הודעה על החמרה ( מידע בטיחות) בעלון לצרכן
advertisement

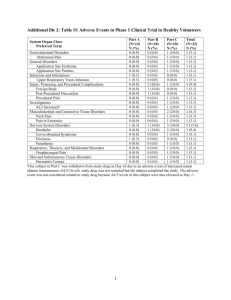
לרופא בעלון לרופא בטיחות) בעלון )מידע בטיחות החמרה (( מידע על החמרה הודעה על הודעה ___________3023 , אפריל32__ תאריך Foscavir :שם תכשיר באנגלית 8206260660 מספר רישום ____________ליברה בע"מ-פארם-או-שם בעל הרישום__טק ים/ים המבוקש/פרטים על השינוי טקסט חדש טקסט נוכחי Foscavir is indicated for induction Foscavir is indicated for induction and maintenance therapy of and maintenance therapy of cytomegalovirus (CMV) retinitis in cytomegalovirus (CMV) retinitis in patients with AIDS. Induction therapy patients with AIDS. Induction of mucocutaneous Herpes Simplex therapy of mucocutaneous Herpes Virus (HSV) infections, unresponsive Simplex Virus (HSV) infections, to acyclovir immunocompromised unresponsive to acyclovir patients. immunocompromised patients. Following induction therapy over 2-3 weeks Foscavir produced stabilisation of retinal lesions in approximately 80% of cases treated. However, since CMV causes latent infections and since Foscavir exerts a virustatic activity, relapses are likely in the majority of patients with persistent immunodeficiency once treatment is discontinued. Following completion of induction therapy, maintenance therapy should be instituted with a once daily regimen at an initial dose of 60 mg/kg increasing to 90-120 mg/kg if tolerated. A number of patients have received 90 mg/kg over a two hour period as a maintenance therapy starting dose. Maintenance therapy has produced a delay in time to retinitis progression. In patients experiencing progression of retinitis while receiving maintenance therapy or off Following induction therapy over 2-3 weeks Foscavir produced stabilisation of retinal lesions in approximately 80% of cases treated. However, since CMV causes latent infections and since Foscavir exerts a virustatic activity, relapses are likely in the majority of patients Therapeutic with persistent Indications immunodeficiency once treatment is discontinued. Following completion of induction therapy, maintenance therapy should be instituted with a once daily regimen at an initial dose of 60 mg/kg increasing to 90-120 mg/kg if tolerated. A number of patients have received 90 mg/kg over a two hour period as a maintenance therapy starting dose. Maintenance therapy has produced a delay in time to retinitis progression. In patients experiencing progression of retinitis while receiving פרק בעלון therapy, reinstitution of induction therapy has shown equal efficacy equivalent to that of the initial course. Foscavir is also indicated for the treatment of mucocutaneous HSV infections, clinically unresponsive to acyclovir in immunocompromised patients. The safety and efficacy of Foscavir for the treatment of other HSV infection (e.g. retinitis, encephalitis); congenital or neonatal disease; or HSV in immunocompetent individuals has not been established. The diagnosis of acyclovir unresponsiveness can be made either clinically by treatment with intravenous acyclovir (510mg/kg t.i.d) for 10 days without response or by in vitro testing. For treatment of acyclovir unresponsive mucocutaneous infections Foscavir was administered at 40mg/kg every 8 hours over 2-3 weeks or until healing. In a prospective randomised study in patients with AIDS, Foscavir treated patients healed within 11-25 days, had a complete relief of pain within 9 days and stopped shedding HSV virus within 7 days. Foscavir is not recommended for treatment of CMV infections other than retinitis or HSV or for use in non-AIDS or nonimmunocompromised patients. Since Foscavir can impair renal maintenance therapy or off therapy, reinstitution of induction therapy has shown equal efficacy equivalent to that of the initial course. Foscavir is also indicated for the treatment of mucocutaneous HSV infections, clinically unresponsive to acyclovir in immunocompromised patients. The safety and efficacy of Foscavir for the treatment of other HSV infection (e.g. retinitis, encephalitis); congenital or neonatal disease; or HSV in immunocompetent individuals has not been established. The diagnosis of acyclovir unresponsiveness can be made either