Receipt of Controlled Drugs Ref 8
advertisement

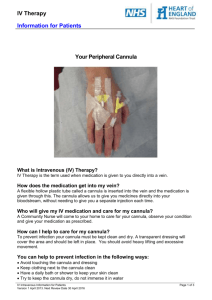
Peripheral IV Cannula Audit Tool (Reference: CDHB Vol 10 and 12) Methodology: Real time observational audit of staff engaged in managing the cannula of a patient who has had the cannula in for over 8hrs plus documentation audit Location of Audit: Month finished: Auditors Name/Designation: Use Y (Yes), N (No) or NA (not applicable) below Section 1 Q 1 Hand hygiene performed prior to accessing cannula Q 2 Asepsis maintained throughout procedure Q 3 Cannula dressed and secured as per policy Q 4 Completed Green ID label placed on dressing Q 5 Completed Green ID label placed in clinical notes Q 6 Extension set attached Q 7 Phlebitis score documented at least 8hrly Q 8 Cannula flushed at least 8hrly Q 9 The cannula is within its date of expiry (i.e. not expired) 1 2 3 4 5 6 7 8 9 10 TOTALS Yes No NA Comments for all audit questions Entered in database by: Date entered: