TM2034 - Calgary Laboratory Services
advertisement

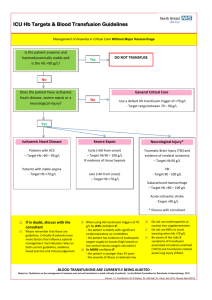
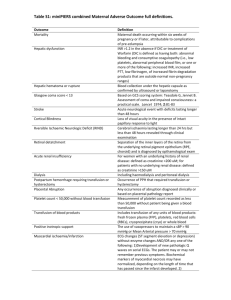
Adverse Reaction to Blood Transfusion or Transfusion Event Notification SITE: ACH FMC PLC RGH SHC Calgary Zone Rural Hospital (specify): Affix addressograph imprint or patient label, or clearly print patient’s full name (full surname, first name), date of birth, gender, Personal Health Number or Medical Record Number. Other (specify): Instructions for Nursing Unit staff: 1. Document all adverse reactions to blood components/products on the patient health record. 2. Report all adverse reactions to the attending physician for follow-up. 3. Perform clerical checks by comparing the patient’s identification with the transfusion tag. 4. Report all transfusion events to the attending physician and to transfusion medicine. Complete a form for each patient if more than one was involved. 5. For further information see www.calgarylabservices.ca. Implicated blood component/ product Clerical check: OK Red cells Plasma Platelets Other (specify) Discrepancy (describe): Volume Transfused: Unit identification /product lot number: Unit/product expiration date: Date Transfused: Time infusion started: Time infusions stopped/completed: Reaction date: Reaction time: Reported to TM: (date/time/technologist) Premedication: No Given under Anesthesia: Patient diagnosis: Yes Drug/Dose/Route: No Local/Regional Hematology/BMT Is the patient immune compromised? General Oncology Yes Medical Surgical Obstetrics/Gynecology Trauma Neonate No Clinical Symptoms (related to transfusion): No clinical signs/symptoms Nausea/vomiting Oliguria Diffuse Hemorrhage Chills/Rigors Dyspnea/Shortness of Breath Hemoglobinuria Jaundice Shock Temp: Pre C Post Pain:(specify)_ C Resp: Pre Rash Hypoxemia O2 sat: Urticaria (hives) Other (Specify) _ __________________ Post BP: Pre Post Pulse: Was the transfusion of red cells discontinued as a result of the reaction? Yes No Is a transfusion reaction investigation requested by the physician? (red cells only) Yes No Was the unit transfused to patient other than the intended recipient? Yes No Was an incorrect component/ product infused? (e.g. 5% instead of 25% albumin) Yes No Is TRALI (Transfusion Related Acute Lung Injury) suspected**? Yes No Is a serious reaction other than TRALI suspected or confirmed? Yes No Was this a febrile reaction? Yes No Pre _______ Post If the answer to ALL of these questions is NO, transfusion reaction investigation will NOT be performed. Complete and return this form to the Laboratory. If the answer to ANY of these questions is YES, a transfusion reaction investigation MUST be done. For reactions to red cells: For reactions to all other components/products: In SCM order: Transfusion RXN Invest Level 1. Collect a 6 mL mauve/lavender top tube from patient**. Label specimen with the patient’s name and number as well as the RTSIS number that the patient is currently wearing. Send the patient specimen and the unit container to the Laboratory with this form. Send the unit/product container to the laboratory with this form. **Contact TM to determine specimen requirements for TRALI and investigation of components /products other than red cells. Measures Taken: None Transfusion stopped Transfusion restarted Antipyretics Antibiotics Other (specify)_ ______ Nurse/physician reporting reaction: TM2034 20150827 Vasopressors Analgesics Antihistamines Steriods Diuretics – Effective? Supplementary O2 ICU required Chest X-ray Blood Culture Mechanical ventilation Duration Name of person completing report: