Induced Nuclear Reactions – Artificial Transmutation

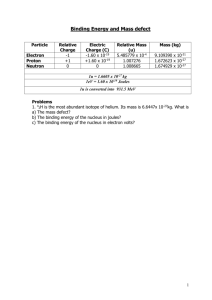
Induced Nuclear Reactions – Artificial Transmutation
So far, we have talked about spontaneously occurring nuclear reactions – when a nucleus is unstable it can naturally emit radiation to become more stable.
Why would a nucleus be unstable? When it has too many protons.
The neutrons are there to help the protons repel each other less.
What is the optimum ration of neutrons to protons? 1:1
Do you remember what number of protons is the most that an atom can have not be considered radioactive? 83
So, when an atom is unstable it can emit a few different types of radiation, and you can look up those types on Table O in your reference tables.
What would the equation look like if an atom of 226
88
Ra spontaneously emits an alpha particle?
226
88
Ra → 222
86
Ra + 4
2
He
How about it an atom of 214
82
Pb spontaneously emits a beta particle?
214
82
Pb → 214
83
Bi + 0
-1 e
What do these two equations have in common?
Do we end up with the same element that we started with? No
This is called transmutation – when the atomic nucleus of one element is changed in to the nucleus of a different element.
How about the location of the particle in the equation?
They’re both on the right side.
That’s how you can tell that the transmutation happened spontaneously – nothing had to be added to the original atom for it to transmutate and the only particles present are the ones that are emitted.
How would the equation change if we wanted to create artificial transmutation by bombarding the nucleus with particles to bring about a change on purpose? The particle would be on the left side of the arrow.
And that’s how you can tell the difference just by looking at the equation. Here’s an example of artificial transmutation:
14
7
N + 4
2
He → 17
8
O + 1
1
H
In this case we’ve bombarded an N-14 atom with an alpha particle, turned the N in to O-17 and emitted a proton. You can predict the outcome of an artificial bombardment reaction the same way that we did with spontaneous decay – the atomic masses and numbers on the left of the arrow has to equal its counterparts on the right. Try this example:
What would we have to bombard an atom of 27
13
Al with to result in the formation of an atom of 30
15
P and the emition of a neutron?
27
13
Al + ? → 30
15
P + 1
0 n
27
13
Al + 4
2
He → 30
15
P + 1
0 n
What’s the charge on an alpha particle?
+2
What’s the charge of a nucleus?
Positive
What does that tell you about how easy it is to cause transmutation by bombarding a nucleus with an alpha particle?
It’s not easy because + repels +
It is possible though because this repulsive force can be overcome if the particle has sufficient energy. Scientists can supply this energy by accelerating the particle in a device called – can you guess? – a particle accelerator. There are different types of particle accelerators with names like “cyclotron” and “synchrotron”, but they all work on the same basic principle. There is a long series of tubes that contain electric or magnetic fields that the particle passes through by attractive and repulsive forces.
Finally, when the particle passes through the last tube it is accelerated in to the target nuclei at close to the speed of light (3 x 10 8 m/s.)
Can all particles be accelerated this way? No, only the charged ones.
Which ones can’t? Neutrons
Would you need to accelerate a neutron to bombard a nucleus with it?
No, since it neutrons don’t have a charge, they aren’t repelled by the target nucleus.
Scientists use neutron bombardment reactions to make stable nuclei radioactive. Tc-99 can be prepared this way.
What is Tc-99 used for? To detect brain tumors.
Take a minute to figure out what type of atom would have to be bombarded with a neutron in order to create an atom of 99
43
Tc. I’ll tell you also that an electron is emitted.
1
0 n + ? → 99
43
Tc + 0
-1 e
1
0 n + 98
42
Mo → 99
43
Tc + 0
-1 e
A R T I I F I I C I I A L T R A N S M U T A T I I O N P r o b l l e m s s
1. In the reaction:
92
U 238 +
2
He 4 X +
0 n 1 the nucleus represented by x is ____
94
Pu 241 _ _____
2. In the reaction:
X +
1
H 1
3
Li 6 +
2
He 4 the nucleus represented by x is ____
4
Be 9 ______
3. In the reaction:
3
Li 6 +
0 n 1
2
He 4 + X the nucleus represented by x is ___
1
H 3 ______
What is that product called? tritium