Hardness of Water - Sites at Penn State
advertisement

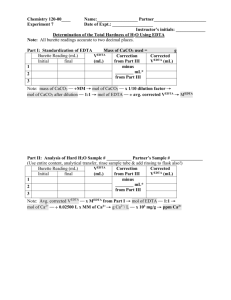
Introduction Water hardness is based on the amount of concentrated dissolved minerals that are cations of charge 2+. There are many minerals that are dissolved in the water, but the two main minerals that are typically measured in the water are Ca2+ and Mg2+. The harder the water is the more 2+ cations are present. The reason that the water hardness is important is because hard water creates a residue and scaling that is very hard and expensive to remove. The harder the water is the more soap is needed to clean dishes, cloths, etc.1. This could cause a lot of trouble for business that use water in their various products. For example a beer brewing company has large boilers that boil water to a certain degree, if the water is really hard the minerals in the water will create scaling that causes big problems when brewing beer. This problem can be ratified with water softening agents. Water hardness can be quantified is several different ways; milligrams per liter (mg/l), parts per million (ppm), and grains per gallon (gpg). There are two main way to determine what the hardness of a water sample. The first that is going to be talked about is atomic absorption spectrophotometry (AA). The second is EDTA titration which will be talked about later on in the document. AA uses an instrument called a spectrophotometer that can measure the concentration of a certain amount of mineral. The way this instrument works is a light is emitted from one end of the machine that is the spectrum of either Mg or Ca, since these are the minerals measured in water to determine hardness. It passes through a flame into a monochrometer. The flame is an air-acetylene mixture that has within it an aerosol sample of the water sample 11. This atomizes the metal cations Mg2+ and Ca2+. The light from the lamp passes through the flame and absorbs 1|Page only the matching electronic energy levels as the lamp used. Say the lamp is a Ca lamp the light will go through the flame that the monochrometer will pick up only that wavelength. The wavelength is then bounced from mirror to grating to a PMT where the concentration is measures. EDTA titration is done by taking a sample of water, with Mg2+, and mixing it with a EBT indicator. This will turn the water maroon because the indicator reacts with Mg2+ and not Ca2+. To determine the hardness the EDTA solution is added, which reacts with Ca2+ creating a chelate. Once the EDTA starts to react with the Mg it will begin to return to the normal blue color. The point at which the color changes from maroon to blue is the titration point and the hardness is calculated by using the equation MEDTA VEDTA = MCa2+ VCa2+. The reason two methods are used to calculate water hardness is because the EDTA titration accommodates for the concentration of all the 2+ cations that are in the water solution, while the AA only accommodates for the Mg2+ and the Ca2+ concentrations. Both tests are conducted so there is no bias to either concentration. Zachary Partner 1 Partner 2 Partner 3 Sample of State City River Pond River Tap PA NJ CA MD Emmaus Washington Janul Gathersburg Table 1: Where the water samples came from Table one shows where the water samples that were used in the experiment were taken from and the location of their source. According to USGS Pennsylvania has generally soft water of about 61 – 120 mg/l, Maryland and New Jersey had the softest water at around 0-60 mg/l, and 2|Page southern California had the hardest of 181 – 250 mg/l 2. After seeing this information a conclusion was determined that partner 1 and 3 would have the softest water, and partner 3 having the softest water because it is from the tap. Zachary would have the third softest water and partner 3 would have the hardest water. Procedure First the hardness of the water was determined using AA. The samples of water were collected, filtered and diluted if necessary, and put through the spectrophotometer to determine the concentrations of Ca2+ and Mg2+. Next a test was done to observe the amount of total dissolved solids that were in the water compared to two given samples. The three samples were the water brought by the student, distilled water, and 1x10-3M Ca2+. Then the titration was practiced on substances with known amounts of Ca2+ and Mg2+. This was used as practice for the student before he/she actually started EDTA titration on their water samples. Next the actual EDTA titration was done on the students’ samples to determine the water’s hardness. After the EDTA titration was done twice on the water samples two different types of water softening tests were done on the samples. First was the commercial waterconditioning agent was used, Arm & Hammer NaHCO3. After a viable solution was prepared EDTA titration was done to determine the new hardness of the water sample. The second softening procedure done was the cation exchange. 