Chemistry Final Review
advertisement

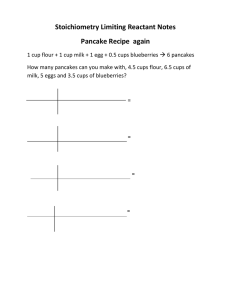
Chemistry Second Quarter Study Guide 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. Smith Convert: 34 m = ? cm 2 L = ? KL List the steps of the scientific method. What is a control? What is a variable? List our safety rules for labs. How do you weigh a fluid? How do you get the volume of an irregular object? List a way to separate a mixture? Put the following into scientific notation: 700m, 0.0054Kg, 5060 s How many significant figures for the following? 72.3 g, 6.20 g, 300m, 3450Km What is an element? What is a compound? What is a mixture? What are the parts of an atom? What is an ion? Cation? Anion? What is an isotope? What is an atomic mass unit? Give the electron configuration for the following: Ne, H, Cl, Mg Give the Lewis dot for the above elements. What is the Octet Rule? How many valence electrons make an atom stable? What is the electron configuration of these stable atoms? Who developed the first periodic table? How is it organized? What are metals, non-metals, noble gases, halides, alkali metals, alkali-earth, metals and metalloids? Know where to find them on the periodic table. Who were Dalton, Thomson and Rutherford? If we have never seen an electron, proton and neutron, how do we know they exist? Draw a Bohr model of oxygen and argon. Label all parts. What is an ionic, covalent and metallic bond? Name the following ionic compounds. MgO, NaBr, KOH, ClO Name the following covalent compounds. HBr, CO, CCl4 Draw a Lewis structure for CO2. What are the parts of a chemical reaction? Balance: H2 + Cl2--> HCl List the 5 basic types of chemical reactions. Give example of each. Write a balanced reaction for the following; Sodium hydroxide reacts with hydrochloric acid to form water and salt. What is the definition of a mole? If 1 smore = 2G + 1M + 1C, how many smores can I make if I have 5 of each reactant? Which is the limiting reactant? Convert the following to moles: 2 g H2, 25g Mg, 36g CO2 2H2 + O2 2H2O How many moles of water will be formed from 2 moles of oxygen and an excess of hydrogen? Show all work with a bca table. P.262 #37 abcd; use a bca table In water, what is the percent composition of hydrogen and oxygen? What is the difference between an empirical formula and a molecular formula? What is a limiting reactant? Excess reactant? What is percent yield? Given H2 + O2 H2O How many grams of water can be made from 20 grams of hydrogen? Why is the law of conservation of mass so important to chemistry?