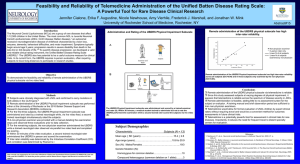
Batten Disease Support and Research Association RFP 2013 Title
advertisement

Batten Disease Support and Research Association RFP 2013 Title Research Grant URL for more info Sponsor Batten Disease Support and Research Association (BDSRA) Sponsor Type Amount Note Unspecified Upcoming Deadlines Date Certainty May 13, 2013 Anticipated Other Type Mandatory Invite Only? Yes No Note Proposals must be submitted by the deadline. Requirements Ph.D./M.D./Other Professional The BDSRA is pleased to announce that funds are available to conduct research directly relevant to all forms of Neuronal Ceroid Lipofuscinosis (NCL, Batten disease), rare, fatal, lysosomal neurodegenerative disorders that strike infants, children and adults. Research is needed across a broad spectrum addressing the underlying mechanisms of neural dysfunction and degeneration in each form of NCL from dysfunctional soluble lysosomal enzymes (PPT1, TPP1, CTSD), transmembrane proteins (CLN3, CLN6, MFSD8, CLN8) and other proteins implicated in lysosome function, membrane trafficking, or neurotransmitter release (CLN5, CSPα). Proposals will be considered for all forms of NCL, with emphasis on CLN1, CLN2, and CLN3. BDSRA also seeks to fund research projects in Batten disease that address immediate challenges of diagnosing and treating patients – epidemiological studies, natural history studies, medication management etc. Investigators are invited to submit proposals in the following areas or submit ideas of their own. BDSRA does not presently support proposals that fall into the category of social science, psychology or family services. 2. 3. Professional Society or Association The maximum award is $100,000 USD. Three-year or more proposals are generally discouraged unless there are extenuating circumstances or the study/project requires it. All grant award(s) must be used to cover direct costs of the project/ investigation and may not be used for indirect or overhead costs. All awards, including Postdoctoral fellowships, will be paid in six month increments following submission of progress reports for review. Citizenship or Unrestricted Residency Activity Location 1. http://www.bdsra.org/research.html Retrospective chart analyses. BDSRA seeks to fund a large-scale retrospective chart analysis in each form of Batten disease. This will be a key step in developing natural histories, informing clinical care, improving existing scales and may help identify potential biophysical markers of disease progression and/or treatment. Cardiomyocytes as comparative cell models to neurons in Batten disease. Recent reports highlight survivable cardiovascular changes in some forms of Batten disease. Because cardiomyocytes exhibit similar characteristics with neurons and withstand more laboratory manipulation, these cells may serve as an important parallel cell model for the primary defects in neurons while providing investigators with new tools and an accessible organ outside the blood brain barrier. Therefore, BDSRA will consider molecular and cellular investigations into the cardiovascular pathophysiology; neural control of cardiovascular function, cell transport and metabolism, cellular electrophysiology and ion channels, and excitation contraction coupling for the purpose of creating additional cell models that address the unsurvivable effects of Batten disease. Neuroscience/Neurobiology of Batten disease. Many questions in Batten disease beg investigation in neurobiology, 4. 5. 6. neurochemistry, neurophysiology, neuropharmacology, etc. BDSRA seeks proposals that answer key questions important for understanding disease pathogenesis and the brain’s response to disease. Drug Discovery. While many questions remain regarding the pathogenesis of Batten disease, BDSRA also seeks proposals that focus on target validation, development and testing of novel of high throughput screening assays, the testing of potentially disease-modifying lead compounds and medical chemistry on lead compounds. The goal of the association is to fund an unbroken and iterative spectrum of activity from basic discovery through clinical implementation. BDSRA seeks rigorous structure activity relationship (SAR) studies of small molecule leads in all forms of NCL. Preliminary evidence suggests that some compounds (e.g. Cystagon, Antiepileptics) produce desirable and undesirable effects in in vitro models, in vivo animal models or children with Batten disease. In some cases, the precise mechanism of action is unknown, unknown in relation to Batten disease, or unproven in humans. Therefore, BDSRA seeks to fund precise biochemical analyses to understand putative drug targets under normal and disease-state conditions. These studies should aim to create a plan for rational drug design, enhance bioavailability, or improve efficacy of promising compounds while reprioritizing others. (NB - Preliminary evidence suggests that Cystagon may be improved through rational design and/or combination therapy). Translation Support. We are in an exciting time in Batten disease research as the number of discoveries is increasing, their potential to be important drug targets is being explored, and the number of compounds available to evaluate for their potential to treat Batten disease is expanding. In order to take full-advantage of these opportunities to advance Batten disease research toward clinical trial success and commercial development, BDSRA will help investigators with promising results obtain resources for advancing their research beyond basic discovery. Examples include engaging FDA consultants to prepare for Investigational New Drug status, reaching out to research methodologists and statisticians to improve clinical trial designs, and locating commercial partners.