clinically by treatment with intravenous acyclovir (510mg/kg t.i.d) for 10 days without response or by in vitro testing. For treatment of acyclovir unresponsive mucocutaneous infections Foscavir was administered at 40mg/kg every 8 hours over 2-3 weeks or until healing. In a prospective randomised study in patients with AIDS, Foscavir treated patients healed within 11-25 days, had a complete relief of pain within 9 days and stopped shedding HSV virus within 7 days. Foscavir is not recommended for treatment of CMV infections other than retinitis or HSV or for use in non-AIDS or non-immunocompromised patients. Since Foscavir can impair renal function, additive toxicity may occur when used in combination with other nephrotoxic drugs such as Interaction with other Medicinal Products and other function, additive toxicity may occur when used in combination with other nephrotoxic drugs such as aminoglycoside antibiotics, amphotericin B and cyclosporin A. Moreover, since Foscavir can reduce serum levels of ionised calcium, extreme caution is advised when used concurrently with other drugs known to influence serum calcium levels, like i.v. pentamidine. Renal impairment and symptomatic hypocalcaemia (Trousseau's and Chvostek's signs) have been observed during concurrent treatment with Foscavir and i.v. pentamidine. Abnormal renal function has been reported in connection with the use of foscarnet in combination with protease inhibitors associated with impaired renal function e.g. ritonavir and saquinavir. The elimination of Foscavir may be impaired by drugs which inhibit renal tubular secretion. There is no evidence of an increased myelotoxicity when foscarnet is used in combination with zidovudine (AZT). Neither is there any pharmacokinetic interaction between the two drugs. There is no pharmacokinetic interaction with ganciclovir, didanosine (ddI) or zalcitabine (ddC). The majority of patients who receive Foscavir are severely immunocompromised and suffering from serious viral infections. Patients’ physical status, the severity of the underlying disease, other infections and concurrent therapies contribute to adverse events observed during use of Foscavir. The following undesirable effects have been observed and reported during treatment with Foscavir with the following frequencies: Very common (≥1/10), common (≥1/100 to <1/10), uncommon (≥1/1,000 to <1/100), rare (≥1/10,000 to <1/1,000), very rare (<1/10,000) Clinical trial experience The adverse events and frequencies shown in Table 2 are based on the foscarnet primary clinical trial database. This includes adverse experiences reported at any time during induction, maintenance or follow-up treatment in 5 clinical trials involving 188 patients with CMV retinitis. aminoglycoside antibiotics, amphotericin B and cyclosporin A. Moreover, since Foscavir can reduce serum levels of ionised calcium, extreme caution is advised when used concurrently with other drugs known to influence serum calcium levels, like i.v. pentamidine. Renal impairment and symptomatic hypocalcaemia (Trousseau's and Chvostek's signs) have been observed during concurrent treatment with Foscavir and i.v. pentamidine. Abnormal renal function has been reported in connection with the use of foscarnet in combination with protease inhibitors associated with impaired renal function e.g. ritonavir and saquinavir. Forms of Interaction The elimination of Foscavir may be impaired by drugs which inhibit renal tubular secretion. There is no evidence of an increased myelotoxicity when foscarnet is used in combination with zidovudine (AZT). Neither is there any pharmacokinetic interaction between the two drugs. In different patient populations Foscavir has been administered to more than 11,500 patients, the majority severely immunocompromised and suffering from serious viral infections. The patient's physical status, the severity of the underlying disease, other infections and concurrent therapy also contribute to the observed adverse event profile of Foscavir. Consistent findings associated with Foscavir administration are renal function impairment, impact on serum electrolytes and haemoglobin concentration, convulsions