pmic7400-sup-0006-suppmat
advertisement

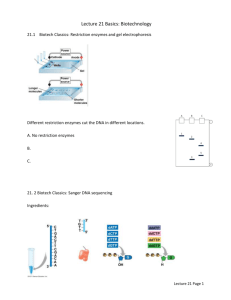
For the Tutorial Programme in Proteomics High-Throughput Cloning and Expression Library Creation for Functional Proteomics - Supplementary Material Basic concepts – Supplementary Material 1. Main enzymes used in cloning methods DNA polymerase. The synthesis of a new strand of DNA is a process mediated by a DNA polymerase. It requires the use of a template, which can be a DNA or RNA molecule, the latter resulting in the complementary copy of the mold. DNA-dependent DNA polymerase was first isolated in Escherichia coli in 1958 (Lehman et al 1958) and in 1986 the Polymerase Chain Reaction (PCR) was developed (Mullis et al 1986, Saiki et al 1988). PCR is an in vitro reaction that allows the copy of a single DNA molecule into millions, an extremely important feature for DNA cloning. In a PCR reaction, short sequences of DNA, called primers, target specific sequences and .then provide a starting point for the amplification. Since the DNA amplification only occurs in the 5 3’ orientation and the two strands of the DNA are anti-parallel, two primers are used in the PCR reaction: one that aligns with the end of the target sequence in the 5’-3’ strand of the template, and another one that aligns with the beginning of the target sequence in the complementary strand (3’-5’). Each cycle, the template DNA is denaturated, the primers align with the template and the DNA is amplified, exponentially amplifying the region flanked by the two primers (Supplementary Figure 1A). RNA-dependent DNA polymerases, also known as transcriptases, are used to copy the information from RNA molecules into DNA, referred as copy-DNA (cDNA) (Supplementary Festa, et al. 1 10/04/2012 For the Tutorial Programme in Proteomics Figure 1B). cDNA synthesized from gene transcripts can be used as templates for PCR reactions and allow the cloning of the transcript into vectors. Restriction enzymes type II and ligases. These two sets of enzymes have complementary activity, restriction enzymes work as “scissors” capable of identifying and cleaving specific DNA sequences (Kelly and Smith 1970, Smith and Wilcox 1970), whereas ligases re-join two DNA strands and reconstitute the phosphodiester bonds (Weiss and Richardson 1967). Restriction enzymes provide bacteria with protection against exogenous DNA from viruses and other bacterial strains, working as a rudimentary immune system (Arber 1965, Roberts 2005). To cleave the DNA, restriction enzymes make two incisions, one in each DNA strand, and depending upon the relative position of the two incisions the product can have a blunt end or, with staggered cuts, overhanging ends (Williams 2003) (Supplementary Figure 2). For DNA cloning, the vector and the gene of interest have to be digested with restriction enzymes that possess compatible products to allow the strands to come together in perfect alignment, for subsequent ligation. Recombinases. Nucleic acid recombination is a process in which DNA fragments are exchanged between two DNA molecules and it is mediated by enzymes called recombinases. These enzymes have the intrinsic activity to cut and ligate DNA fragments, within a single complex. Recombination was originally described in maize by Barbara McClintock in 1931 (Creighton and McClintock 1931, McClintock 1931), however it took five decades for the first recombinase and its precise mechanism of action to be elucidated (Abremski and Hoess 1984, Hoess and Abremski 1984). Recombinases may present distinct properties regarding the mechanism of action, substrate preferences and types of products generated. For DNA cloning it is important that the recombination happens precisely in specific sequences and generates consistent products every time, without adding or removing any nucleotide during the ligation of the two DNA molecules. Enzymes with those properties are called site-specific recombinases. Festa, et al. 2 10/04/2012 For the Tutorial Programme in Proteomics Depending on the configuration of sequences recognized by these enzymes, the recombination reaction can be unidirectional, between distinct DNA sequences (Supplementary Figure 3A), or bidirectional, among identical sequences (Supplementary Figure 3B). This process is largely independent of the sequence to be cloned, making it suitable for high-throughput cloning projects. 2. Vector features In functional genomics the term plasmid refers to a circular double stranded DNA with the ability to replicate within the cell, independently of the genomic DNA. The process to introduce a plasmid (clone or empty vector) into bacteria is called transformation and it is commonly used for the amplification and the isolation of plasmids. In eukaryotic cells, the introduction of a plasmid into cells is designated transfection (if it is done chemically) or transduction (if it is done using viral delivery). The basic elements common to all vectors include: the replication site, antibiotic resistance, and cloning site (Supplementary Figure 4). The replication site, or origin of replication, comprises a nucleotide sequence that can be recognized by the cellular DNA replication machinery as a start point for the synthesis of the new copy (Nagata and Meselson 1968). Different organisms recognize distinct replication sites, therefore the vector must have the origin of replication appropriate for the host cell of choice (Errico and Costanzo 2010). Once in the cell, the vector is copied multiple times and shared among daughter cells after the cell divides. Alternatively, some vectors, usually the ones derived from virus, are integrated into the host genome and passed to the progeny cells as part of the genome. Festa, et al. 3 10/04/2012 For the Tutorial Programme in Proteomics Selection markers are genes that alter the proliferation capabilities of the host cell. The purpose of these markers is to provide growth differences between cells with and without the plasmid, enriching the cell population for