*Please contact Dr Kerkovich (dkerkovich@beyondbatten.org) to discuss your ideas before attaching a free-form justification to administrative pages of the application form. Practice Guidelines for the management of Batten disease. BDSRA seeks to fund the development of practice guidelines for the diagnostic confirmation and management of individuals with Batten disease. Literature reviews, consensus statements created by a panel of international experts in clinical and laboratory diagnosis or treatment and management and endorsement from appropriate international professional societies such as the American Academy of Neurology, American Association of Neuroscience Nurses, etc. is required. These guidelines will serve as an educational resource for clinicians new to Batten disease. Therefore, it is expected that they will follow the Institute of Medicine’s Standards for Developing Trustworthy Clinical Practice Guidelines (http://www.iom.edu/Reports/2011/Clinical-Practice-Guidelines-We-Can-Trust/Standards.aspx) and be widely disseminated in the most appropriate form and forum. The authors will be provided with $5,000 to create practice guidelines based upon the current state of science and clinical care and $10,000 if their plans include the creation of an international working group dedicated to developing additional guidelines across multiple levels of care -- seizure management, occupational therapy, nutrition etc. .*Applicants must send an email request for consideration of their topic area to Dr Frazier (mfrazier@bdsra.org) for approval before attaching a free-form grant application to administrative pages of the application form. A note on research priorities: In addition to scientific merit, proposals are also scored for their potential significance to provide information important for the development of therapies to treat the primary effects of Batten disease. Applicants are encouraged to contact BDSRA for more information prior to developing their proposals. Keywords Lysosomal Storage Disease Neurodegenerative Disease Neurobiology Neurochemistry Drug Discovery High-throughput screening Metabolic Diseases Cardiovascular Practice Guidelines Cell Models Funding Type PhD Postdoctoral Award MD, PhD, or DO Independent Investigator Award MD, MD/PhD, or DO fellow enrolled or through the third year of training in neurology, pediatrics or other appropriate discipline Margie Frazier, PhD Executive Director | Batten Disease Support and Research Association 1175 Dublin Road Columbus, OH 43215 United StatesPhone: +1 (800) 448-4570/+1 (740) 927-4298 Sponsor mfrazier@bdsra.org Contact Info www.bdsra.org Danielle M Kerkovich, PhD Scientific Officer | Batten Disease Support and Research Association +1 202-812-6462 dkerkovich@beyondbatten.org . All proposals must be submitted electronically using BDSRA’s application form found on BDSRA’s website at http://www.bdsra.org/research.html and sent to mfrazier@bdsra.org AND dkerkovich@beyondbatten.org identified by the investigator’s last name. Recipients of awards from BDSRA, in addition to providing progress reports, are required to provide to BDSRA copies of any publications, abstracts, etc. resulting in whole or in part, directly or indirectly, from research funded by the award. Appropriate acknowledgment of BDSRA funding must be noted in such publications. In addition, if a recipient's research that has been funded by the BDSRA results in any new discovery or invention that may provide benefit to individuals with Neuronal Ceroid Lipofuscinosis (Batten disease), the BDSRA shall retain the right to use, as it deems appropriate, such discovery or invention for non-commercial research purposes. Furthermore, the recipient and/or the recipient's institution shall immediately notify the BDSRA, if BDSRA funds have supported research that results in the recipient and/or the recipient's institution seeking to make any commercial gain, patent application or secure any other proprietary rights to legally protect any discovery or invention resulting, in whole or in part, directly or indirectly, from the research. The BDSRA shall be entitled to an equitable share of any patent, proprietary right and/or commercial gain that accrue to the recipient and/or the recipient's institution as a result, either in whole or in part, directly or indirectly, of BDSRA funding. The BDSRA's equitable share shall be based upon the level of contribution the BDSRA funding had on the research. BDSRA requires models, cells and mice developed with BDSRA funds be openly shared with the scientific community upon description of their development or use in a peer-reviewed publication. As a condition of funding, resources must be placed in a repository and/or the commercial sector for their continued development, management and dissemination. Applicants are encouraged to contact BDSRA with questions associated with this requirement or any other issue with respect to funding.