הודעה על החמרה ( מידע בטיחות) בעלון לצרכן
advertisement

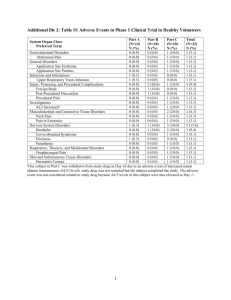
)בטיחות )מידע בטיחות החמרה (( מידע על החמרה הודעה על הודעה 116..60.11 תאריך BOOSTRIX שם תכשיר באנגלית 121 34 30059 00 מספר רישום GlaxoSmithKline (Israel) Ltd בעל הרישום השינויים בעלון מסומנים על רקע צהוב לרופא בעלון לרופא בעלון ים/ים המבוקש/פרטים על השינוי טקסט חדש Addition: Extremely rare cases of collapse or shock-like state (hypotonichyporesponsiveness episode) and convulsions within 2 to 3 days of vaccination have been reported in DTPa and DTPa combination vaccines. Syncope (fainting) can occur following, or even before, any vaccination as a psychogenic response to the needle injection. It is important that procedures are in place to avoid injury from faints. טקסט נוכחי פרק בעלון Warning and Precautions Approximately 3000 vaccinees have received a dose of Boostrix in clinical studies. The safety profile presented below is based on data from clinical trials where Boostrix was administered to 839 children (from 4 to 8 years of age) and 1931 adults, adolescents and children (from 10 to 76 years of age). Approximately 3000 vaccinees have received a dose of Boostrix in clinical studies. The most common events occurring after vaccine administration were local injection site The most common events occurring reactions (pain, redness after Boostrix vaccine administration in and swelling) reported both groups were local injection site by 50-92% of subjects reactions (pain, redness and swelling) in each trial. These reported by 23.7-80.650-92% of usually had their onset subjects in each trial. These usually had within the first 48 their onset within the first 48 hours hours after vaccination. after vaccination. All resolved without All resolved without sequelae. sequelae. Adverse events with at least a Adverse events with at suspected causal relationship to least a suspected causal vaccination are listed below. relationship to vaccination are listed Frequencies are reported as: Adverse below. Undesirable reactions reported are listed according to the following frequency: Frequencies are effects reported as: Very common: (1/10) Very common: (1/10) Common: ( 1/100 to < 1/10) Common: ( Uncommon: ( 1/1,000 to < 1/100) Rare: ( 1/10,000 to < 1/100 to < 1/10) Uncommon: ( 1/1,000) 1/1,000 to < 1/100) Very rare: (< 1/10,000) Rare: Within each frequency grouping, ( undesirable effects are presented in 1/10,000 to < 1/1,000) order of decreasing seriousness. Very rare: (< 1/10,000) Clinical trials Within each frequency - For children from the age of 4 years grouping, undesirable onwards Subjects aged 4 – 8 years effects are presented in (N=839436) order of decreasing seriousness. Clinical trials: - For children from the Infections and infestations age of 4 years onwards Uncommon: upper respiratory tract (N=436) infection Clinical trials: Metabolism and nutrition disorders Common: anorexia Psychiatric disorders Very common: irritability Nervous system disorders Very common: somnolence Common: headache Uncommon: disturbances in attention Eye disorders Uncommon: conjunctivitis Gastrointestinal disorders Common: diarrhoea, vomiting, gastrointestinal disorders Skin and subcutaneous tissue disorders Uncommon: rash Metabolism and nutrition disorders Very common: anorexia General disorders and administration site conditions Very common: pyrexia (fever > 37.5°C), injection site reactions (such as redness and/or swelling), injection site pain, fatigue Common: pyrexia (fever ≥37.5ºC including fever >39.0ºC) oedematous swelling of the injected limbextensive swelling of vaccinated limb (sometimes involving the adjacent joint), pyrexia (fever 39.0°C), Uncommon: other injection site reactions (such as induration), pain Psychiatric disorders Very common: irritability Post-marketing surveillance: Nervous system disorders Hypotonic-hyporesponsiveness episodes, convulsion Immune system disorders Anaphylactic reaction, allergic reaction - For adults and adolescents from the age of 10 