signal pep
advertisement

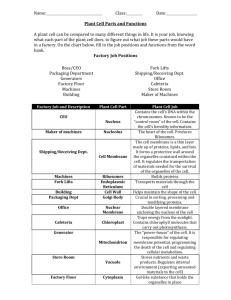
The Topological Relationships of Membrane-enclosed Organelles Can Be Interpreted in Terms of Their Evolutionary Origins To understand the relationships between the compartments of the cell, it is helpful to consider how they might have evolved. The precursors of the first eucaryotic cells are thought to have been simple organisms that resembled bacteria, which generally have a plasma membrane but no internal membranes. The plasma membrane in such cells therefore provides all membranedependent functions, including the pumping of ions, ATP synthesis, protein secretion, and lipid synthesis. Typical present-day eucaryotic cells are 10–30 times larger in linear dimension and 1000–10,000 times greater in volume than a typical bacterium such as E. coli. The profusion of internal membranes can be seen in part as an adaptation to this increase in size: the eucaryotic cell has a much smaller ratio of surface area to volume, and its area of plasma membrane is presumably too small to sustain the many vital functions for which membranes are required. The extensive internal membrane systems of a eucaryotic cell alleviate this imbalance. Figure 12-3 Development of plastids Figure 12-3 . Development of plastids (A) Proplastids are inherited with the cytoplasm of plant egg cells. As immature plant cells differentiate, the proplastids develop according to the needs of the specialized cell: they can become chloroplasts (in green leaf cells), storage plastids that accumulate starch (e.g., in potato tubers) or oil and lipid droplets (e.g., in fatty seeds), or chromoplasts that harbor pigments (e.g., in flower petals). (B) Development of the thylakoid. As chloroplasts develop, invaginated patches of specialized membrane from the proplastid inner membrane pinch off to form thylakoid vesicles, which then develop into the mature thylakoid. The thylakoid membrane forms a separate compartment, the thylakoid space, which is structurally and functionally distinct from the rest of the chloroplast. Thylakoids can grow and divide autonomously as chloroplasts proliferate. The evolution of internal membranes evidently accompanied the specialization of membrane function. Consider, for example, the generation of thylakoid vesicles in chloroplasts. These vesicles form during the development of chloroplasts from proplastids in the green leaves of plants. Proplastids are small precursor organelles that are present in all immature plant cells. They are surrounded by a double membrane and develop according to the needs of the differentiated cells: they develop into chloroplasts in leaf cells, for example, and into organelles that store starch, fat, or pigments in other cell types (Figure 12-3A ). In the process of differentiating into chloroplasts, specialized membrane patches form and pinch off from the inner membrane of the proplastid. The vesicles that pinch off form a new specialized compartment, the thylakoid, that harbors all of the chloroplast's photosynthetic machinery (Figure 12-3B ). Figure 12-4 Hypothetical schemes for the evolutionary origins of (more...) Figure 12-4 . Hypothetical schemes for the evolutionary origins of some membrane-enclosed organelles The origins of mitochondria, chloroplasts, ER, and the cell nucleus can explain the topological relationships of these intra-cellular compartments in eucaryotic cells. (A) A possible pathway for the evolution of the cell nucleus and the ER. In some bacteria the single DNA molecule is attached to an invagination of the plasma membrane. Such an invagination in a very ancient procaryotic cell could have rearranged to form an envelope around the DNA, while still allowing the DNA access to the cell cytosol (as is required for DNA to direct protein synthesis). This envelope is presumed to have eventually pinched off completely from the plasma membrane, producing a nuclear compartment surrounded by a double membrane. As illustrated, the nuclear envelope is penetrated by communicating channels called nuclear pore complexes. Because it is surrounded by two membranes that are in continuity where they are penetrated by these pores, the nuclear compartment is topologically equivalent to the cytosol; in fact, during mitosis the nuclear contents mix with the cytosol. The lumen of the ER is continuous with the space between the inner and outer nuclear membranes and topologically equivalent to the extracellular space. (B) Mitochondria (and plastids) are thought to have originated when a bacterium was engulfed by a larger pre-eucaryotic cell. They retain their autonomy. This may explain why the lumens of these organelles remain isolated from the membrane traffic that interconnects the lumens of many other intracellular compartments. Figure 12-5 Topological relationships between compartments of the (more...) Figure 12-5 . Topological relationships between compartments of the secretory and endocytic pathways in a eucaryotic cell Topologically equivalent spaces are shown in red. In principle, cycles of membrane budding and fusion permit the lumen of any of these organelles to communicate with any other and with the cell exterior by means of transport vesicles. Blue arrows indicate the