3|Page Results Absorbance Value (@ 422.7 nm) Concentration vs Absorbance of Ca2+ y = 0.0096x + 0.014 R² = 0.9959 0.60000 0.50000 0.40000 0.30000 0.20000 0.10000 0.00000 0.00 10.00 20.00 30.00 40.00 50.00 60.00 Concentration (ppm) Figure 1: Calibration of the Calcium spectrophotometer Absorbance Value (@ 202.5 nm) Concentration vs Absorbance of Mg2+ y = 0.0143x + 0.0197 R² = 0.9984 0.5 0.45 0.4 0.35 0.3 0.25 0.2 0.15 0.1 0.05 0 0.00 5.00 10.00 15.00 20.00 25.00 30.00 35.00 Concentration (ppm) Figure 2: Calibration of the Magnesium spectrophotometer 4|Page Zachary Partner 1 Partner2 Partner3 Ca2+ Mg2+ Hardness Ca2+ Hardness Mg2+ Total Hardness 19.86 18.59 60.63 27.78 12.03 2.01 50.96 6.83 49.66 46.48 151.56 69.45 49.53 8.26 209.70 28.09 99.19 54.74 361.27 97.54 Table 2: Calculated hardness using AA in ppm Zachary Partner 1 Partner2 Partner3 MEDTA VEDTA VCa2+ MCa2+ 0.0002 0.0002 0.0002 0.0002 7 5 5 9 1 1 1 1 0.0014 0.001 0.001 0.0018 Table 3: Calculated MCa2+ using the equation M1*V1 = M2*V2 Zachary Partner 1 Partner2 Partner3 MCa2+ Concentration in ppm 0.0014 0.001 0.001 0.0018 140 100 100 180 Concentration in grains per gallon 8.18713 5.84795 5.84795 10.5263 Table 4: Calculated concentration un softened in ppm and grain per gallon Zachary Partner 1 Partner2 Partner3 MEDTA VEDTA VCa2+ MCa2+ 0.0002 0.0002 0.0002 0.0002 5 2 3 4 1 1 1 1 0.001 0.0004 0.0006 0.0008 Table 5: Calculated MCa2+ from the Arm & Hammer Baking Soda used to soften the water samples 5|Page Zachary Partner 1 Partner2 Partner3 MCa2+ Concentration in ppm 0.001 0.0004 0.0006 0.0008 100 40 60 80 Concentration in grains per gallon 5.847953 2.339181 3.508772 4.678363 Table 6: Concentrations that occurred after the Arm & Hammer baking soda was used to soften the water samples Zachary Partner 1 Partner2 Partner3 MEDTA VEDTA VCa2+ MCa2+ 0.0002 0.0002 0.0002 0.0002 1 1 1 3 1 1 1 1 0.0002 0.0002 0.0002 0.0006 Table 7: Calculated MCa2+ from the Resin ion exchange used to soften the water samples note for all the VEDTA that are equal to 1 were actually less than one Zachary Partner 1 Partner2 Partner3 MCa2+ Concentration in ppm 0.0002 0.0002 0.0002 0.0006 20 20 20 60 Concentration in grains per gallon 1.169591 1.169591 1.169591 3.508772 Table 8: Concentrations that occurred after the Resin ion exchange was used to soften the water samples 6|Page Dilution for AA AA Ca Absorbance AA Mg Abosorbance Dilution for EDTA EDTA Unsoften Drops EDTA Baking Soda Softened Drops EDTA Resin Softened Drops pH Before Resin pH After Resin TBS Observation Zachary Partner 1 Partner 2 Partner 3 n/a 0.2047 0.1918 n/a 7 5 n/a 0.1925 0.0484 n/a 5 2 1:4 0.596 0.7484 1:4 5 3 n/a 0.2807 0.1173 n/a 9 4 <1 8 3 Sample had a larger ring with more white residue in a larger ring 1 2 3 Sample had less white residue in a larger ring <1 7 2 Sample had a thicker white residue in a larger ring 3 2.50 2 Sample had more white residue but in a smaller ring Table 9: Overview of all observations taken form Chem 111 Labratory Notebooks of Katie Horneff, Luke Hess, Patrick Huang, and Zachary Horan 7 8 9 10 The equation below is to change the hardness taken from the AA machine that is in ppm and change it to ppm hardness. 100 g CaCO3 19.86 ppm Ca2+ × [ 1 mole2+ ] = 49.65 ppm CaCO3 = 49.65 ppm hardness 40.0 g Ca 1 mole The equation below is used to determine the molarity of the water solution; it was used he Table 3 to determine MCa2+. MEDTA VEDTA = MCa2+ VCa2+ → MCa2+ = MEDTA VEDTA VCa2+ 7|Page The equation below is for determining the concentration of CaCO3 from EDTA titration; it was used to determine the concentration in Table 4. 1.4 × 10−3 moles CaCO3 100 g CaCO3 1000 mg CaCO3 140 mg CaCO3 × × = 1 liter of solution 1 moleCaCO3 1 g CaCO3 1 liter of solution = 140 ppm The equation below is to convert the hardness of ppm to grains per gallon; it was used in Table 4 to determine the concentration in grains per gallon 140 ppm × 1 grain per gallon = 8.19 grains per gallon 17.1 ppm Equations derived from PSU Chemtrek Discussion While using AA to determine the water hardness it was discovered that the softest water came from Partner 1, the second softest came from Partner 3, the third softest came from Zachary, and the hardest came from Partner 2. According to the hypothesis the only part that was wrong was that Partner 3 had harder water than Partner 1. As stated above it was expected that the tap water would be the hardest but the pond water from New Jersey ended up being the softest. After further research is was discovered that since the tap water travels through all sorts of pipes it can pick up bits