal diffusion controlled condition. For alloy B2, the evaluated dendrite growth rate is excess 2 10-3 m/s. Yttrium tends to segregate at grain boundaries and between dendrite arms in Mg-Zn-Y system during conventional solidification. Small amounts of Y additives to Mg-Zn base alloy is in solid solution if rapidly solidified. Using 1.3%Y-3.2%Y to alloy Mg-Zn base alloys can get reasonable amount of dispersed micro W particles when the alloys contain 5.5%Zn to 6%Zn. References 1. K. Kubota, M. Mabuch, K. Higashi, Review - Processing and mechanical properties of fine-grained magnesium alloys, Journal of Materials Science, 34 (1999) 2255 – 2262 2. B.L. Mordike, T. Ebert, Magnesium properties — applications — potential, Materials Science and Engineering, A302 (2001) 37–45 3. H. Jones, A perspective on the development of rapid solidification and nonequilibrium processing and its future, Materials Science and Engineering, A304–306 (2001) 11–19 4. M. M. Avedesian, H. Baker (eds.), Magnesium and magnesium alloys, The materials information society, Materials Park, OH, 1999 5. E. A. Ball and P. B. Prangnell, Tensile-compressive yield asymmetries high strength wrought magnesium alloys, Scripta metallurgica et Materialia, 31 (2), (1994) 111-116 6. C. Shaw, and H. Jones, Structure and mechanical properties of two Mg-Al-Ca alloys consolidated from atomised powder, Materials Science and Technology, 15, (1999) 78-84 7. T. Shibata, M. Kawanishi, J. Nagahora, A. Inoue and T. Masumoto, High specific strength of extruded Mg-Al-Ge alloys produced by rapid solidification processing, Materials Science and Engineering, A179–180 (1994) 632–636 8. T. Miyazaki, J. Kaneko and M. Sugamata, Structures and properties of rapidly solidified Mg-Ca based alloys, Materials Science and Engineering, A181–182 (1994) 1410–11414 9. C. Shaw, and H. Jones, The contributions of different alloying additions to hardening in rapidly solidified magnesium alloys, Materials Science and Engineering, A226–228 (1997) 856–860 279 10. R. S. Busk (ed.), Magnesium products design, Marcel Dekker Inc., New York, pp 57-84 11. C. Sanchez, G. Nussbaum, P. Azavant and H. Octor, Elevated temperature behavior of rapidly solidified magnesium alloys containing rare earths, Materials Science and Engineering, A221 (1996) 48-57 12. Yuan Guangyin, Sun yangshan, and Ding Wenjiang, Effects of Sb addition on the microstructure and mechanical properties of AZ91 magnesium alloy, Scripta mater., 43 (2000) 1009-1013 13. Z. Trojanova, P. Lukac, et al., Influence of composition and prestraining on the damping behavior of magnesium alloys and composites, in B. L. Mordike, K. U. Kainer, (eds.), Magnesium alloys and applications, Wolfsburg, WerkstoffInformationsgesellschat mbH, Hamburger Allee 26, 1998, pp241246 14. R. W. Cahn, Background to rapid solidification processing, in H. H. Liebmann, (ed.), Rapidly solidified alloys, Marcel Dekker, Inc., New York, 1993, pp 1-17 15. S. Krishnamurthy, E. Robertson and F. H. Froes, Rapidly solidified magnesium alloys containing rare earth additions, in H. Paris and W. H. Hunt (eds.), Advances in magnesium alloys and composites, The minerals, metals, and materials society, 1988, pp. 77-88 16. S. S. Cho, B. S. Chun, C. W. Kim, S. D. Kim, B. S. Lee, H. Baek and C. Suryanarayana, Structure and properties of rapidly solidified Mg-Al alloys, Journal of Materials Science, 34 (17) (1999) 43114320 17. A. Kato, H. Horikiri, A. Inoue and T. Masumoto, Microstructure and mechanical properties of bulk Mg70Ca10Al20 alloys produced by extrusion of atomized amorphous powders, Materials Science and Engineering, A179–180 (1994) 707–711 18. H. Iwasaki, T. Morri, and A. Tamura, Microstructure and mechanical properties of rapidly solidified magnesium alloys, Materials Science and Engineering, A179–180 (1994) 712–716 19. S. Krishnamurthy, M. Khobaib, E. Robertson and F. H. Froes, Corrosion behavior of rapidly solidified Mg-Nd and Mg-Y alloys, Materials Science and Engineering, 99 (1988) 507–511 20. J. A. Juarez-Islas, Rapidly solidification of Mg-Al-Zn-Si