Synthesis Booklet 2013 (1)
advertisement

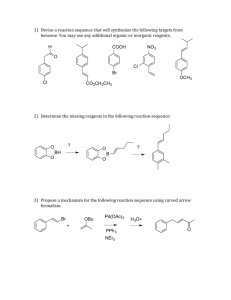
4.2.3 Synthesis Synthetic chemists use synthesis to make complex organic molecules from simpler organic molecules. A key difference between synthesis and the organic chemistry that you have met so far is that the product of one reaction is then used in another transformation. An understanding of how we change one functional group into another, in terms of reagents and conditions needed and sometimes mechanism, is key to successful synthesis.This is Casodex, a cancer drug launched in 1995 and first manufactured in Cheshire. Student activity 1 1. Name the functional groups shown below 2. Answer the following questions about the structure of casodex. (a) Circle the amide bond. Circle the nitrile/cyano group. Circle the alcohol. (b) What is the molecular formula?..................................... (c) Complete the structures of the carboxylic acid and amine that might be used to make this drug. What we need to be able to do at A2 is analyse reaction sequences involving familiar and unfamiliar molecules and apply our knowledge, with rigour. That way we can determine: conditions needed, reagents required, intermediates, starting molecules, and final product molecules. Here’s a synthetic sequence involving two reaction steps in which both the reagents, conditions and intermediates are shown. We need to bring together all the different reactions we know and see that we can use them to help us solve synthetic problems. Student Activity 2: Below is a list of transformations needed at A2 (they do not always represent reaction equations, as the product which is not the organic molecule has often been left out). Write next to them (or above the arrow) the reagents/conditions required for the conversions. Other reactions may be present on the exam. Now we can look at different ways of thinking about synthesis. Using flowcharts, molecular formula, structural, skeletal and displayed formula. Student activity 4 Draw a flow diagram starting from (a) propene and (b) benzene. Use a pencil, the page should be landscape orientation, include the organic structures but not the reagents, number each arrow. Student Activity 5: Deducing reagents/conditions in short reaction sequences Above/below each arrow, write the reagents/conditions required for the conversion 1. 2. 3. 4. 5. Student Activity 6: Deduce the intermediates. 4. Student Activity 7: Complete the flowcharts January 2012 AQA January 2012 Combining mechanistic knowledge and synthesis Calculating or using yields for organic reactions 1. Reactant to product- work out yield (Mod 4 collection). Page 23 June 02 2. Reactant to product- use yield to work out mass produced (Jan 06 Q4b-this has been used on a HW in 2013, so figures have been changed for the yield part). (b) Assuming a yield by mass of 24% for 4-nitrophenol, calculate the mass of 4-nitrophenol that would be produced from 120g of phenol. Show your working. Give your answer to an appropriate number of significant figures. 3. Reactant to product- use yield to work out mass of reactant needed (June 06 Q3c). This is much harder and has only come up once. Work out how many moles of product are needed, this equals the number of moles of reactant needed (in a one to one reaction). Then multiply by the inverse of the fraction eg. multiply by 100 over 40 if the reaction goes with 40% yield. Then multiply by the Mr of the reactant. 4. Impure substance to pure substance (recrystallisation of benzoic acid, normally on the coursework but not on the theory examination). In these situations there is no need to use moles as there is no reactant/product. 5. Limiting factor questions. If masses of two reactants are given then it may be necessary to calculate the number of moles of each, so that you can work out which is the limiting factor. If 0.5 moles of A is combined with 1 mole of B and the reacting ratio is 1+1 turns into 1 mole of product, then the maximum amount of product (theoretical amount) will be 0.5 moles. Appendix A Flow diagram for aliphatic (not arenes) compounds Student activity Write the reagents and conditions for the flow diagram. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 16. 17. 18.