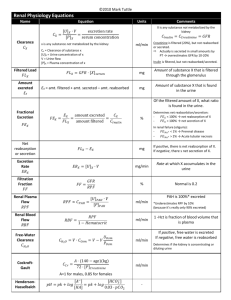
ACID-BASE - SUMMARY
advertisement

Acid-Base Rules – Summary 21/7/10 1. pH – acidaemia or alkalaemia - net deviation from normal indicates presence of an acidosis or alkalosis 2. Assess the pattern - each of the simple disorders produce predictable changes in either PCO2 or HCO33. Look for associated clues – certain conditions produce certain changes in biochemistry 4. Assess for compensation response HCO3 will change for a 10mmHg change in PaCO2 Respiratory Acidosis Respiratory Alkalosis ACUTE 1 -> 2 -> CHRONIC 4 5 Metabolic Acidosis - the ‘1.5 + 8 Rule’ -> expected PaCO2 at max compensation = 1.5 x HCO3- + 8 Metabolic Alkalosis - the ‘Point Seven plus Twenty Rule’ -> expected pCO2 = 0.7 [HCO3] + 20 5. Other Indices in the Assessment of a Metabolic Acidosis - Lactate Anion Gap Delta ratio Osmolar Gap Urinary anion Gap Stewart equation When to calculate what: RAGMA – look for lactate, calculate Delta ratio, Stewart Equation NAGMA – calculate Urinary anion gap, Steward Equation Osmolarity – calculate Osmolar gap Lactate CAUSES (Cohen & Woods classification) Type A - Inadequate Oxygen Delivery (i) anaerobic muscular activity (sprinting, generalised convulsions) (ii) tissue hypoperfusion (shock, cardiac arrest, regional hypoperfusion -> mesenteric ischaemia) Jeremy Fernando (2010) (iii) reduced tissue oxygen delivery (hypoxaemia, anaemia) or utilisation (CO poisoning) Type B - No Evidence of Inadequate Tissue Oxygen Delivery B1: associated with underlying diseases - LUKE: leukaemia, lymphoma - TIPS: thiamine deficiency, infection, pancreatitis, short bowel syndrome - FAILURES: hepatic, renal, diabetic failures B2: associated with drugs & toxins - phenformin cyanide beta-agonists methanol adrenaline salicylates nitroprusside infusion ethanol intoxication in chronic alcoholics anti-retroviral drugs paracetamol salbutamol biguanides fructose sorbitol xylitol isoniazid B3: associated with inborn errors of metabolism - congenital forms of lactic acidosis with various enzyme defects (eg pyruvate dehydrogenase deficiency) Anion Gap = (Na+ + K+) – (Cl- + HCO3-) - normal 12 to 16 - the normal anion gap depends on serum phosphate and serum albumin - the normal AG = 0.2 x [albumin] (g/L) + 1.5 x [phosphate] (mmol/L) Causes of an Anion Gap Metabolic Acidosis – accumulation of organic acids or impaired H+ excretion (LTKR) Lactate Toxins – methanol, metformin, penformin, paraldehyde, propylene glycol, pyroglutamic acidosis, acid, Fe, isoniazid, ethanol, ethylene glycol, salicylates, solvent Ketones Renal Failure Jeremy Fernando (2010) Expanded Causes (MUDPILES) Methanol Uraemia DKA Phenformin, Paracetamol, Pyroglutamic metabolic acidosis, Paraldehyde Iron, isoniazid Lactate Ethylene glycol, ethanol Salicylates, solves Causes of a Non-anion Gap Metabolic Acidosis – loss of HCO3- from ECF (CAGE) Chloride Acetazolamide/Addisons GI loss (diarrhoea, vomiting, enterostomies, fistulae, ileostomies) Extra: RTA type 1 Expanded Causes (HARDUP) Hyperchloraemia Acetazolamide, Addison’s disease Renal tubular acidosis Diarrhoea, vomiting, ileostomies, fistulae Ureteroenterostomies Pancreatoenterostomies Causes of a Low Anion Gap Decrease in unmeasured anions (albumin, dilution) Increase in unmeasured cations (multi-myeloma, hypercalcaemia, hypermagnesaemia, lithium OD, bromide OD, polymixin B) Non random analytical errors (increased Na+, increased viscosity, iodide ingestion, increased lipids) Delta