Review Worksheet #1
advertisement

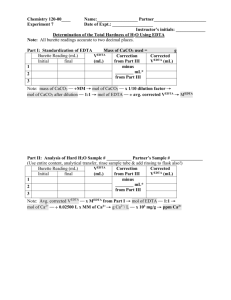
CHE 230 Review Worksheet – Exam #1 Name ____________________ 1. Standard and sample preparation a. Describe how to prepare 100 mL of a 2.0 M Fe3+ stock solution from iron (III) nitrate solid. b. You need to prepare standard from the stock solution (in part a) in the linear range from 0.010 M to 0.050 M Fe3+. Describe how to make 25.0 mL of the lowest and highest standard. c. An iron ore is suspected to contain 10% (w/w) iron (III). If you dissolved 0.25 g of the ore and diluted it to volume in a 250.0-mL volumetric flask, would the sample likely be in the linear range (see part b)? If not, propose a way to may it “in range”. 2. Conversions a. The LD-50 for caffeine is 10 g by oral digestion. A 12 oz can of Mt. Dew contains 55 mg of caffeine. How many 2-L bottles of Mt. Dew would you have to consume to reach the LD-50? b. One cup of low fat milk contains 30% of the recommended daily allowance of calcium. The RDA for calcium is 1000 mg. How many cups of milk should a person drink to get sufficient calcium? How many mg of calcium are contained in one cup of milk? c. Tea contains antioxidant compounds called flavonoids. Tea allowed to brew 40-60 s contains approximately 140 mg of flavonoids. Let’s assume that the 60 s extraction is 40% effective in pulling out the flavonoids. How many mg of flavonoids naturally occur in one tea bag? If one tea bag contains 2.0 g of tea, what is the concentration of favonoids in ppm (ug/g) in the tea (prior to brewing)? 3. Statistics a. The blood alcohol content of a DUI suspect was analyzed in quadruplicate by GC. The results are provided below. Based on statistics, is this driver legally drunk (0.08)? Explain. 0.083 0.067 0.071 0.065 b. The density of 10 pennies (post 1982) was determined. The following results were obtained: 6.90, 6.52, 7.26, 6.88, 7.03, 6.74, 6.79, 6.24, 6.37, 6.54 (g/cm3). Calculate the 95% confidence interval for these results. Comment on the accuracy and precision of the results. (After 1982, pennies were 97.6% zinc and 2.4% copper and had a density of 6.94 g/cm3.) c. The intoxilyzer is also used to test DUI suspects. The same person was tested by GC (the courtapproved method) and by the intoxilyzer five times. The results are provided below. Use a bar graph to explain whether the results from the intoxilyzer should stand up in court. Trial 1 2 3 4 5 GC Intoxilyzer 0.052 0.055 0.049 0.048 0.051 0.045 0.046 0.042 0.50 0.044 4. Titration a. For the complexometric titration of Ni2+ with EDTA, 0.537 g of nickel chloride was dissolved in 50.0 mL of water. How much volume of a 0.35 M EDTA solution should be required to reach the end point? b. A 50.0 mL sample containing Ni2+ was treated with 25.0 mL of 0.0500M EDTA to complex all the Ni2+ and leave excess EDTA in solution. The excess EDTA was then back-titrated, required 5.00 mL of 0.0500 M Zn2+. What was the concentration of Ni2+ in the original solution? (EDTA and metals complex in a 1:1 molar ratio.)