TPJ_4524_sm_Legends
advertisement

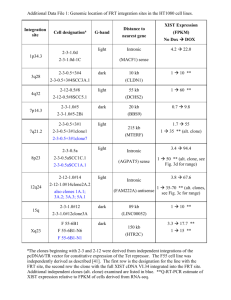
Supplemental Figure Legends Figure S1. The recombineering procedure (the DH10B to SW102 transfer step) does not induce unwanted alterations in the large TAC clones. Fingerprint of 6 TAC clones before (C lanes) and after (lanes1 through 5) transferring from DH10B to SW102. DNA from 5 independent clones (lanes 1 to 5) for each of the 6 selected genes were digested with HindIII and the fragments were separated by electrophoresis on a 1% agarose 1xTAE gel. The names of tagged genes and the sizes of the corresponding JAtY clones are as indicated. Figure S2. The recombineering procedure (the SW102 to Agrobacterium transfer step) does not induce unwanted alterations in the large TAC clones. Fingerprint of 6 TAC clones before (C lanes) and after (lanes1 through 3) transferring from SW102 to Agrobacterium. To fingerprint these clones, the JAtY clones were transferred back to SW102 to obtain DNA of sufficient quality to enable fingerprinting. DNAs from 3 independent clones for each of the 6 selected genes were digested with HindIII and the fragments were separated by electrophoresis on a 1% agarose 1xTAE gel. The names of tagged genes and the sizes of the corresponding JAtY clones are as indicated. Figure S3. Large JAtY clones can be stably maintained in Agrobacterium. Three independent clones (lanes 2 to 4 of each gel) were fingerprinted after approximately 48 generation. To fingerprint these clones, the JAtY clones were transferred back to SW102 to obtain DNA of sufficient quality to enable fingerprinting. DNAs from 3 independent clones for each of the two selected genes were digested with HindIII and the fragments were separated by electrophoresis on a 1% agarose 1xTAE gel. The original clones kept in SW102 were used as controls. Figure S4. Schematic representation of six JAtY clones. The relative sizes, positions, and orientations of the genes in TAC clones are represented by arrows. Black arrows indicate the tagged gene in each TAC clone. Light gray arrows mark tandem duplications http://bioinformatics.psb.ugent.be/plaza Figure S5. Expression of the tagged Aux/IAA transgenes mimics that of the corresponding endogenous genes. Primers spanning exon-exon junctions were designed to specifically amplify mRNA transcribed from the endogenous IAA5 gene and the IAA5-GFP transgene. Fold change was determined by comparing the expression level in pooled IAA5-GFP T2 seedlings treated with 0.1mM IAA for 1h to that of the corresponding mock-treated seedlings. The experiment was performed with 3 biological replicates (3 independent IAA5-GFP transgenic lines were used and the names of these lines are as displayed). The PCR products were sequenced to confirm the specificity of the assay. Figure S6. Protein alignment of domain IV of Aux/IAAs. The multi-alignment was generated by ClustalW. Amino acids of predicted domain IV and part of domain III were included. Box in the figure displays 22 amino acids that are highly 1 conserved across all Aux/IAAs and that were replaced by Ypet in the recombineering experiments. 2