and local genital irritation/ulceration. The adverse events discussed and tabulated below refer to results for 188 AIDS patients in prospective clinical trials and include those events related, unrelated and of unknown relationship to Foscavir, The adverse Undesirable Effects1 Adverse events are presented by event profile from the market is frequency and body System-Organ similar to that reported in clinical Class (SOC). In these clinical trials, studies. hydration and attention to electrolyte balance was not consistently given; the Renal function impairment: Twentyfrequency of some adverse events will seven percent of the above 188 study be lower when current patients experienced renal functional recommendations are followed (see impairment recorded as a rise in ‘Posology and method of serum creatinine (19%), decreases in administration’ and ‘Special warnings creatinine clearance (6%), abnormal and special precautions for use’). renal function (9%), acute renal Table 2 Frequency of adverse events failure (2%), uraemia (1%) and from the primary clinical trial database polyuria in 2%. Metabolic acidosis was seen in 1%. The overall pattern of SOC Frequency Event these symptoms is consistent with Blood and Very Granulocytop previous experiences although the lymphatic Common enia incidence may vary. Most patients system (≥10%) with increased serum creatinine have disorders shown normalisation or return to preCommon Leukopenia, treatment levels within 1-10 weeks of (≥1% and Thrombocyto treatment discontinuation. <10%) penia Metabolis m and nutrition disorders Very Common (≥10%) Common (≥1% and <10%) Nervous system disorders Uncommon (≥0.1% and <1%) Very Common (≥10%) Common (≥1% and <10%) Gastrointes tinal disorders Very Common (≥10%) Common (≥1% and <10%) Skin and subcutaneo us disorders Very Common (≥10%) Anorexia, Hypokalaemi a, Hypomagnesa emia, Hypocalcaemi a Hyperphosph ataemia, Hyponatraemi a, Hypophospha taemia, Blood alkaline phosphatase increased, Blood lactate dehydrogenas e increased Acidosisb Dizziness, Headache, Paraesthesia Coordination abnormal, Convulsion, Hypoaesthesi a, Muscle contractions involuntary, Neuropathy, Tremor Diarrhoea, Nausea, Vomiting Abdominal pain, Constipation, Dyspepsia Rash Electrolytes: Among the above 188 patients, hypocalcaemia was recorded in 14%. Also, hypomagnesaemia was recorded in 15%. Frequently recorded were also hypokalemia in 16% and hvpophosphataemia and hyperphosphataemia in 8 and 6% respectively. Foscarnet chelates with metal ions (Ca2+ Mg2+, Fe2+, Zn2+) and acute hypocalcaemia, sometimes symptomatic, has been a common observation in some 30% of AIDS patients receiving foscarnet. Experimental and clinical data have shown that foscarnet acutely decreases ionised calcium in a dose-related manner. The drop in serum calcium is reversible. It is reasonable to assume that the infusion rate significantly affects the decrease rate of ionised calcium. Convulsions: Among the AIDS patients referred to above, convulsions including grand mal were recorded in 10%, Based on the occurrence of convulsions among immunocompromised patients receiving foscarnet, an association between foscarnet induced hypocalcaemia or a direct action of foscarnet per se and convulsions has been discussed. Although many of the patients experiencing convulsions had pre-existing CNS abnormalities such as cryptococcal meningitis, space occupying lesions or other CNS tumours, an association with foscarnet can not be excluded. General disorders and administrat ion site conditions Investigati ons Very Common (≥10%) Asthenia, Chills, Fatigue, Pyrexia Common (≥1% and <10%) Very Common (≥10%) Malaise, Oedema Common (≥1% and <10%) Immune system disorders Psychiatric disorders Cardiac disorders Vascular disorders Hepatobiliary disorders Reproducti ve system and breast disorders Renal and urinary disorders a Common (≥1% and <10%) Common (≥1% and <10%) Common (≥1% and <10%) Common (≥1% and <10%) Common (≥1% and <10%) Common (≥1% and <10%) Common (≥1% and <10%) Blood creatinine increased, Haemoglobin decreased Creatinine renal clearance decreased, Electrocardio gram abnormala Sepsis