CARBONYL COMPOUNDS – ALDEHYDES & KETONES
advertisement

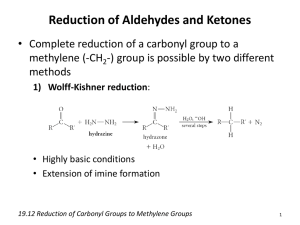
CARBONYL COMPOUNDS – ALDEHYDES & KETONES Introduction: Aldehydes and ketones are known as carbonyl compounds because they contain the carbonyl group. The carbon-oxygen bond is polar because of the electronegativity of the oxygen. C O Name and Structure: Aldehydes: The name aldehyde is derived from alcohol dehydrogenated. The systematic name for aldehydes is alkanals. The carbonyl group is always on the end, and a hydrogen is attached to the carbonyl carbon. R H C O The name ends with the suffix –al. e.g. methanal, ethanal, propanal etc Methanal is also known as formaldehyde and ethanal as acetaldehyde. Ketones: Alkanone is the systematic name for ketones. The functional group is not at the end and its position must be indicated in the name. R' R C O The name ends in the suffix –one. Propanone (also known as acetone). pentan-2-one etc. Bonding, Properties & Uses Intermolecular bonding results from relatively strong dipole-dipole interaction due to the polar nature of the carbonyl group in aldehyde and ketone molecules. Melting and boiling points increase with increasing molecular mass. Most ketones and aldehydes are volatile liquids with definite odours – aldehydes tend to have unpleasant, pungent smells, while ketones tend to have pleasant sweet smells. The oxygen of the carbonyl group is able to hydrogen bond with water molecules hence C1-C3 aldehydes and C3 & C4 ketones are water soluble. Aldehydes C4-C6 and ketones C5 are slightly soluble. LRY 2009 Both tend to be soluble in non-polar solvents. Formalin is a solution about 40% methanal and 60% water, is used as a preservative and disinfectant. Acetone is a major industrial solvent (IPA). It is used in nail polish remover. Butanone is used in model aeroplane glue. Preparation of carbonyl compounds. Aldehydes are made by the oxidation of primary alcohols. To stop the product being further oxidised to carboxylic acids the aldehyde is removed by fractional distillation. CH3CH2OH Cr2O72-- H + CH3CHO Cr2O72-- H + CH3COOH Ketones are prepared by oxidising secondary alcohols. It is a slow reaction and to ensure all alcohol is oxidised it is carried out under reflux. CH3CH2CH2CHOHCH3 Cr2O72-- H + CH3CH2CH2COCH3 Distinguishing Aldehydes and Ketones: Aldehydes are more reactive than ketones because: The carbonyl group of the aldehyde is more polarised. In ketones, the polarity of the carbonyl group is decreased by the presence of the second alkyl group. In ketones the large alkyl groups attached to the carbonyl group get in the way of reactant species. The carbon to hydrogen bond of the aldehyde carbonyl group can be oxidised to a C-O-H bond. The fact that aldehydes are easily oxidised and ketones are not is the basis of the tests to distinguish these two carbonyl compounds. Tests for Aldehydes: Aldehydes are easily oxidised to carboxylic acids. As they do they reduce the other reactant. This is the basis of tests for aldehydes. Oxidising Agent Potassium dichromate (acidified) Potassium permanganate (acidified) Tollens’ reagent – ammoniacal silver nitrate Fehling’s solution Benedict’s solution LRY 2009 Colour Change Orange solution to green Purple solution to colourless Silver precipitate or silver mirror Blue solution forms orange/red precipitate Blue solution forms orange/red precipitate Tollens’ Reagent: Tollens’ reagent contains diamminesilver(I) complex ion [Ag(NH3)2]+, made up by: Putting 2 – 3 mL aqueous silver nitrate in a clean test tube. Adding one drop of NaOH(aq) so that a precipitate of silver hydroxide forms. Adding just enough dilute ammonia NH3(aq) to redissolve the precipitate forming [Ag(NH3)2]+ Aldehydes reduce the silver ions Ag+ to form metallic silver, which is seen as a silver mirror on the inside of the test tube (or a black/grey precipitate) Ketones do not react with Tollens’ reagent. Benedict’s Test: Benedict’s solution contains aqueous copper (II) ions Cu2+ which (this makes to solution blue coloured), in alkaline conditions. To prevent the copper from precipitating out as Cu(OH)2, the Cu2+ is complexed with citrate ions. When Benedict’s solution is added to an aldehyde and warmed, a reddish, brown precipitate of Cu (I) oxide, (Cu2O) is formed. CH3CHO + Ethanal 4OH- + 2Cu2+ Benedict’s soln. CH3COOH + Cu2O Fehling’s solution is another reagent with copper(II) ions in alkaline solution. It behaves exactly the same as Benedict’s solution. Ketones do not react with Benedict’s solution and Fehling’s solution because the ketones cannot reduce metal ions. Reaction Summary for Ethanal Tollens’ reagent Silver mirror/ppt Ethanol CH3CH2OH H+/Cr2O72- ethanal cold CH3CHO H+/Cr2O72 warm ethanoic acid CH3COOH Benedict’s solution Blue to orange LRY 2009