years onwards Subjects aged Nervous system disorders Very common: somnolence Gastrointestinal disorders Common: diarrhoea, vomiting, Metabolism and nutrition disorders Very common: anorexia General disorders and administration site conditions Very common: pyrexia (fever > 37.5°C), injection site reactions (such as redness and/or swelling), injection site pain, Common: oedematous swelling of the injected limb pyrexia (fever 39.0°C), Psychiatric disorders Very common: irritability Post-marketing surveillance: Nervous system disorders Hypotonichyporesponsiveness episodes, convulsion Immune system disorders Anaphylactic reaction, allergic reaction - For adults and adolescents from the age of 10 years 10 – 76 years (N = 19311,515) onwards (N = 1,515) Clinical trials: Clinical trials: Infections and infestations Uncommon: upper respiratory tract infection, pharyngitis Blood and lymphatic system disorders Uncommon: lymphadenopathy Nervous system disorders Very common: headache Common: dizziness Uncommon: hypertonia syncope Respiratory, thoracic and mediastinal disorders Uncommon: cough Gastrointestinal disorders Common: nausea, gastrointestinal disorders Uncommon: diarrhoea, vomiting Skin and subcutaneous tissue disorders Uncommon: hyperhidrosis, pruritispruritus, rash Musculoskeletal and connective tissue disorders Uncommon: arthralgia, myalgia, joint stiffness, musculoskeletal stiffness Infections and infestations Uncommon: pharyngitis General disorders and administration site conditions Very common: injection site reactions (such as redness and/or swelling), malaise, fatigue, injection site pain Common: pyrexia (fever 37.5°C), injection site reactions (such as injection site mass and injection site abscess sterile) malaise, fatigue Uncommon: pyrexia (fever > 39.0°C), influenza like illness injection site mass, injection site abscess sterile, pain Post-marketing surveillance Blood and lymphatic system disorders Uncommon: lymphadenopathy Nervous system disorders Very common: headache Common: dizziness Uncommon: hypertonia Gastrointestinal disorders Uncommon: vomiting Skin and subcutaneous tissue disorders Uncommon: hyperhidrosis, pruritis Musculoskeletal and connective tissue disorders Uncommon: myalgia, joint stiffness, Infections and infestations Uncommon: pharyngitis General disorders and administration site conditions Very common: injection site reactions (such as redness and/or swelling), injection site pain Common: pyrexia (fever 37.5°C), malaise, fatigue Uncommon: pyrexia (fever > 39.0°C), Because these events were reported spontaneously, it is not possible to reliably estimate their frequency. Blood and lymphatic system disorders Angioedema Immune system disorders Allergic reactions, including anaphylactic and anaphylactoid reactions Nervous system disorders Hypotonic-hyporesponsiveness episodes, convulsions (with or without fever) Skin and subcutaneous tissue disorders Urticaria General disorders and administration site conditions Oedematous swelling of the injected limb Asthenia Immune system disorders Anaphylactic reaction, allergic reaction Data on 146 subjects suggest that there might be a small increase in local reactogenicity (pain, redness, swelling) with repeated vaccination according to a 0, 1, 6 months schedule in adults (> 40 years of age). Data suggest that in subjects primed with DTP in childhood a booster dose might give an increase of local reactogenicity. Following administration of tetanus toxoid containing vaccine injection site mass, injection site abscess sterile, pain Post-marketing surveillance General disorders and administration site conditions Oedematous swelling of the injected limb Immune system disorders Anaphylactic reaction, allergic reaction Data on 146 subjects suggest that there might be a small increase in local reactogenicity (pain, redness, swelling) with repeated vaccination according to a 0, 1, 6 months schedule in adults (> 40 years of age). Following administration of tetanus toxoid containing vaccine