extensive network of outbound and inbound traffic routes, which we discuss in Chapter 13. Some organelles, most notably mitochondria and (in plant cells) plastids do not take part in this communication and are isolated from the traffic between organelles shown here. Other compartments in eucaryotic cells may have originated in a conceptually similar way (Figure 12-4A ). Pinching off of specialized intracellular membrane structures from the plasma membrane, for example, would create organelles with an interior that is topologically equivalent to the exterior of the cell. We shall see that this topological relationship holds for all of the organelles involved in the secretory and endocytic pathways, including the ER, Golgi apparatus, endosomes, and lysosomes. We can therefore think of all of these organelles as members of the same family. As we discuss in detail in the next chapter, their interiors communicate extensively with one another and with the outside of the cell via transport vesicles that bud off from one organelle and fuse with another (Figure 12-5 ). As described in Chapter 14, mitochondria and plastids differ from the other membrane-enclosed organelles in containing their own genomes. The nature of these genomes, and the close resemblance of the proteins in these organelles to those in some present-day bacteria, strongly suggest that mitochondria and plastids evolved from bacteria that were engulfed by other cells with which they initially lived in symbiosis (discussed in Chapters 1 and 14). According to the hypothetical scheme shown in Figure 12-4B , the inner membrane of mitochondria and plastids corresponds to the original plasma membrane of the bacterium, while the lumen of these organelles evolved from the bacterial cytosol. As might be expected from such an endocytic origin, these two organelles are surrounded by a double membrane, and they remain isolated from the extensive vesicular traffic that connects the interiors of most of the other membrane-enclosed organelles to each other and to the outside of the cell. The evolutionary scheme described above groups the intracellular compartments in eucaryotic cells into four distinct families: (1) the nucleus and the cytosol, which communicate through nuclear pore complexes and are thus topologically continuous (although functionally distinct); (2) all organelles that function in the secretory and endocytic pathways—including the ER, Golgi apparatus, endosomes, lysosomes, the numerous classes of transport intermediates such as transport vesicles, and possibly peroxisomes; (3) the mitochondria; and (4) the plastids (in plants only). Proteins Can Move Between Compartments in Different Ways All proteins begin being synthesized on ribosomes in the cytosol, except for the few that are synthesized on the ribosomes of mitochondria and plastids. Their subsequent fate depends on their amino acid sequence, which can contain sorting signals that direct their delivery to locations outside the cytosol. Most proteins do not have a sorting signal and consequently remain in the cytosol as permanent residents. Many others, however, have specific sorting signals that direct their transport from the cytosol into the nucleus, the ER, mitochondria, plastids, or peroxisomes; sorting signals can also direct the transport of proteins from the ER to other destinations in the cell. Figure 12-6 A simplified “roadmap” of protein traffic (more...) Figure 12-6 . A simplified “roadmap” of protein traffic Proteins can move from one compartment to another by gated transport (red), transmembrane transport (blue), or vesicular transport (green). The signals that direct a given protein's movement through the system, and thereby determine its eventual location in the cell, are contained in each protein's amino acid sequence. The journey begins with the synthesis of a protein on a ribosome in the cytosol and terminates when the final destination is reached. At each intermediate station (boxes), a decision is made as to whether the protein is to be retained in that compartment or transported further. In principle, a signal could be required for either retention in or exit from a compartment. We shall use this figure repeatedly as a guide throughout this chapter and the next, highlighting in color the particular pathway being discussed. To understand the general principles by which sorting signals operate, it is important to distinguish three fundamentally different ways by which proteins move from one compartment to another. These three mechanisms are described below, and their sites of action in the cell are outlined in Figure 12-6 . The first two mechanisms are detailed in this chapter, while the third (green arrows in Figure 12-6 the subject of Chapter 13. ) is Figure 12-7 Vesicle budding and fusion during vesicular transport (more...) Figure 12-7 . Vesicle budding and fusion during vesicular transport Transport vesicles bud from one compartment (donor) and fuse with another (target) compartment. In the process, soluble components (red dots) are transferred from lumen to lumen. Note that membrane is also transferred, and that the original orientation of both proteins and lipids in the donor-compartment membrane is preserved in the target-compartment membrane. Thus, membrane proteins retain their asymmetric orientation, with the same domains always facing the cytosol. 