alloys, Materials Science and Engineering, A134 (1991) 1193–1196 280 21. F. hehmann, F. Sommer and B. Predel, Extension of solid solubility in magnesium by rapid solidification, Materials Science and Engineering, A125 (1990) 249–265 22. D. Dube´, M. Fiset, A. Couture, I. Nakatsugawa, Characterization and performance of laser melted AZ91D and AM60B, Materials Science and Engineering, A299 (2001) 38–45 23. H. Watanabe, H. Tsutsui, T. Mukai, M. Kohzu, S. Tanabe, K. Higashi, Deformation mechanism in a coarse-grained Mg-Al-Zn alloy at elevated temperatures, International Journal of Plasticity 17 (2001) 387-397 24. S. Spigarelli, M. Cabibbo, E. Evangelista, M. Talianker, V. Ezersky, Analysis of the creep behaviour of a thixoformed AZ91 magnesium alloy, Materials Science and Engineering, A289 (2000) 172–181 25. J.C. Gebelin, M. Suery, D. Favier, Characterisation of the rheological behaviour in the semi-solid state of grain-refined AZ91 magnesium alloys, Materials Science and Engineering, A272 (1999) 134–144 26. M. Mabuchi, K. Higashi, Strengthening mechanisms of Mg-Si alloys, Acta mater, 44 (11) (1996) 4611-4618 27. M. Sugamata, S. Hanawa, J. Kaneko, Structures and mechanical properties of rapidly solidified Mg-Y based alloys, Materials Science and Engineering, A226–228 (1997) 861–866 28. H. Horikiri, A. Kato, A. Inoue and T. Masumoto, New Mg-based amorphous alloys in Mg-Y-mish metal systems, Materials Science and Engineering, A179-A180 (1994) 702–706 29. F. Sommer, F. Hehmann, The transformation behavior of the rapidly solidified solid solution of yttrium in magnesium, Journal of the lesscommom metals, 159 (1990) 237-259 30. A. I. Anthony, Y. Kitaguchi, Y. Harima, et al., Aging characteristics and tensile properties of forged Mg-Gd-Y-Zr alloys, in E. Aghion and D. Elieser, (eds.), magnesium 97, Proceedings of the first Israeli international conference on magnesium science and technology, pp. 127-132 31. F. von Buch, J. Lietzau, B.L. Mordike, A. Pisch, R. Schmid-Fetzer, Development of Mg–Sc–Mn alloys, Materials Science and Engineering, A263 (1999) 1–7 281 32. J. F. Nie and B. C. Muddle, Characterisation of strengthening precipitation phases in a Mg-Y-Nd alloy, Acta mater., 48 (2000) 1691-1703 33. J. F. Nie and B.C. Muddle, Precipitation in magnesium alloy WE54 during isothermal ageing at 250 °C, Scripta Materialia, 40 (10), (1999)1089–1094J.F. Nie and B.C. Muddle, Precipitation in magnesium alloyWE54 during isothermal ageing at 250 °C, Scripta Materialia, 40 (10), (1999)1089–1094 34.. M. M. Avedesian, H. Baker (eds.), Magnesium and magnesium alloys, The materials information society, Materials Park, OH, 1999 35. J. F. Nie, B. C. Muddle, Precipitation in magnesium alloy WE54 during isothermal ageing at 250 oC, Scripta Materialia, 40 (10) (1999) 1089-1094 36. W. J. Boettinger, S. R. Coriell, R. Trivedi, in: R Mehrabian and P A Parrish, (eds.), Rapid Solidification Processing: Principle and Technologes IV, Claitor’s Publishing Division, Baton Rouge, LA, 1988, p13 37. R. Willnecker, D. M. Herlach, and B. Feuerbacher, Evidence of nonequilibrium processes in rapid solidification of undercooled metals. Phys Rev Lett 62 (5) (1989) 2707~2710 38. T. Z. Kattamis, M. C. Flemings, Dendrite structure and grain size of undercooled melts. Trans Met Soc AIME, 236(1966) 1523~1532 39. A. A. Байкоа, (ed.), magnesium alloys with yttrium, Science, Moscow, 1979, p 85 40. K. Kubota, M. Mabuchi, and K. Higashi, Processing and mechanical properties of fine-grained magnesium alloys, Journal of Materials Science, 34 (10) (1999) 2255-2262 41. H. Watanabe, T. Mukai, M. Kohzu, S. Tanabe and K. higashi, Effect of temperature and grain size on the dominant diffusion process for superplastic flow in an AZ61 magnesium alloy, Acta mater, 47 (14)(1999) 3753-3758 42. S. Krishnamurthy, E. Robertson, and F. H. Froes, Rapidly solidified magnesium alloys containing rare earth additions, in: H. Paris and W. H. Hunt (Eds.), Advances in magnesium alloys and composites, The minerals, metals, and materials society, 1988, pp. 77-88 282