Chemical Formula
advertisement

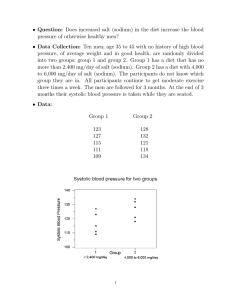
DAILY 5 SQUARE October 30 QUESTION If Sodium and Chlorine can react chemically to form table salt. How does the mass of the table salt formed in this reaction compare to the mass of the sodium and chlorine before the reaction takes place? A. The mass of the table salt formed is less than the sodium and chlorine. B. The mass of the table salt formed is greater than the sodium and chlorine. C. The mass of the table salt formed is twice the mass of the sodium and chlorine. D. The mass of the table salt formed is the same as the mass of the sodium and chlorine. SOL NOTES Closed system Law of conservation of mass RELATED WORDS Chemical equation Reactants Product Coefficient Subscript Chemical Formula Nov Science Benchmark – Tuesday, November 5 Study all 5 squares since Day 1 Elements # 6- #10 Test tomorrow Atomic number Element symbol # of protons, neutrons, electrons Group #, Period # Valence electrons , # of energy levels Type – metal, nonmetal, metalloid Occurs as – solid, liquid, gas Element test #1 1 2 3 4 5