Aggression, Agitation, Anxiety, Confusional state, Depression, Nervousness Palpitations Haemoglobin concentration: Decreases of the haemoglobin concentration have been observed in 25-33% of patients. Generally, there has been no consistent pattern of simultaneous decreases in white blood cell and platelet counts. Some 30% of the above study patients were also on concurrent AZT treatment. Many AIDS patients were anaemic already before foscarnet administration. Local irritation in terms of thrombophlebitis in peripheral veins following infusion of undiluted foscarnet solution and genital irritation/ulcerations have been observed. Since foscarnet is excreted in high concentrations in the urine local irritation/ulceration may ensue especially during induction therapy when high doses of foscarnet are being administered. Other Adverse Events: Other adverse events that were recorded in the 188 study patients include a variety of symptoms varying in frequency from 1% to approximately 60%, the latter being the incidence for fever. Subgrouped by body system the Hypertension, following adverse events, related, Hypotension, unrelated or of unknown relationship Thrombophle to foscarnet therapy were recorded. bitis Hepatic function abnormal, Gammaglutamyltrans ferase increased, Alanine aminotransfer ase increased, Aspartate aminotransfer ase increased Genital irritation and ulceration Renal impairment, Renal failure acute, Dysuria, Polyuria This frequency is based on 2 reports of electrocardiogram abnormal from 188 patients in the primary clinical trial database; the post-marketing reporting rate is ‘Very rare’. b This frequency is based on 1 report of acidosis from 188 patients in the Body as a whole: Asthenia, fatigue, malaise and chills were observed in 12, 20, 7 and 13% respectively and sepsis in 7%. Gastro-intestinal system disorders: Nausea and vomiting were observed in 45 and 25% respectively and diarrhoea in 32%. Abdominal pain and occasionally dyspepsia and constipation were observed in 10, 3 and 6% respectively. Isolated cases of pancreatitis have been reported from marketed use. Metabolic and nutritional disorders: Hyponatremia and oedema in legs were seen in 4 and 1% respectively and increase in LDH and alkaline phosphatases in 2 and 3% respectively. Increased levels of amylase have been reported from marketed use. Central/Peripheral nervous system disorders: Paraesthesia was observed in 18%, headache in 25% and primary clinical trial database; the postmarketing reporting rate is ‘Rare’. Post-marketing and other experience Reporting rates for events detected in studies other than those in the primary clinical trial database and/or from spontaneous post-marketing reports are shown in Table 3. Table 3 Reporting rates for events detected in other clinical studies or from spontaneous post-marketing reports a SOC Renal and urinary disorders Blood and lymphatic system disorders Endocrine disorders Gastrointes tinal disorders Skin and subcutaneo us disorders Musculosk eletal disorders Investigati ons Frequency Common (≥1% and <10%) Unknown Event Renal paina Unknown Unknown Diabetes insipidus Pancreatitis Unknown Pruritus Cardiac disorders Unknown Musculosk eletal disorders Unknown Investigati ons Unknown Neutropenia Unknown Myalgia Unknown Blood amylase increased Electrocardio gram QT prolongedb, Ventricular arrhythmia Muscular weakness, Myopathy, Myositis, Rhabdomyoly sis Blood creatine phosphokinas e increased This reporting rate is based on 7 reports of renal pain from two prospective clinical trials involving 107 patients (trials 90FP48 and 91FP49). There were no reports in the primary clinical trial database; the postmarketing reporting rate is ‘Very rare’. b This reporting rate is based on 3 spontaneous reports of QT prolongation from 80000 patients. dizziness in 12%. Involuntary muscle contractions and tremor were seen in 9 and 5% respectively. Hypoaesthesia, ataxia and neuropathy were observed in 7, 4 and 6% respectively. Psychiatric disorders: Anorexia, anxiety and nervousness were observed in 15 and 5% respectively and depression in 10%, confusion in 7%, psychosis in 1%, agitation in 3% and aggressive reaction in 