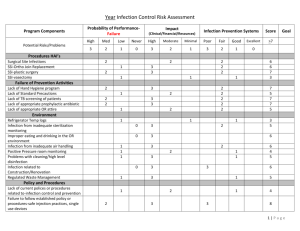
A cellular model for pandemic influenza outbreak simulation and
advertisement

A cellular model for pandemic influenza outbreak simulation and mitigation Paper Presenter: Satish K Ramchurn, Department of Physics, Faculty of Science, University of Mauritius Author(s): Satish K Ramchurn, Department of Physics, Faculty of Science, University of Mauritius Smita SD Goorah, Department of Medicine, Faculty of Science, University of Mauritius B Thakoor, Department of Physics, Faculty of Science, University of Mauritius RL Roopchund, Department of Physics, Faculty of Science, University of Mauritius Introduction Influenza is an airborne infectious disease which has a huge public health impact worldwide. Only in the twentieth century, there have been three pandemic outbreaks of influenza: in 1918, 1957 and 1968 causing widespread mortality especially amongst healthy people. The 1918 outbreak was the most deadly killing 20-40 million people worldwide with nearly half of these deaths in young adults in the 20-40 year age group. It is generally agreed that the world is moving closer to another pandemic. The country most affected by the current avian H5N1 influenza virus is Indonesia where there have been several suspected cases on the virus being directly transmitted to humans and causing deaths. Disease surveillance systems have been stepped up throughout the world to act quickly should there be the beginnings of a new pandemic. Since this infection is unpredictable and can propagate rapidly, a high state of alert must be maintained and timely public health action initiated. Measures to mitigate the effects of this pandemic must necessarily have a strong medical component. However, the simulation of the dynamical evolution of an outbreak is also crucial to inform on the appropriateness and timing of public health interventions. This is important particularly for a small island state like Mauritius where resources (human, financial and others) are limited and should be utilized to maximal effectiveness. Method A cellular model for simulating the spatio-temporal evolution of an airborne viral infection like influenza was developed. The region of interest was divided into cells. Each cell was assumed to be homogeneous. The development of the viral infection within a cell was characterized using an SIR (susceptible-infectious-removed) compartmental approach. Intercellular interactions depended on the distance between the cells. These interactions were described by a set of differential equations with time as the independent variable. The differential equations were computationally integrated to simulate the propagation of the virus throughout the population for a population homogeneously distributed in the cells and with one infected person randomly located in one of the cells. Results In our model, the wavefront of the infection was found to evolve radially from the cell containing the index case. Discussion The proposed model captures the intuitive radial spread of the airborne infection for a randomly located index case in homogeneously distributed populations. This is the simplest application of the model. By ascribing features to each cell to mimic more realistic actual conditions, one should also be able to describe the spread of the infection in cases such as in populations distributed inhomogeneously in the cells and for cells with geographic structures. Since the propagation of airborne infectious diseases is preventable by timely public health measures, rapid identification of index cases and modeling the potential spread of the infection in the community can help public health authorities rapidly identify vulnerable geographic locations and take disease mitigating measures in them. Dynamical models such as the one proposed here could be vital tools in mitigating the effects of an influenza pandemic outbreak in Mauritius and in controlling the propagation of infectious diseases in general.