S06.Chromo
advertisement

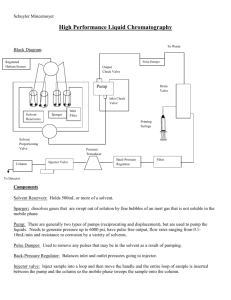
Lecture #6 HPLC HPLC have mobile phase , column , pump and detector . HPLC (High Pressure Liquid Cromatography) → maen that I have pump . HPLC (High Performance Liquid Chromatgraphy) → mean that give good resolution of the peak . HPLC mobile phase needs : HPLC good solvent which contain aqueous part and organic modifier (Why?) Because here we use reverse phase (not normal, polar→water ) .. but if mobile phase all of it is polar and the column is non-polar there will be no competition ,so we use organic modifier (which means :water soluble organic solvent , semi-polar) like: ethanol , methanol , acetonitrate and tetra hydrochloric . Acetonitrate and methanol are most commonly used after (microfiltration) .. Methanol manufacturing : We use sugar source like grabs and bacteria to make fermentation and convert the glucose to ethanol (96% ethanol) why? Because alcohol always have a 4% of water during distillation process . To have 100% ehanol they make isosbestic distillation by adding benzene to ethanol with make eutectic mix then benzene and ethanol evaporate together leaving water , then separation ethanol from benzene by simple distillation to have absolute ethanol , but it is expensive (high purity ) Eutectic mix : when we mix 2 substances together , it will cause boiling point elevation or melting point depression. HPLC code : Solvent should be free from insoluble impurities to avoid scattering of UV , AND free of soluble impurities to avoid chromophore impurities which may lead to interference with UV absorbtion . Cut-off value : ↓λ ↑energy Higher energy → absorbtion of α bond Lower energy → absorbtion of π bond Methanol should be transparent on π region (low energy) to prevent making inteferance. At (blue) higher absorbtion ,, it means that the methanol absorbtion is higher than analyte Blank sample → low absorbance ,, if it was high absorbance analyte will not be clear. if mobile phase (blank) →high absorbance at low energy and will make excitation of analyte .. analyte won't be clear methanol absorbtion at low λ → due to impurities cut-off value → λ → better when it lower so we need solvent with high purity (free of soluble and insoluble impurities ) and we can relay on cut-off value .