2%. White blood cells: Adverse events related to white blood cells included leukopenia 9%, granulocytopenia 17%. In these patients over 90% had some degree of leukopenia already before foscarnet administration, in 8% severe or even life-threatening. Moreover in some patients, it is noteworthy that mean WBC counts increased during treatment with foscarnet. Although a few patients worsened in this respect, there is no clear evidence to indicate that foscarnet is myelosuppressive. Platelet, bleeding, clotting disorders: Thrombocytopenia was observed in 4%. Skin and appendages: Rash was observed in 16%. Liver and biliary system disorders: Abnormal liver function was observed in 4% and increase in serum ALAT and ASAT in 3 and 2% respectively and gamma GT in 2%. Cardiovascular disorders: Abnormal ECG, hypertension and hypotension were observed in 1, 4 and 2% respectively. Heart rate and rhythm disorders: Ventricular arrhythmia has been reported in 2 patients from marketed use. Urinary system disorders: A few cases of diabetes insipidus, usually of the nephrogenic type, have been reported from marketed use. Musculo-skeletal disorders: Muscle weakness has been reported from marketed use. Overdose symptoms, Overdose symptoms, emergency procedures, emergency Overdose antidotes procedures, antidotes Overdose has been reported during the use of Foscavir in 33 patients, the highest dose being some 20 about 10 times the recommended prescribed dose. Some of the cases were relative overdoses, in that the dose of drug used had not been promptly adjusted for a patient experiencing reduced renal function. There are cases where it has been reported that no clinical sequelae were consequent on the overdose. 28 of the patients experienced adverse events and 5 patients suffered no ill effects in connection with foscarnet overdosing. 4 patients died, one from respiratory/cardiac arrest 3 days after stopping foscarnet, one from progressive AIDS and renal failure approximately 2 months after the last foscarnet dose, one from end stage AIDS and bacteraemia 2 weeks after overdosing and one from multi-organ failure 11 days after stop of foscarnet. The pattern of adverse events reported in association connection with an overdose of Foscavir was is in accordance correspondence with the known adverse event profile of the drug symptoms previously observed during foscarnet therapy. Foscarnet is not compatible with dextrose 30% solution, amphotericin B, acyclovir sodium, ganciclovir, pentamidine isethionate, trimethoprim-sulfamtoxazole and vancomycin hydrochloride. Neither is foscarnet compatible with solutions containing calcium. It is recommended that other drugs should not be infused concomitantly in the same line. until further experience is gained. 1 Overdose has been reported during the use of Foscavir, the highest dose being some 20 times the recommended dose. Some of the cases were relative overdoses, in that the dose of drug used had not been promptly adjusted for a patient experiencing reduced renal function. There are cases where it has been reported that no clinical sequelae were consequent on the overdose. The pattern of adverse events reported in association with an overdose of Foscavir is in accordance with the known adverse event profile of the drug. Haemodialysis increases Foscavir elimination and may be of benefit in relevant cases. Foscarnet is not compatible with dextrose 30% solution, amphotericin B, acyclovir sodium, ganciclovir, pentamidine isethionate, trimethoprim-sulfamtoxazole and vancomycin hydrochloride. Neither is foscarnet compatible Incompatibilities with solutions containing calcium. It is recommended that other drugs should not be infused concomitantly in the same line, until further experience is gained. :הערות the section "Undesirable Effects" was rewritten in the manufacturer's format שבו מסומנים השינויים מודפסים באדום הועבר בדואר אלקטרוני,העלון ...__________..בתאריך .קיים עלון לרופא והוא מעודכן בהתאם דרישת היצרן:אסמכתא לבקשה השינוי הנ"ל אושר על ידי רשויות הבריאות ב :בריטניה אני ,הרוקח הממונה של חברת טק-או-פארם-ליברה מצהיר בזה כי אין שינויים נוספים בעלון. _____________________________ חתימת הרוקח הממונה