1 In gated transport, the protein traffic between the cytosol and nucleus occurs between topologically equivalent spaces, which are in continuity through the nuclear pore complexes. The nuclear pore complexes function as selective gates that actively transport specific macromolecules and macromolecular assemblies, although they also allow free diffusion of smaller molecules. 2 In transmembrane transport, membrane-bound protein translocators directly transport specific proteins across a membrane from the cytosol into a space that is topologically distinct. The transported protein molecule usually must unfold to snake through the translocator. The initial transport of selected proteins from the cytosol into the ER lumen or from the cytosol into mitochondria, for example, occurs in this way. 3 In vesicular transport, membrane-enclosed transport intermediates—which may be small, spherical transport vesicles or larger, irregularly shaped organelle fragments—ferry proteins from one compartment to another. The transport vesicles and fragments become loaded with a cargo of molecules derived from the lumen of one compartment as they pinch off from its membrane; they discharge their cargo into a second compartment by fusing with that compartment (Figure 12-7 ). The transfer of soluble proteins from the ER to the Golgi apparatus, for example, occurs in this way. Because the transported proteins do not cross a membrane, vesicular transport can move proteins only between compartments that are topologically equivalent (see Figure 12-5 discuss vesicular transport in detail in Chapter 13. ). We Each of the three modes of protein transfer is usually guided by sorting signals in the transported protein that are recognized by complementary receptor proteins. If a large protein is to be imported into the nucleus, for example, it must possess a sorting signal that is recognized by receptor proteins that guide it through the nuclear pore complex. If a protein is to be transferred directly across a membrane, it must possess a sorting signal that is recognized by the translocator in the membrane to be crossed. Likewise, if a protein is to be loaded into a certain type of vesicle or retained in certain organelles, its sorting signal must be recognized by a complementary receptor in the appropriate membrane. Signal Sequences and Signal Patches Direct Proteins to the Correct Cellular Address Figure 12-8 Two ways in which a sorting signal can be built into (more...) Figure 12-8 . Two ways in which a sorting signal can be built into a protein (A) The signal resides in a single discrete stretch of amino acid sequence, called a signal sequence, that is exposed in the folded protein. Signal sequences often occur at the end of the polypeptide chain (as shown), but they can also be located internally. (B) A signal patch can be formed by the juxtaposition of amino acids from regions that are physically separated before the protein folds (as shown). Alternatively, separate patches on the surface of the folded protein that are spaced a fixed distance apart can form the signal. There are at least two types of sorting signals in proteins. One type resides in a continuous stretch of amino acid sequence, typically 15–60 residues long. Some of these signal sequences are removed from the finished protein by specialized signal peptidases once the sorting process has been completed. The other type consists of a specific three-dimensional arrangement of atoms on the protein's surface that forms when the protein folds up. The amino acid residues that comprise this signal patch can be distant from one another in the linear amino acid sequence, and they generally persist in the finished protein (Figure 12-8 ). Signal sequences are used to direct proteins from the cytosol into the ER, mitochondria, chloroplasts, and peroxisomes, and they are also used to transport proteins from the nucleus to the cytosol and from the Golgi apparatus to the ER. The sorting signals that direct proteins into the nucleus from the cytosol can be either short signal sequences or longer sequences that are likely to fold into signal patches. Signal patches also direct newly synthesized degradative enzymes into lysosomes. Each signal sequence specifies a particular destination in the cell. Proteins destined for initial transfer to the ER usually have a signal sequence at their N terminus, which characteristically includes a sequence composed of about 5–10 hydrophobic amino acids. Many of these proteins will in turn pass from the ER to the Golgi apparatus, but those with a specific sequence of four amino acids at their C terminus are recognized as ER residents and are returned to the ER. Proteins destined for mitochondria have signal sequences of yet another type, in which positively charged amino acids alternate with hydrophobic ones. Finally, many proteins destined for peroxisomes have a signal peptide of three characteristic amino acids at their C terminus. Table 12-3 Some Typical Signal Sequences Table 12-3 Some Typical Signal Sequences Some specific signal sequences are presented in Table 