the recombinant cells of interest. The selection can be positive or negative; meaning that cells carrying the plasmid will survive or die, respectively. Positive selection is usually accomplished by the use of an antibiotic in the growth media and a gene, coded by the plasmid, that makes the bacterium carrying the gene resistant to the drug. A classical example is ampicillin and beta-lactamase gene, usually referred as bla gene or AmpR (Ampicillin Resistance). Recombinant cells expressing bla can grow in media containing ampicillin, while the parental cells, without the plasmid, are sensitive to the antibiotic and cannot proliferate. Negative selection can be accomplished by the use of toxic genes, such as ccdB (DNA gyrase inhibitor) and barnase (ribunuclease), as the selection marker. Once expressed, the toxic gene kills the host cell. This type of selection is important to avoid the growth of cells carrying by-products of the cloning process. Negative selection markers can be propagated in special strains that are resistant to them. Cloning sites. Vectors also have specific sequences to allow the cloning of the insert of interest. Depending on the cloning methodology used, those sequences can be sites recognized by restriction enzymes or recombinases. Restriction sites for many distinct enzymes are often present in tandem in a region referred as multiple cloning sites (MCS). Festa, et al. 4 10/04/2012 For the Tutorial Programme in Proteomics 3. References Abremski K, Hoess R (1984). Bacteriophage P1 site-specific recombination. Purification and properties of the Cre recombinase protein. J Biol Chem 259: 1509-1514. Arber W (1965). Host-controlled modification of bacteriophage. Annu Rev Microbiol 19: 365-378. Creighton HB, McClintock B (1931). A Correlation of Cytological and Genetical Crossing-Over in Zea Mays. Proc Natl Acad Sci U S A 17: 492-497. Errico A, Costanzo V (2010). Differences in the DNA replication of unicellular eukaryotes and metazoans: known unknowns. EMBO Rep 11: 270-278. Hoess RH, Abremski K (1984). Interaction of the bacteriophage P1 recombinase Cre with the recombining site loxP. Proc Natl Acad Sci U S A 81: 1026-1029. Kelly TJ, Jr., Smith HO (1970). A restriction enzyme from Hemophilus influenzae. II. J Mol Biol 51: 393409. Lehman IR, Bessman MJ, Simms ES, Kornberg A (1958). Enzymatic synthesis of deoxyribonucleic acid. I. Preparation of substrates and partial purification of an enzyme from Escherichia coli. J Biol Chem 233: 163-170. McClintock B (1931). The Order of the Genes C, Sh and Wx in Zea Mays with Reference to a Cytologically Known Point in the Chromosome. Proc Natl Acad Sci U S A 17: 485-491. Mullis K, Faloona F, Scharf S, Saiki R, Horn G, Erlich H (1986). Specific enzymatic amplification of DNA in vitro: the polymerase chain reaction. Cold Spring Harb Symp Quant Biol 51 Pt 1: 263-273. Nagata T, Meselson M (1968). Periodic replication of DNA in steadily growing Escherichia coli: the localized origin of replication. Cold Spring Harb Symp Quant Biol 33: 553-557. Roberts RJ (2005). How restriction enzymes became the workhorses of molecular biology. Proc Natl Acad Sci U S A 102: 5905-5908. Saiki RK, Gelfand DH, Stoffel S, Scharf SJ, Higuchi R, Horn GT et al (1988). Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science 239: 487-491. Festa, et al. 5 10/04/2012 For the Tutorial Programme in Proteomics Smith HO, Wilcox KW (1970). A restriction enzyme from Hemophilus influenzae. I. Purification and general properties. J Mol Biol 51: 379-391. Weiss B, Richardson CC (1967). Enzymatic breakage and joining of deoxyribonucleic acid, I. Repair of single-strand breaks in DNA by an enzyme system from Escherichia coli infected with T4 bacteriophage. Proc Natl Acad Sci U S A 57: 1021-1028. Williams RJ (2003). Restriction endonucleases: classification, properties, and applications. Mol Biotechnol 23: 225-243. Figures Legends Supplementary Figure 1 - Polymerase Chain reaction and cDNA synthesis. DNA Polymerases are largely employed in cloning techniques. (A) DNA-Dependent DNA polymerase is used in PCR reactions for the amplification of the desired insert. Every cycle is composed of a denaturation step, followed by annealing of the primers and extension (DNA synthesis). After 20-30 cycles, millions copies of the sequence flanked by the primers is obtained. (B) RNA-dependent DNA polymerase is used to create a DNA copy (cDNA) of the mRNA. Supplementary Figure 2 - Restriction enzymes and ligases. Type II restriction enzymes cut the DNA at specific recognition sequences (often palindromes) creating blunt ends (A) or cohesive ends (B), depending on the relative position each strand of the DNA is cleaved. This reaction can be reversed by the action of the ligases, enzymes that ligate two DNA molecules by reforming the phosphodiester bond. Supplementary Figure 3 – DNA Recombinases. Site-specific recombinases have the combined activities of a restriction enzyme and a ligase. They are capable of identifying specific sequences in the DNA and exchanging those sequences between two distinct DNA Festa, et al. 6 10/04/2012 For the Tutorial Programme in Proteomics molecules. The recombination can be: (A) unidirectional, when distinct sites are used, or (B) bidirectional, using identical sites. Supplementary Figure 4 – Vector Features. Vectors are circular DNA molecules capable of replicating independently of the genomic DNA in bacteria, using their origin of replication (ori). In biotechnology, they usually carry a promoter to drive the expression of a selection marker, for example beta-lactamase that confers resistance to ampicillin. Cloning sites are also added to facilitate the insertion of the gene of interest. Depending on the sequence, the cloning site can be recognized by restriction enzymes or recombinases. In addition to these features, expression vectors also include a promoter to drive the transcription of the insert and often a fusion tag in frame with the coding sequences. Festa, et al. 7 10/04/2012