of Ca2+ and Mg2+ that will make the water harder, so in this case the tap water might be harder that the pond3. The pond water is fairly stagnant which would not bring much of any metals into the pond than whatever might run into it during a rain storm so it is now relevant that the pond should have the softest water. The hypothesis stated that the partners 1 and 3 would 8|Page have the softest which they did, and that partner 2 would have the hardest and Zachary would have the second hardest. The rest of this hypothesis was correct. When looking at the EDTA titration of the water samples the findings were completely different. The order from softest to hardest is now Partner 1, Partner 2, Zachary, Partner 3. This is completely different, and the hardness are higher on average. Since AA only measures the Ca2+ and Mg2+ it would read a lower overall hardness, but the EDTA measures all 2+ cations. This could bring the hardness level up which would cause the data to differ. The way to measure the EDTA titration is also not the most precise way because the VEDTA has to be a whole number and it is taken from observation alone, when the experimenter thinks the color in the wells change the right amount. When the water samples were softened with the Arm & Hammer it changed the hardness order by one person the order, from soft to hard, is now; Partner1, Partner2, Partner3, and Zachary. The softening agent softened the water about 40 -80 ppm, and brings all the samples into the moderately hard water category according to the department of environmental quality 4. The Resin softened water softened the water so much that it was unable to determine the exact hardness of the water sample. While using the EDTA titration the very first well was turned to blue meaning that the hardness could be 20 ppm or it could be less. This would mean that the VEDTA could equal one drop or it could be less but since the equipment is not scaled small enough it has to be assumed that the softness is less than 20 ppm. This type of water softener brings all the water samples down to soft water according to mecc.edu5. The AA technique is more accurate because it measures just the Ca2+ and Mg2+ that is in the water and can measure a smaller amount to get a more precise reading. The error in the AA 9|Page technique is much smaller than the error in the EDTA titration. In the EDTA titration one missed drop or a piece of sediment in the sample could give the wrong reading. If the water sample was not filtered are diluted right it could give the wrong readings. Also the pipet used could be defective and not be dropping the same size drops every time. All these are factors that could go into giving a false or wrong reading of the hardness. According to the Allentownpa.gov the water sample of Zachary was a little softer that the average, the site states that the average is about 14.2 grains per gallon where Zachary’s was 8.17 grains per gallon 6. This could be due to that fact that the water sample was taken from a slightly higher elevation than Allentown because it was taken on the outskirts of Allentown. Conclusion The hypothesis was generally correct for the AA but differed when it came to the EDTA titration as can be seen in table 2 and table 4. The difference in the records for hardness using the AA verses the EDTA titration was unexpected, and could possibly be redone to confirm conclusions, but the fact that all the partners’ readings were all off would lead one to believe that it is probably not just coincidence. The hardest water ended up coming from Janul, California out of a river and the softest came from a pond in Washington, New Jersey. In conclusion the north coasts of the US contain softer water where the southern coasts and central US contain harder water. 10 | P a g e References 1. “Web Site Title” http://www.fcwa.org/water/hardness.htm 2. “Web Site Title” http://water.usgs.gov/owq/hardness-alkalinity.html 3. “Web Site Title” http://www.mwra.state.ma.us/04water/html/qual4concerns.htm 4. “Web Site Title” http://www.gallatin.mt.gov/Public_Documents/GallatinCoMT_WQDFactSheets/WaterSo ftenersSepticsGW.pdf 5. “Web Site Title” http://water.mecc.edu/exam_prep/hardness.html 6. “Web Site Title” http://www.allentownpa.gov/Portals/0/files/Water/2009finalCCR.pdf 7. Hess, Luke, Chem 111 Laboratory Notebook 8. Horan, Zachary, Chem 111 Laboratory Notebook 9. Horneff, Katie, Chem 111 Laboratory Notebook 10. Huang, Patrick, Chem 111 Laboratory Notebook 11. PSU Chemtrek Small-Scale Experiments for General Chemistry by Stephen Thompson Hayden McNeil, Englewood Cliffs, NJ 2012-2013, pgs 10.1 – 10.22 12. Chemistry 111 Student Packet Department of Chemistry Penn State by Joseph T. Keiser Hayden McNeil, USA, 2012-2013, pgs 59 - 62 11 | P a g e