Salt Iodine Titration Method
advertisement

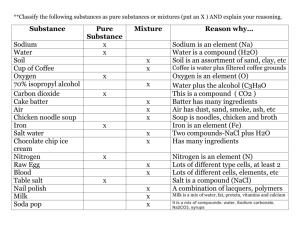
Salt Iodine Titration Method The reference method for measuring the iodine content of salt is iodometric titration with the thiosulfate-starch reaction as the external indicator. Reagents for testing iodate salt include 0.005 M sodium thiosulfate (Na2S2O3), 2 N sulfuric acid (H2SO4), 10% (w/v) KI, and the starch indicator solution. For salt fortified with KI, additional reagents comprise methyl orange indicator, bromine water, sodium sulfite solution, and phenol solution. Salt iodized with KIO3: 1. Ten grams of the iodate salt sample are dissolved with 50 mL water in a 250-mL Erlenmeyer flask. 2. One milliliter of 2 N sulfuric acid is added to release the free iodine from the iodate. 3. Five milliliters of 10% KI is then added, which, in the presence of iodine, turns the solution pale yellow. 4. A stopper is placed in the flask; the flask is placed in a dark space for 10 minutes. 5. Upon removal of the flask from the dark space, sodium thiosulfate from the titration burette is added until the solution turns pale yellow. 6. Approximately 2 mL of the starch indicator solution is then added, turning the solution dark purple. 7. Titration is continued until the solution becomes colorless. 8. The burette reading indicates the total amount of thiosulfate added to the solution, which is converted to mg/kg of iodine in salt: “x” mL titrated sodium thiosulfate * 0.1058 * 100 = Iodine mg/kg. Salt iodized with KI: 1. Salt fortified with iodide requires a slightly different procedure. 2. After dissolving 10 g of salt in 50 mL water, a few drops of methyl orange indicator are added until the solution turns pale orange. 3. Next, 1 drop of the 2N sulfuric acid is added, which results in a color change to pink. Bromine water (500 l) is then added, changing the solution’s color to yellow. 4. Drops of sodium sulfite solution are added to the solution until it turns to pale yellow. 5. Three drops of phenol solution are added turning the solution clear. 6. After 1 mL H2SO4 and 5 mL KI solution are added, titration with sodium thiosulfate is started. 7. Once the solution turns pale yellow, 1 mL of the starch indicator solution is added, turning the solution dark purple. 8. Titration is resumed until the solution is colorless. The burette reading is recorded and converted to mg/kg of iodine in salt. Reagents: 1. Methyl Orange Indicator - Dissolve 0.01 g methyl orange in 100 mL water. 2. Bromine Water - Place 5mL in a small flask, (keep in fume hood due to dangerous fumes). 3. Sodium Sulfite Solution - Dissolve 1g sodium sulfite in 100mL water. 4. Phenol Solution - Dissolve 5g phenol in 100mL water. 5. Starch Solution - Weigh 1 g soluble starch into a 100 mL beaker, add 10 mL water, heat to dissolve. Add saturated NaCl solution to the hot starch solution to make up to 100 mL. (Store all reagents in a cool, dark place). Use of trade names and commercial sources is for identification only and does not imply an endorsement by the U.S. Department of Health and Human Services (DHHS) and the Centers for Disease Control and Prevention (CDC).