Ratio = the increase in Anion Gap/the decrease in HCO3- indicates what has happen to the denominator (HCO3-) - used in RAGMA to see whether change in HCO3- is appropriate (ie. whether there is a RAGMA component to disorder) - normal value = 1 to 1.5 - if normal there is only one pathology (uncomplicated RAGMA) Interpretation < 0.4 - hyperchloraemic normal anion gap acidosis 0.4 - 0.8 - consider combined high AG & normal AG acidosis BUT note that the ratio is often < 1 in acidosis associated with renal failure Jeremy Fernando (2010) 1 – 2 - usual for uncomplicated high-AG acidosis (lactic acidosis: average value 1.6, DKA: around 1) > 2 - a high delta ratio - > an elevated bicarbonate at onset of the metabolic acidosis -> preexisting metabolic alkalosis or compensated respiratory acidosis. Osmolar Gap - the osmolality is measured in lab calculated osmolarity = (2 x [Na+]) + [glucose] + [urea] the osmolar gap = Osmolality - Osmolarity an osmolar gap > 10 mOsm/l is often stated to be abnormal. Significance - indirect evidence for the presence of an abnormal solute which is present in significant amounts. - ethanol, methanol & ethylene glycol -> will cause an elevated osmolar gap. - [NB: To convert ethanol levels in mg/dl to mmol/l divide by 4.6. For example, an ethanol level of 0.05% is 50mg/dl. Divide by 4.6 gives 10.9mmols/l] Causes of Raised Osmolar Gap (MEPMELK) Methanol/mannitol Ethanol P – isopropyl alcohol Methylene glycol Ethylene glycol Lactate Ketones Causes of a Normal Osmolar Gap Metabolic Acidosis - Pyroglutamic acid - Salicyclates Urinary Anion Gap - the cations normally present in urine are Na+, K+, NH4+, Ca++ and Mg++. - the anions normally present are Cl-, HCO3-, sulphate, phosphate and some organic anions. - only Na+, K+ and Cl- are commonly measured in urine so the other charged species are the unmeasured anions (UA) and cations (UC). Urinary Anion Gap = [Na+]+ [K+] - [Cl-] Clinical Use - the urinary anion gap can help to differentiate between GIT and renal causes of a hyperchloraemic metabolic acidosis. - hyperchloraemic acidosis can be caused by: Jeremy Fernando (2010) (i) loss of base via the kidney (eg renal tubular acidosis) (ii) loss of base via the bowel (eg diarrhoea), or (iii) gain of mineral acid (eg HCl infusion). - if the acidosis is due to loss of base via the bowel then the kidneys can respond appropriately by increasing ammonium excretion to cause a net loss of H+ from the body -> the UAG would tend to be decreased -> increased NH4+ (with presumably increased Cl-) -> increased UC -> decreased UAG. - if the acidosis is due to loss of base via the kidney -> it is not able to increase ammonium excretion and the UAG will not increase. Stewart Equation - pH is dependent on other ions in solution not just H+ and HCO3- there are dependent and independent variables Dependent: - H+ OHHCO3CO32HA (weak acids) A- (weak bases) Independent: - PaCO2 - ATOT (total of weak non-volatile acids) - SID Strong Ion Difference - a strong ion = an ion that totally dissociates at a given pH - SID = strong cations – strong anions SID = (Na+ + K+ + Ca2+ +Mg2+) – (Cl- - other anions) Modified SID = (Na+ - K+) – Cl- SID > 0 = alkalosis SID < 0 = acidosis normal SID of plasma = 40mEq/L (slightly alkalaemic) any movement from this is roughly equal to the standard base excess Simple SID calculator Expected BE = (Na+ - Cl-) – 38 - if expected BE < observed BE -> there is a mixed RAGMA + NAGMA Jeremy Fernando (2010)