12-3. The importance of each of these signal sequences for protein targeting has been shown by experiments in which the peptide is transferred from one protein to another by genetic engineering techniques. Placing the Nterminal ER signal sequence at the beginning of a cytosolic protein, for example, redirects the protein to the ER. Signal sequences are therefore both necessary and sufficient for protein targeting. Even though their amino acid sequences can vary greatly, the signal sequences of all proteins having the same destination are functionally interchangeable, and physical properties, such as hydrophobicity, often seem to be more important in the signal-recognition process than the exact amino acid sequence. Signal patches are far more difficult to analyze than signal sequences, so less is known about their structure. Because they often result from a complex three-dimensional protein-folding pattern, they cannot be easily transferred experimentally from one protein to another. Both types of sorting signals are recognized by complementary sorting receptors that guide proteins to their appropriate destination, where the receptors unload their cargo. The receptors function catalytically: after completing one round of targeting, they return to their point of origin to be reused. Most sorting receptors recognize classes of proteins rather than just an individual protein species. They therefore can be viewed as public transportation systems dedicated to delivering groups of components to their correct location in the cell. Panel 12-1 Approaches to Studying Signal Sequences and Protein (more...) The main ways of studying how proteins are directed from the cytosol to a specific compartment and how they are translocated across membranes are illustrated in Panel 12-1. Most Membrane-enclosed Organelles Cannot Be Constructed From Scratch: They Require Information in the Organelle Itself When a cell reproduces by division, it has to duplicate its membrane-enclosed organelles. In general, cells do this by enlarging the existing organelles by incorporating new molecules into them; the enlarged organelles then divide and are distributed to the two daughter cells. Thus, each daughter cell inherits from its mother a complete set of specialized cell membranes. This inheritance is essential because a cell could not make such membranes from scratch. If the ER were completely removed from a cell, for example, how could the cell reconstruct it? As we shall discuss later, the membrane proteins that define the ER and perform many of its functions are themselves products of the ER. A new ER could not be made without an existing ER or, at the very least, a membrane that specifically contains the protein translocators required to import selected proteins into the ER from the cytosol (including the ER-specific translocators themselves). The same is true for mitochondria, plastids, and peroxisomes (see Figure 12-6 ). Thus, it seems that the information required to construct a membrane-enclosed organelle does not reside exclusively in the DNA that specifies the organelle's proteins. Epigenetic information in the form of at least one distinct protein that preexists in the organelle membrane is also required, and this information is passed from parent cell to progeny cell in the form of the organelle itself. Presumably, such information is essential for the propagation of the cell's compartmental organization, just as the information in DNA is essential for the propagation of the cell's nucleotide and amino acid sequences. As we discuss in more detail in Chapter 13, however, the ER sheds a constant stream of membrane vesicles that incorporate only specific proteins and therefore have a different composition from the ER itself. Similarly, the plasma membrane constantly produces specialized endocytic vesicles. Thus, some membrane-enclosed compartments can form from other organelles and do not have to be inherited at cell division. Summary Eucaryotic cells contain intracellular membranes that enclose nearly half the cell's total volume in separate intracellular compartments called organelles. The main types of membrane-enclosed organelles present in all eucaryotic cells are the endoplasmic reticulum, Golgi apparatus, nucleus, mitochondria, lysosomes, endosomes, and peroxisomes; plant cells also contain plastids, such as chloroplasts. Each organelle contains a distinct set of proteins that mediate its unique functions. Each newly synthesized organelle protein must find its way from a ribosome in the cytosol, where it is made, to the organelle where it functions. It does so by following a specific pathway, guided by signals in its amino acid sequence that function as signal sequences or signal patches. Signal sequences and patches are recognized by complementary sorting receptors that deliver the protein to the appropriate target organelle. Proteins that function in the cytosol do not contain sorting signals and therefore remain there after they are synthesized. During cell division, organelles such as the ER and mitochondria are distributed intact to each daughter cell. These organelles contain information that is required for their construction so that they cannot be made from scratch.